Treatment options in metastatic colorectal cancer (mCRC) become increasingly limited once the disease progresses after standard therapies. In this setting, clinicians often face a difficult question: how can they extend disease control when conventional lines of treatment have already been exhausted? One promising strategy has been the re-use of anti-EGFR therapy in carefully selected patients, particularly when resistance-associated alterations are no longer detectable in circulating tumor DNA (ctDNA). The PARERE study was designed to address this question in a randomized setting and to clarify how panitumumab re-treatment should be positioned relative to regorafenib in chemorefractory disease.
The study was published as an Original Article in Annals of Oncology in January 2026.
Title: Re-treatment with panitumumab followed by regorafenib versus the reverse sequence in chemorefractory metastatic colorectal cancer patients with RAS and BRAF wild-type circulating tumor DNA: the PARERE study by GONO
Authors: P. Ciracì, M.M. Germani, F. Pietrantonio, P. Manca, S. Lonardi, A. Busico, F. Bergamo, V. Burgio, F. Mannavola, S. Di Donato, E. Fenocchio, F. Palermo, I. Capone, M.C. De Grandis, N. Pella, M. Scartozzi, L. Antonuzzo, A. Passardi, M. Claravezza, L. Salvatore, S. Tamberi, G. Randon, E. Conca, V. Conca, C. Antoniotti, R. Moretto, G. Masi, L. Boni, D. Rossini, and C. Cremolini, on behalf of the GONO Foundation Investigators.
Read about Colon Cancer Success Rate on OncoDaily.
Methods
PARERE (NCT04787341) was an open-label, multicenter, randomized phase II trial conducted across 37 Italian oncology units. The trial enrolled patients with histologically confirmed RAS and BRAF wild-type mCRC, ECOG performance status of 0–1, and prior exposure to fluoropyrimidines, oxaliplatin, irinotecan, antiangiogenic therapy, and first-line anti-EGFR-based treatment. To enter the study, patients also had to have experienced benefit from prior first-line anti-EGFR therapy, defined as response or stable disease for at least 6 months, followed by at least one intervening anti-EGFR-free line and an anti-EGFR-free interval of at least 4 months.
A key feature of the study was prospective molecular selection using liquid biopsy. Only patients without RAS mutations or BRAF-V600E mutations in ctDNA were eligible for randomization. Patients were then assigned 1:1 to one of two strategies: arm A, panitumumab followed by regorafenib after progression, or arm B, regorafenib followed by panitumumab after progression.
Primary and Secondary Endpoints
The primary endpoint was overall survival (OS). Secondary endpoints included first and second progression-free survival (PFS), first and second objective response rate (ORR), first and second disease control rate (DCR), and safety.
Results
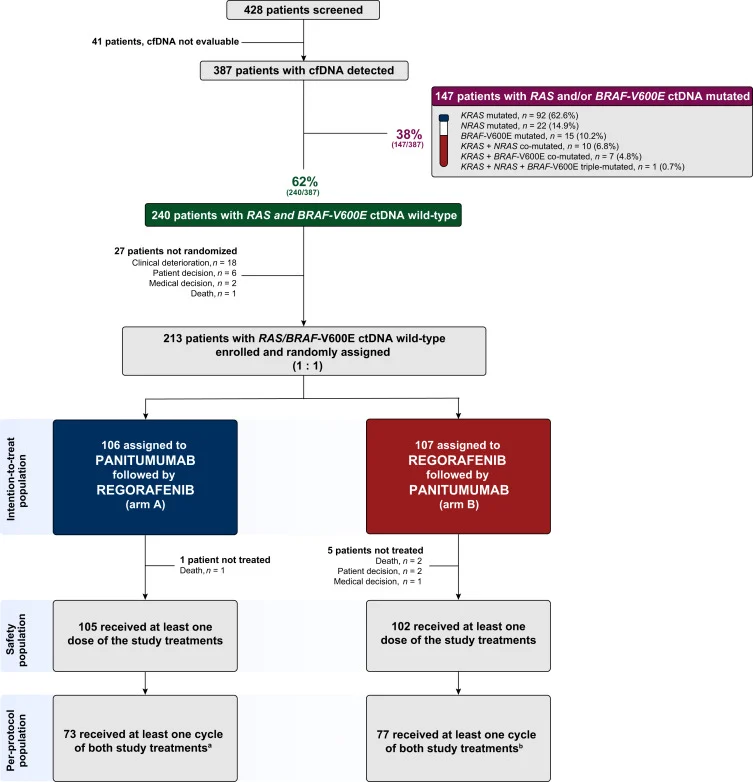
Between December 2020 and December 2024, 428 patients underwent molecular screening. Cell-free DNA was evaluable in 387 cases (90.4%), with a median turnaround time of 12 days. Among these 387 screened patients, 147 (38.0%) were found to harbor RAS and/or BRAF-V600E mutations in ctDNA and were therefore excluded from randomization. This is an important finding in itself, showing that liquid biopsy excluded more than one-third of clinically eligible patients from anti-EGFR re-treatment on molecular grounds.
The most common alterations detected were KRAS and NRAS codon 61 mutations. Compared with patients who remained ctDNA wild-type, those with RAS/BRAF-V600E-mutant ctDNA were more likely to have progressed during first-line anti-EGFR treatment or within 3 months of the last anti-EGFR dose (74.2% vs 60.4%, P = 0.006) and had a shorter anti-EGFR-free interval (11.2 vs 14.3 months, P = 0.004). They were also more likely to harbor co-mutations in MAP2K1 (13.6% vs 2.5%, P < 0.001) and EGFR (11.6% vs 1.7%, P < 0.001).
Patient population
Of the 240 ctDNA wild-type patients, 213 (88.8%) were randomized: 106 to arm A and 107 to arm B. The median age was 62 years, and most patients had ECOG 0, left-sided primary tumors, multiple metastatic sites, and had received a median of two prior lines of therapy. Among tumors with known status, 3 patients (1.5%) were MSI-high/dMMR, and 6 patients (5.0%) were HER2-positive.
Overall survival
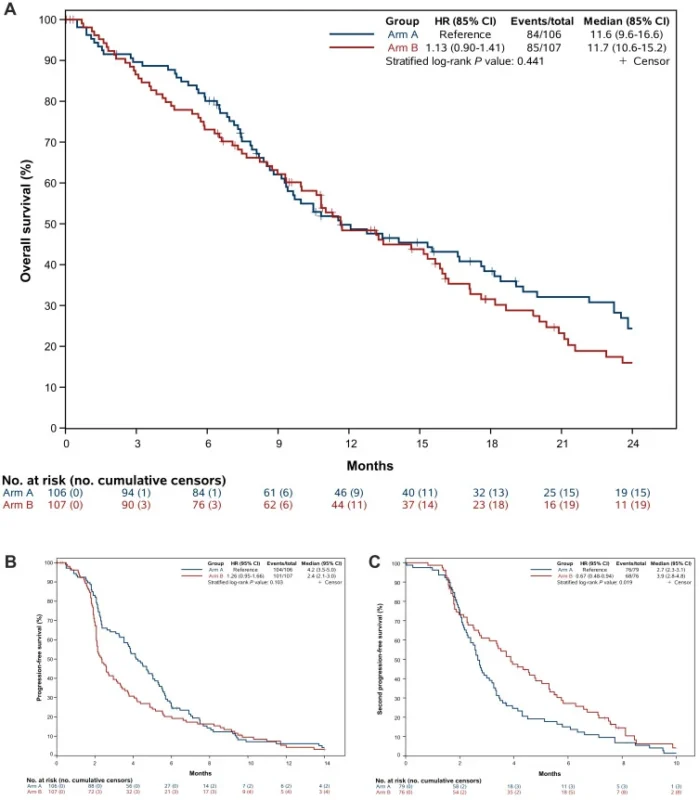
At a median follow-up of 31.9 months, 169 overall survival events were recorded. The study did not demonstrate a difference in overall survival between treatment sequences. Median OS was 11.6 months in the panitumumab-first arm and 11.7 months in the regorafenib-first arm (HR 1.13; 85% CI 0.90–1.41; P = 0.441). Similar results were observed in the per-protocol population. These findings suggest that the order in which the two treatments were administered did not significantly affect overall survival.
Treatment activity and progression-free survival
Although overall survival was similar between the two strategies, clear differences emerged in tumor response and disease control, which consistently favored panitumumab over regorafenib.
When used as the first treatment, panitumumab demonstrated higher activity:
- Objective response rate: 16% with panitumumab vs 2% with regorafenib (P = 0.003)
- Disease control rate: 61% vs 36% (P < 0.001)
- Median PFS: 4.2 vs 2.4 months
A similar pattern was observed after disease progression when the treatments were crossed over. Patients receiving panitumumab as second treatment achieved:
- ORR: 18% vs 0% with regorafenib (P = 0.013)
- DCR: 62% vs 38% (P = 0.003)
- Median second PFS: 3.9 vs 2.7 months (P = 0.019)
These results indicate that anti-EGFR re-treatment provides greater tumor activity than regorafenib, regardless of treatment sequence.
Molecular hyperselection
Exploratory analyses investigated whether additional genomic alterations detected in ctDNA might influence response to anti-EGFR re-treatment. Among 212 randomized patients with complete coverage of relevant genes, 19 patients (9.0%) had at least one alteration potentially associated with anti-EGFR resistance, including alterations in AKT1, MAP2K1, EGFR, ERBB2, non-BRAF-V600E class I or II, and PIK3CA exon 20.
In this gene-altered ctDNA subgroup, there was no clear efficacy difference between panitumumab and regorafenib. By contrast, the benefit of panitumumab over regorafenib was more evident in the ctDNA-negative hyperselected population. Likewise, in the subgroup with known MMR-proficient/microsatellite-stable and HER2-negative tumors, efficacy results were consistent with those seen in the intention-to-treat population.
Safety
Safety findings also distinguished the two agents. Grade 3–4 events of any cause occurred in 43.0% of patients in arm A and 51.0% in arm B. Panitumumab was more often associated with grade ≥3 acneiform rash than regorafenib, both as first treatment (19.2% vs 2.9%, P < 0.001) and as second treatment (13.7% vs 0%, P = 0.002).
In contrast, regorafenib was associated with more grade ≥3 hypertension (13.7% vs 1.0% as first treatment; 10% vs 0% as second treatment), fatigue (9.8% vs 2.9% as first treatment), and hand–foot skin reaction (5.9% vs 0% as first treatment). Most patients started regorafenib at reduced doses, and only about a quarter escalated to the full 160 mg/day dose.
Conclusion
The PARERE study did not identify a superior treatment sequence in terms of overall survival, but it did provide strong randomized evidence that panitumumab re-treatment is more active than regorafenib in chemorefractory RAS/BRAF ctDNA wild-type mCRC. Across both treatment lines, panitumumab consistently achieved higher response rates, higher disease control rates, and longer progression-free survival, with a generally manageable safety profile.
Just as importantly, PARERE highlights the clinical value of liquid biopsy-guided selection, showing that 38% of screened patients carried ctDNA resistance mutations and would likely not have benefited from anti-EGFR re-treatment. The exploratory findings further suggest that deeper molecular hyperselection may refine patient selection even more, and that patients with an anti-EGFR-free interval of 6 months or less may be less suitable candidates for this approach.
Overall, PARERE supports anti-EGFR re-treatment as a meaningful option in the continuum of care for chemorefractory mCRC, particularly when tumor shrinkage is needed and ctDNA confirms the absence of acquired resistance alterations.
The full article is available in Annals of Oncology.