A major international clinical trial, NRG-LU005, has provided important new insight into how immunotherapy should be used in limited-stage small cell lung cancer (SCLC).
While immune checkpoint inhibitors have significantly improved outcomes in extensive-stage SCLC, it has remained unclear whether adding immunotherapy earlier—during potentially curative treatment—could further improve survival.
The LU005 trial set out to answer this question by testing whether adding the PD-L1 inhibitor atezolizumab to standard chemoradiotherapy could improve outcomes in patients with limited-stage disease.
Study design
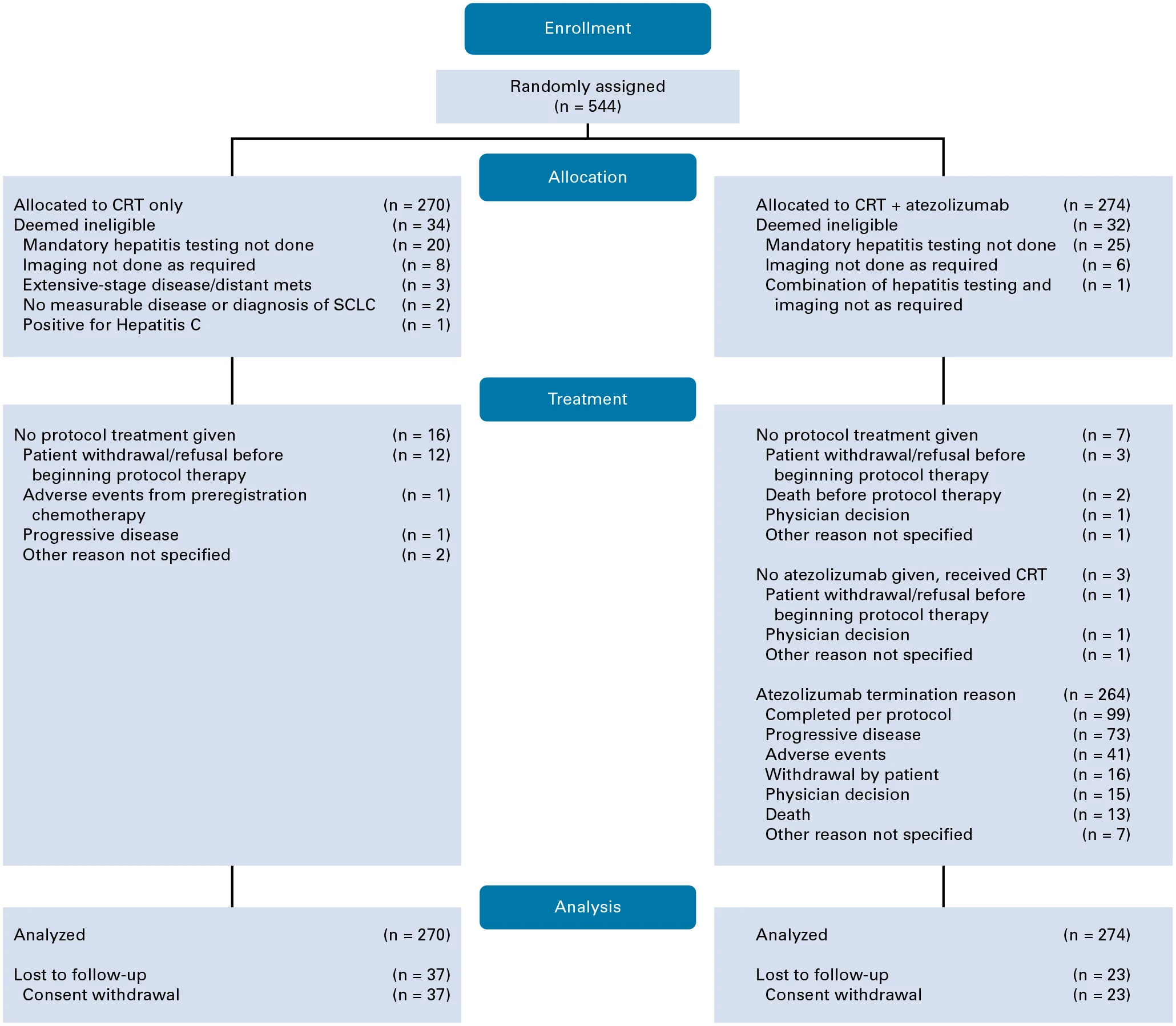
The phase III trial enrolled 544 patients with limited-stage SCLC across 218 centers in the United States and Japan.
All patients received standard concurrent chemoradiation, consisting of:
- platinum chemotherapy (cisplatin or carboplatin)
- etoposide
- thoracic radiation therapy
Participants were then randomized to receive either:
- Chemoradiation alone, or
- Chemoradiation plus atezolizumab, given every three weeks during and after treatment.
The primary goal of the study was to determine whether adding atezolizumab could improve overall survival.
Key results
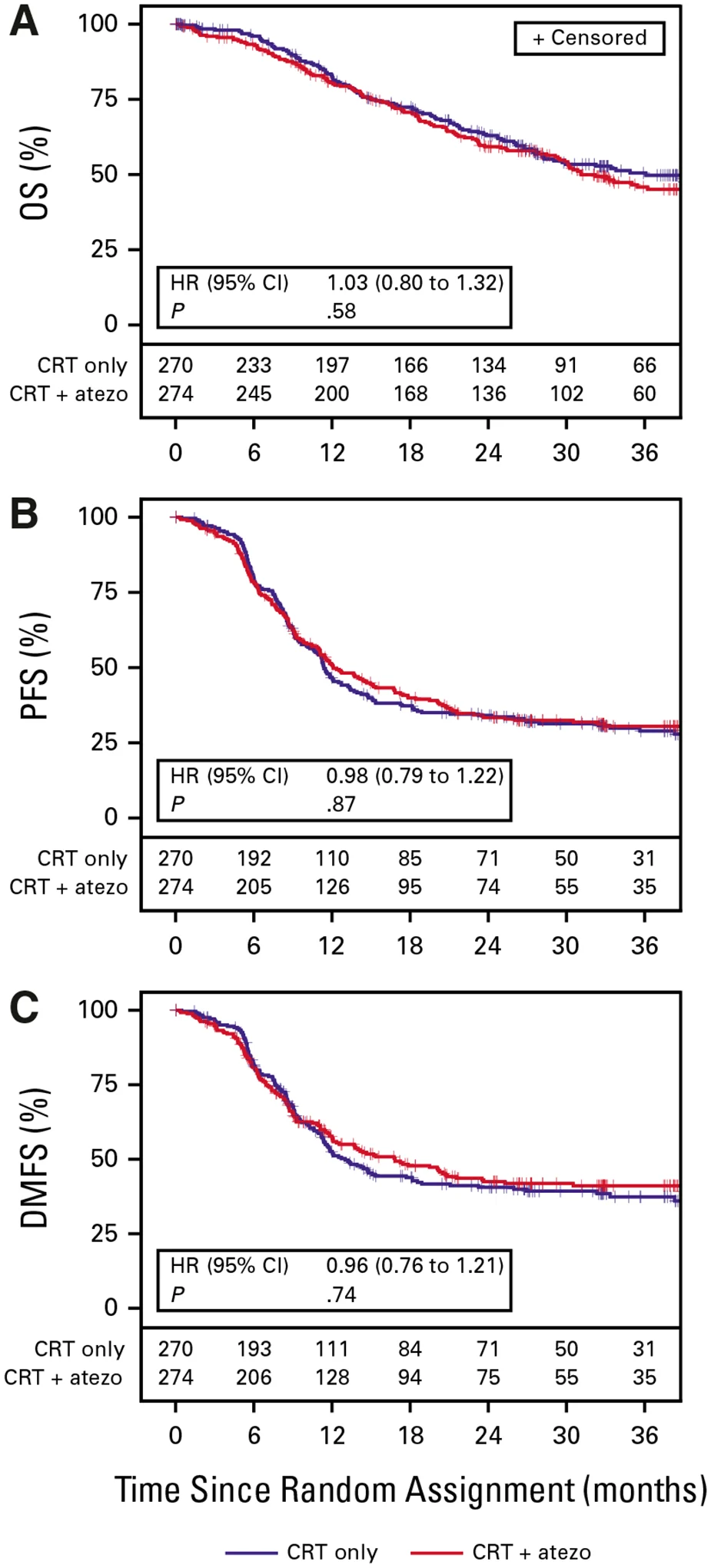
The trial did not meet its primary endpoint.
Adding atezolizumab to chemoradiotherapy did not significantly improve survival.
- Median overall survival was 36.1 months with chemoradiation alone
- Median overall survival was 31.1 months with chemoradiation plus atezolizumab
Similarly, progression-free survival was nearly identical:
- 11.4 months with chemoradiation alone
- 12.1 months with the addition of atezolizumab
Importantly, the study did not identify new safety concerns, suggesting that combining immunotherapy with chemoradiation is feasible, even if it did not improve outcomes.
An unexpected but important finding
Although immunotherapy did not improve survival, the trial revealed a striking observation related to radiation therapy scheduling.
Patients who received twice-daily radiation therapy had better survival outcomes compared with those treated with once-daily radiation.
This finding is particularly notable because twice-daily radiation has been supported by clinical evidence since the 1990s, yet it remains underused in routine clinical practice.
Currently, only about 20% of patients in the United States receive twice-daily thoracic radiation, largely due to logistical challenges for patients and treatment centers.Despite not being randomly assigned in this trial, the LU005 analysis again suggests that twice-daily radiation may meaningfully improve outcomes in limited-stage SCLC.
Why this trial matters
The results highlight an important reality in cancer research: not every promising strategy improves outcomes when tested in large clinical trials. While immunotherapy has revolutionized treatment for many cancers, LU005 suggests that simply adding checkpoint inhibitors to standard chemoradiation is not enough to improve survival in limited-stage SCLC.
At the same time, the trial reinforces the importance of optimizing existing treatments, particularly radiation therapy schedules that may already improve survival but remain underutilized.
Key Takeaways
- Adding atezolizumab to chemoradiotherapy did not improve survival in patients with limited-stage small cell lung cancer (SCLC) in the phase III NRG-LU005 trial.
- Twice-daily thoracic radiation was associated with improved survival outcomes, reinforcing evidence from earlier studies that this radiation schedule may offer meaningful clinical benefit.
- Despite decades of supporting data, twice-daily radiation remains underused in routine practice, highlighting an opportunity to optimize existing treatment strategies.
- Chemoradiotherapy continues to be the backbone of treatment for patients with limited-stage SCLC.
- Ongoing research aims to determine how immunotherapy can be more effectively integrated into earlier-stage SCLC treatment strategies.
You Can Read Full Article Here