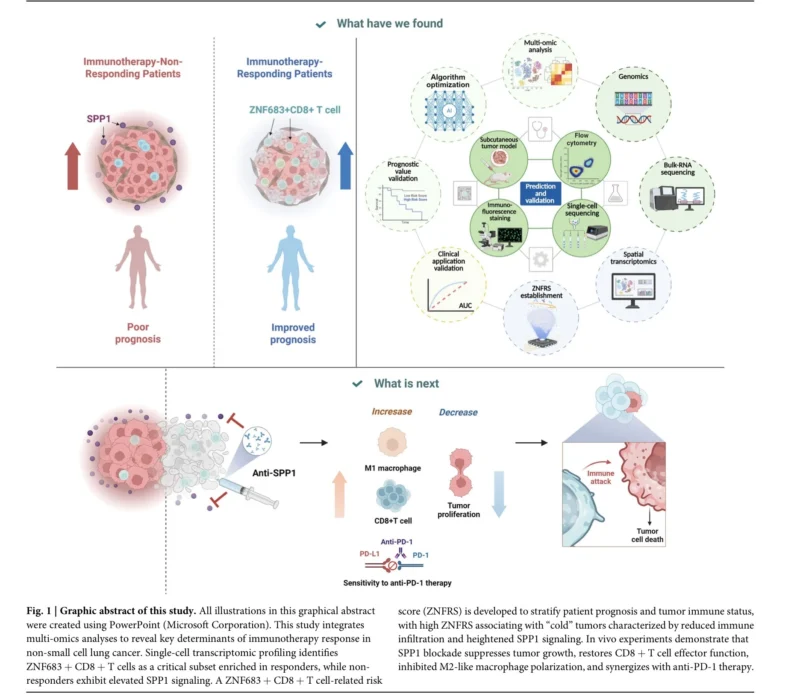
A new multi-omics study published in npj Precision Oncology provides a detailed view of how the tumor microenvironment may shape immunotherapy response in non-small cell lung cancer (NSCLC). By integrating bulk RNA sequencing, single-cell transcriptomics, spatial transcriptomics, machine-learning-based risk modeling, and mouse experiments, Zhang and colleagues identified ZNF683+ CD8+ T cells as a potentially important immune subset linked to response, while highlighting SPP1 signaling as a driver of immunosuppression and resistance (Zhang et al., 2026).
The study addresses one of the central problems in lung cancer immunotherapy: many patients either do not respond to immune checkpoint blockade or eventually develop resistance. While PD-L1, tumor mutational burden, and clinical factors remain useful, they do not fully explain why some tumors are immunologically active and others remain “cold.” This work suggests that the balance between ZNF683+ CD8+ T-cell infiltration and SPP1-driven myeloid suppression may help define that difference.
© The Author(s) 2026
Why This Study Matters
Immune checkpoint inhibitors have transformed the treatment of advanced NSCLC and are now also moving earlier in the disease course, including neoadjuvant chemoimmunotherapy. However, response remains heterogeneous. Some tumors show strong CD8+ T-cell infiltration and pathologic response, while others remain dominated by suppressive myeloid cells, stromal signals, and poor immune activation.
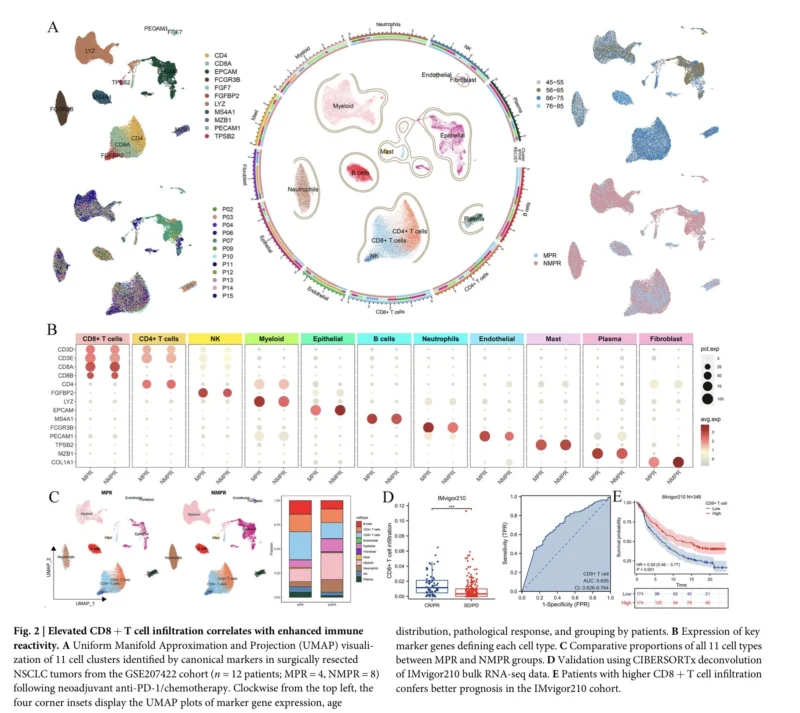
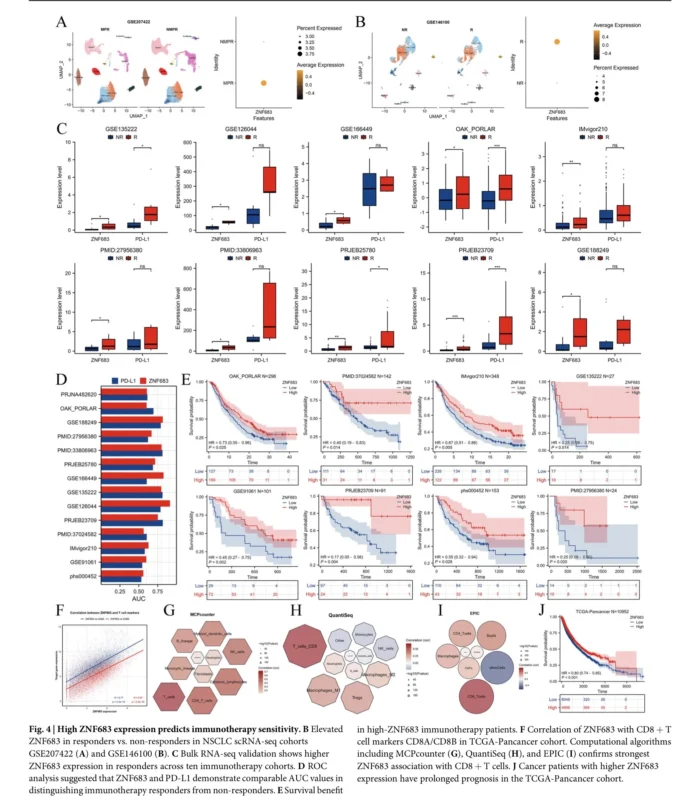
To investigate these mechanisms, the authors analyzed 31 datasets, including bulk RNA-seq, single-cell RNA-seq, and spatial transcriptomic datasets. They first focused on NSCLC samples from patients treated with neoadjuvant anti-PD-1 therapy plus chemotherapy, comparing tumors with major pathological response (MPR) against non-major pathological response (NMPR).
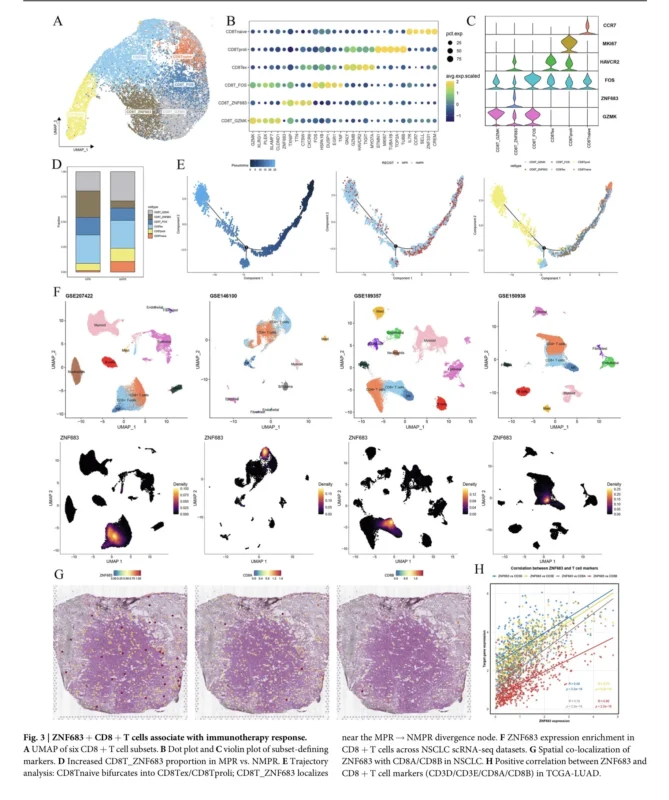
The analysis showed that MPR tumors had higher immune infiltration, particularly CD8+ T cells. Among CD8+ T-cell subsets, the ZNF683+ CD8+ T-cell population was specifically enriched in responders, suggesting that not all CD8+ T cells have the same relevance for immunotherapy benefit.
ZNF683+ CD8+ T Cells Emerged as a Response-Linked Subset
ZNF683, also known as Hobit, is a transcription factor associated with tissue-adapted and cytotoxic lymphocyte states. In this study, ZNF683 expression was predominantly enriched in CD8+ T cells across multiple NSCLC single-cell datasets and showed spatial co-localization with CD8A and CD8B.
Importantly, ZNF683 expression was higher in immunotherapy responders across several independent cohorts. In bulk RNA-seq datasets from NSCLC and other cancers, ZNF683 performed comparably to PD-L1 in distinguishing responders from non-responders. Patients with higher ZNF683 expression also showed stronger immune infiltration and better survival in several analyses.
This does not mean ZNF683 is ready to replace PD-L1 in clinical practice. But it suggests that ZNF683 may capture a more functional immune state, reflecting the presence of CD8+ T cells with stronger anti-tumor potential.
© The Author(s) 2026
ZNFRS Identified “Cold” Tumors With Poor Prognosis
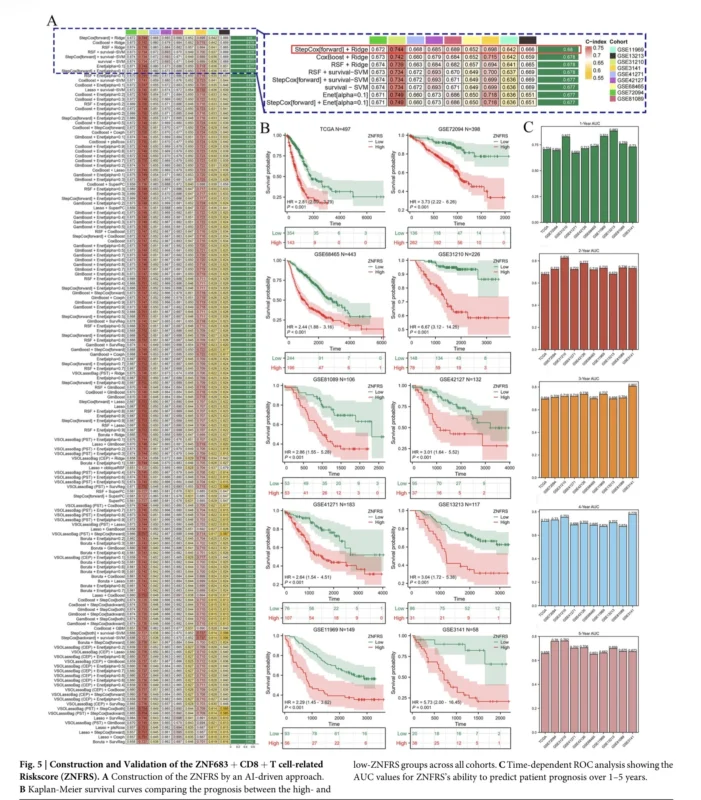
The authors then developed a ZNF683+ CD8+ T-cell-related Risk Score, called ZNFRS, using 19 marker genes from the ZNF683+ CD8+ T-cell population. They screened 296 algorithm combinations and selected the StepCox-forward plus Ridge model as the optimal method.
Across ten lung adenocarcinoma cohorts, high ZNFRS consistently predicted worse prognosis. The score also outperformed many clinical variables and compared favorably with previously published gene signatures.
Biologically, high-ZNFRS tumors showed features of an immunologically “cold” tumor microenvironment. These tumors had reduced immune cell infiltration, lower antigen presentation and immune signaling, and weaker chemokine activity. In contrast, low-ZNFRS tumors showed more active immune features and better prognosis.
This is an important distinction: ZNF683 expression itself was associated with immune activation, while high ZNFRS reflected a broader adverse transcriptional program linked to immune exclusion and tumor aggressiveness.
SPP1 Signaling Was Linked to Immunosuppression
One of the strongest mechanistic findings involved SPP1, also known as osteopontin. Cell-cell communication analyses showed that SPP1 signaling was enriched in non-responding tumors and high-ZNFRS tumors.
The SPP1 signal appeared to originate mainly from myeloid cells and malignant epithelial cells in the discovery cohort, and from SPP1+ macrophages in the independent validation cohort. These cells communicated with CD8+ T cells, CD4+ T cells, and NK cells through SPP1-CD44 and SPP1-ITGA4/ITGB1 axes.
This suggests that SPP1 may help maintain an immunosuppressive microenvironment by limiting cytotoxic lymphocyte infiltration and function. In practical terms, SPP1 signaling may be one mechanism by which tumors remain “cold” and resistant to anti-PD-1 therapy.
© The Author(s) 2026
SPP1 Blockade Improved Anti-PD-1 Activity in Mice
The preclinical validation strengthens the translational relevance of the study. In a Lewis lung carcinoma mouse model, anti-SPP1 therapy suppressed tumor growth. Anti-PD-1 also suppressed tumor growth, but the combination of anti-SPP1 plus anti-PD-1 showed the strongest anti-tumor effect.
SPP1 blockade increased tumor-infiltrating CD8+ T cells and enhanced production of key effector molecules, including IFN-γ and granzyme B. It also reduced M2-like macrophage polarization and shifted the tumor microenvironment toward a more immune-active state.
These findings support the idea that targeting SPP1 could help convert immunologically “cold” tumors into “hotter” tumors that are more responsive to checkpoint blockade.
© The Author(s) 2026
Clinical Interpretation
This study provides a useful framework for thinking about immunotherapy resistance in NSCLC. Responding tumors were enriched for ZNF683+ CD8+ T cells, while resistant tumors showed stronger SPP1-driven myeloid suppression. The combination of immune-state profiling and risk modeling may help identify patients with poor prognosis and low likelihood of immunotherapy benefit.
However, the findings remain largely translational and hypothesis-generating. Most analyses were based on public datasets, and the SPP1-targeting strategy was tested in preclinical models rather than in patients. The specific ligand-receptor mechanisms, including SPP1-CD44 and SPP1-integrin signaling, also require further functional validation.
Still, the study adds an important message to the NSCLC immunotherapy field: resistance may not be explained by one marker alone. It may require understanding the immune architecture of the tumor, including the quality of CD8+ T cells, the suppressive role of macrophages, and the signaling networks that prevent immune infiltration.
© The Author(s) 2026
Clinical Takeaway
The study identifies ZNF683+ CD8+ T cells as a promising marker of immunotherapy-responsive immune biology in NSCLC and introduces ZNFRS as a prognostic tool for lung adenocarcinoma. High-ZNFRS tumors showed “cold” immune features, poor prognosis, and increased SPP1 signaling.
Preclinical data suggest that SPP1 blockade may remodel the tumor microenvironment, restore CD8+ T-cell activity, reduce M2-like macrophage polarization, and enhance anti-PD-1 efficacy.
For thoracic oncology, this work points toward a future where immunotherapy response prediction may depend not only on PD-L1 or TMB, but also on the functional immune state of the tumor microenvironment.

© The Author(s) 2026