After nephrectomy, patients with high-risk clear cell renal cell carcinoma (ccRCC) remain vulnerable to recurrence. Adjuvant pembrolizumab became a key standard based on KEYNOTE-564, where pembrolizumab improved DFS versus placebo (DFS HR 0.68, 95% CI 0.53–0.87). LITESPARK-022 asks the next logical question:
can we improve on adjuvant PD-1 alone by adding a biology-matched agent?
Why belzutifan makes mechanistic sense
Belzutifan is an oral HIF-2α inhibitor, targeting a central oncogenic dependency in ccRCC (VHL/HIF axis). Pairing HIF-2α inhibition with PD-1 blockade is conceptually attractive: one agent disrupts hypoxia-driven tumor programs, while the other sustains anti-tumor T-cell activity—potentially reducing micrometastatic persistence after surgery.
Study Design and Methods (LITESPARK-022)
- Phase 3, multicenter, randomized, double-blind
- Population: ccRCC following nephrectomy (adjuvant setting; “certain patients with earlier-stage RCC” / increased recurrence risk)
Arms:
- Belzutifan 120 mg PO daily (~1 year) + pembrolizumab 400 mg IV q6w (~1 year)
- Pembrolizumab + placebo
- Primary endpoint: Disease-free survival (DFS)
- Key secondary endpoints: Overall survival (OS), safety, quality-of-life outcomes
Results
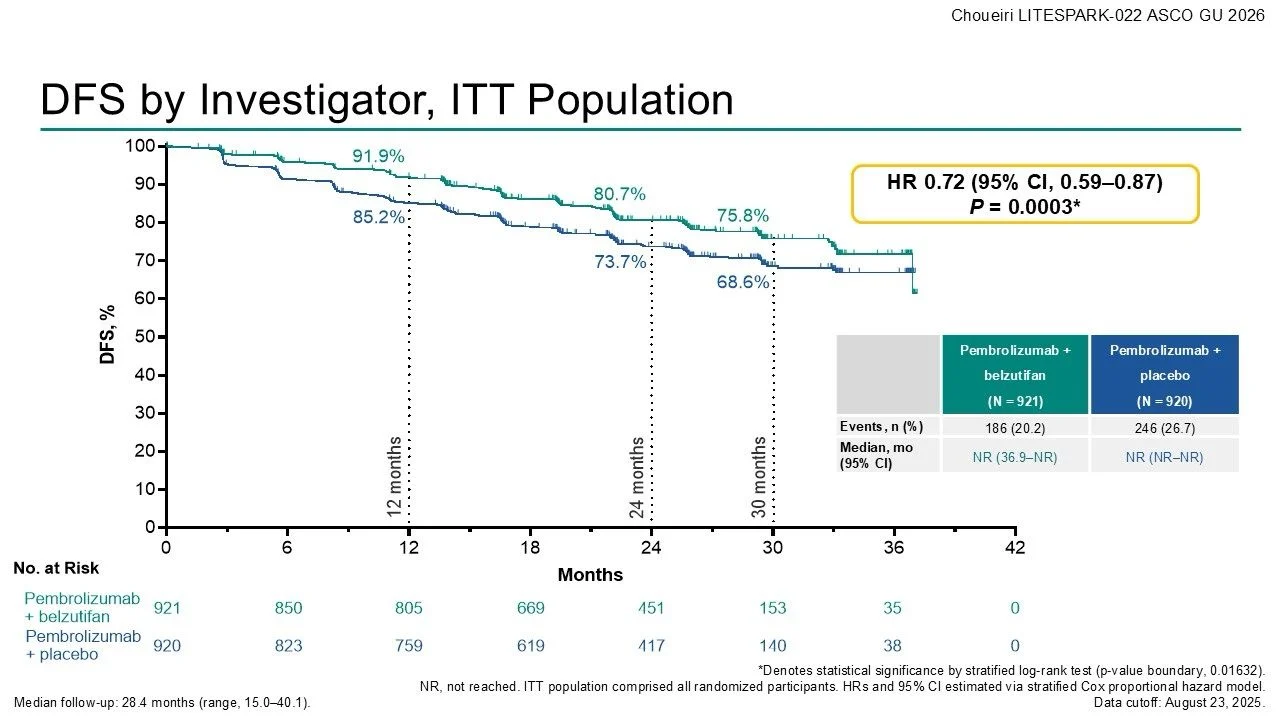
At the first pre-specified interim analysis, the LITESPARK-022 trial demonstrated a clear improvement in disease-free survival with the addition of belzutifan to pembrolizumab in the adjuvant setting.
With a median follow-up of 28.4 months (range 15.0–40.1), the combination therapy significantly reduced the risk of recurrence or death compared with pembrolizumab alone (HR 0.72; 95% CI 0.59–0.87; P = 0.0003), corresponding to a 28% relative risk reduction.
At the 24-month landmark analysis, disease-free survival rates favored the combination strategy:
- 80.7% with pembrolizumab + belzutifan
- 73.7% with pembrolizumab + placebo
Importantly, median DFS had not yet been reached in either treatment arm, reflecting ongoing disease control at the time of analysis. The DFS benefit appeared consistent across predefined clinical subgroups, supporting the robustness of the treatment effect.
Overall Survival: Overall survival data remain immature, and continued follow-up is ongoing, with OS retained as a key secondary endpoint of the study.
Safety
Safety was described as consistent with known profiles; however, toxicity intensity increased with the combination:
Treatment completion
- Completed assigned therapy: 69.5% combo vs 71.1% control
- Grade ≥3 TEAEs
- 52.1% combo vs 30.2% control
Most common Grade ≥3 TEAEs
- Anemia: 12.1% vs 0.5%
- ALT increased: 6.4% vs 2.0%
- Hypoxia: 4.6% vs 0%
Grade 5 events
- Treatment-emergent: 1.1% vs 1.2%
- Treatment-related: 0.3% vs 0.3%
Clinical takeaway: toxicity aligns with HIF-2α on-target effects (anemia/hypoxia) plus immune-related risks from pembrolizumab, emphasizing the need for proactive monitoring and dose-management discipline.
Why This Is Practice-Relevant
This is positioned as:
- First positive Phase 3 trial for belzutifan in earlier-stage disease
- First positive Phase 3 readout for a HIF-2α inhibitor + immunotherapy combination
- Critically: first adjuvant RCC combination study showing DFS superiority vs an active IO comparator (pembrolizumab)
That last point matters: improving on pembrolizumab (not placebo) raises the evidentiary bar—and LITESPARK-022 appears to clear it for DFS.
Regulatory Status (U.S.)
Based on LITESPARK-022, the FDA has accepted for priority review supplemental applications for belzutifan + pembrolizumab (including pembrolizumab QLEX) as adjuvant therapy in adults with RCC with a clear cell component at increased risk of recurrence after nephrectomy.
PDUFA target action date: June 19, 2026

Bottom Line
LITESPARK-022 delivers a statistically robust and clinically meaningful DFS improvement with adjuvant belzutifan + pembrolizumab versus pembrolizumab alone, at the cost of higher grade ≥3 toxicity dominated by anemia and hypoxia—consistent with HIF-2α inhibition. With OS pending and priority review underway, this regimen is a plausible next standard for appropriately selected high-risk ccRCC patients—especially where recurrence risk justifies escalation beyond PD-1 alone.
Kidney Cancer: Symptoms ,Causes, Stages, Diagnosis and Treatment