This week in OncoDaily Immuno-Oncology, advances across melanoma, lung cancer, and solid tumor immunobiology highlight a clear shift toward personalization, microenvironment reprogramming, and treatment optimization.
Neoadjuvant immunotherapy in melanoma continues to evolve into a biomarker-driven strategy, with IFNγ and PD-L1 guiding escalation and de-escalation, while long-term PRADO data reinforce durable survival benefits. In parallel, innovative platforms such as nanobody-based stem cell therapies targeting PD-1 and CSF1-R demonstrate the potential to locally rewire the tumor microenvironment and overcome resistance.
In advanced NSCLC, emerging meta-analyses challenge current practice by showing survival advantages of chemo-immunotherapy over monotherapy in high PD-L1 disease, while real-world and prospective data support safe discontinuation of immunotherapy at 2 years in selected patients. Beyond checkpoint blockade, next-generation strategies—including CAR-T evolution toward ecosystem targeting (uPAR), NMD inhibition to expand neoantigen landscapes, and macrophage-T cell reprogramming—are redefining tumor immunogenicity.
Across indications, these insights underscore a transition from uniform protocols to precision-guided immunotherapy, balancing efficacy, toxicity, and patient-centered decision-making in the next era of oncology.
This Week’s Expert Highlights in Immuno-Oncology
Marco Donia (Professor at University of Copenhagen):
“Neoadjuvant immunotherapy in melanoma: moving into personalization
Key question: which regimen for which patient?
Nature Reviews Immunology, Francesca Fallarino and Prof Dr Christian Blank, September 22, 2025
Immunity, Minke Lucas et al., January 13, 2026
Nature Medicine, Lotte Hoeijmakers et al., January 28, 2026
Key Points
1. Neoadjuvant immunotherapy is now established
Beyond proof-of-concept and into routine clinical application in many regions
2. Long-term outcomes support durable benefit
In PRADO, 5-year EFS was 71% and 5-year OS 86%
Patients achieving major pathologic response (MPR, <10% live tumor) had particularly favorable outcomes, with 5-year RFS 86% and DMFS 91%
3. Biomarkers may help guide de-escalation and escalation
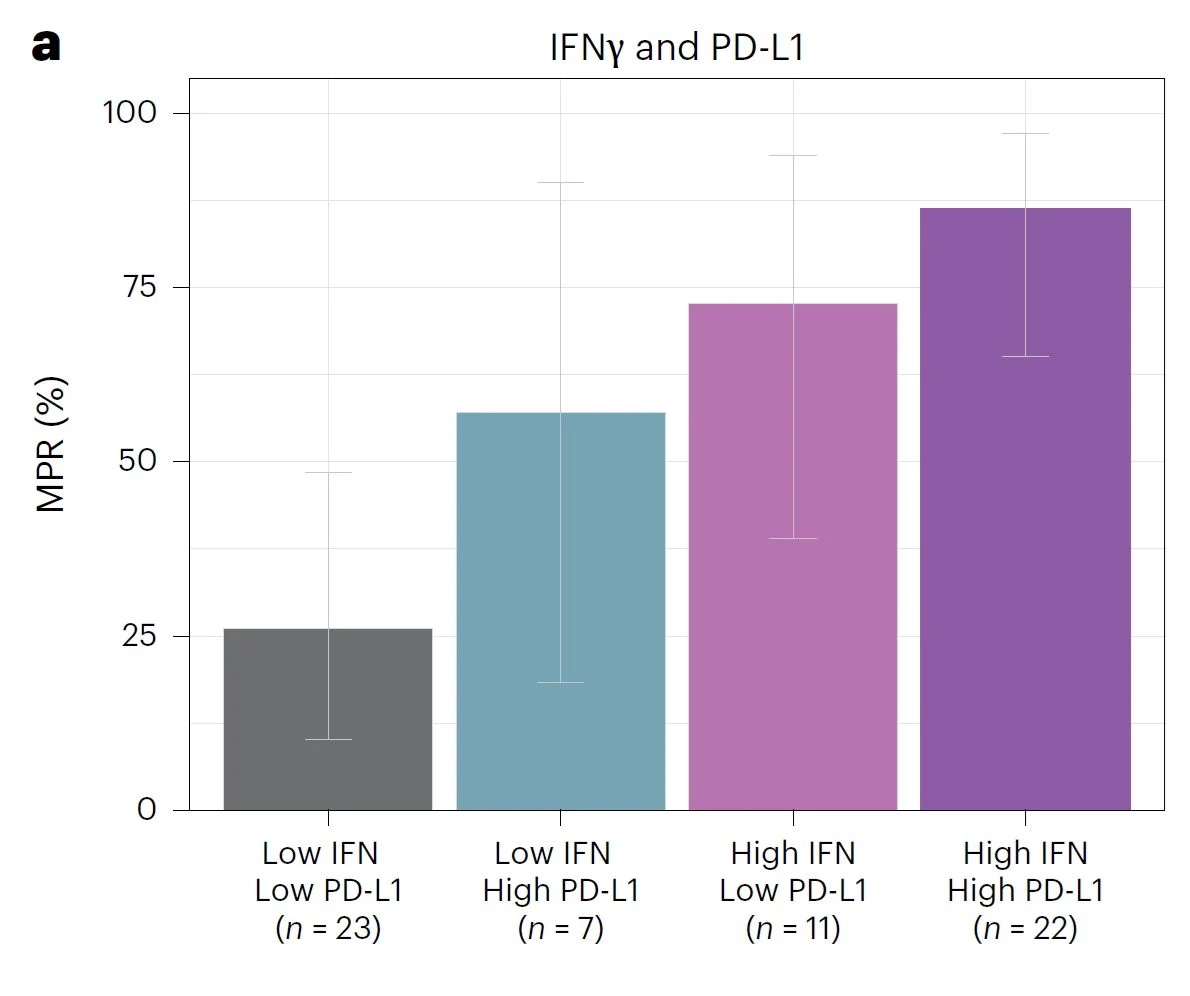
Lucas et al. suggest that baseline IFNγ signatures may help uncouple efficacy from toxicity
In IFNγ-high tumors, MPR was high across regimens (71% with anti-PD-1, 73% with ipi1+nivo3, 78% with ipi3+nivo1), while grade ≥3 irAEs increased (0%, 19%, 44%)
In IFNγ-low tumors, MPR increased with escalation (29%, 39%, 64%), while grade ≥3 irAEs remained relatively low (14%, 21%, 18%)
4. PRADO provides a practical biomarker example
Baseline IFNγ and PD-L1 further separated outcomes
Low IFNγ / low PD-L1: MPR 27%, 5-year EFS 48%
High IFNγ / high PD-L1: MPR 86%, 5-year EFS 91%
This supports biomarker-guided de-escalation for favorable biology, and escalation or novel combinations for less favorable disease
Take-Home
Neoadjuvant immunotherapy is now a standard strategy in resectable macroscopic, stage III hashtag#melanoma
Long-term data support durable benefit, especially in patients achieving MPR
The field is moving toward biomarker-driven treatment selection
Clinical Implications
A one-size-fits-all neoadjuvant immunotherapy approach is unlikely to be optimal
Next frontier: validation (who can be safely de-escalated and who needs escalation?)
Next frontier: PRADO also suggests that patients achieving MPR in the ILN may be candidates for surgical de-escalation, a strategy now being prospectively tested in the OMIT and MSLT-3 trials
Many congratulations to all investigators whose extraordinary efforts, especially with investigator-initiated neoadjuvant melanoma trials, are shaping the field, such as Alexander van Akkooi, Georgina Long, Prof Dr Christian Blank and many other esteemed colleagues!”
Khalid Shah (Vice Chair of Research at Brigham and Women’s Hospital, Director of Center for Stem Cell and Translational Immunotherapy at Brigham and Women’s Hospital, Professor at Harvard Medical School):
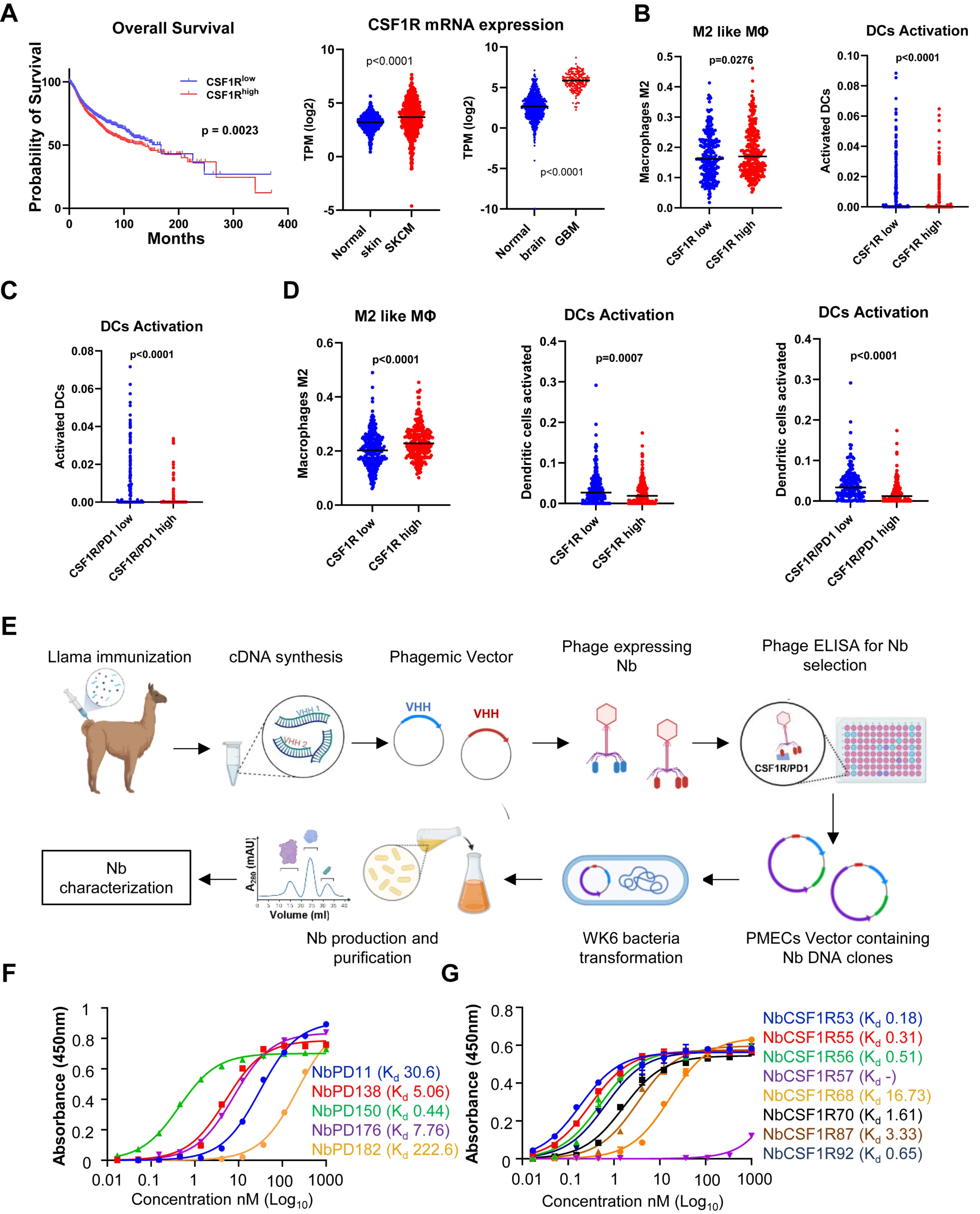
“Excited to share our recently published study in Lancet EbioMedicine
Solid tumors often evade immune-based therapies by establishing an immunosuppressive tumor microenvironment (TME), which actively shuts down the immune system. So we asked a different question: What if we could reprogram that environment locally, rather than fight it systemically?
In this study, we first generated nanobodies against key immune targets, PD-1 and CSF1-R, in llamas and developed a novel nanobody-stem cell platform that targets both innate and adaptive immune suppression via PD-1 and CSF1-R. Our findings show effective macrophage reprogramming, restoration of T cell activity, and enhanced antitumor immunity across multiple preclinical tumor models. This approach sidesteps some of the biggest limitations of systemic immunotherapy and opens a new path for treating resistant solid tumors.
Many congratulations to Ioulita Vogiatzi, collaborative teams of Jan Gettemans and Mohammad Rashidian, and the entire Center for Stem Cell and Translational Immunotherapy research team for their dedication and innovation in advancing cancer treatment.”
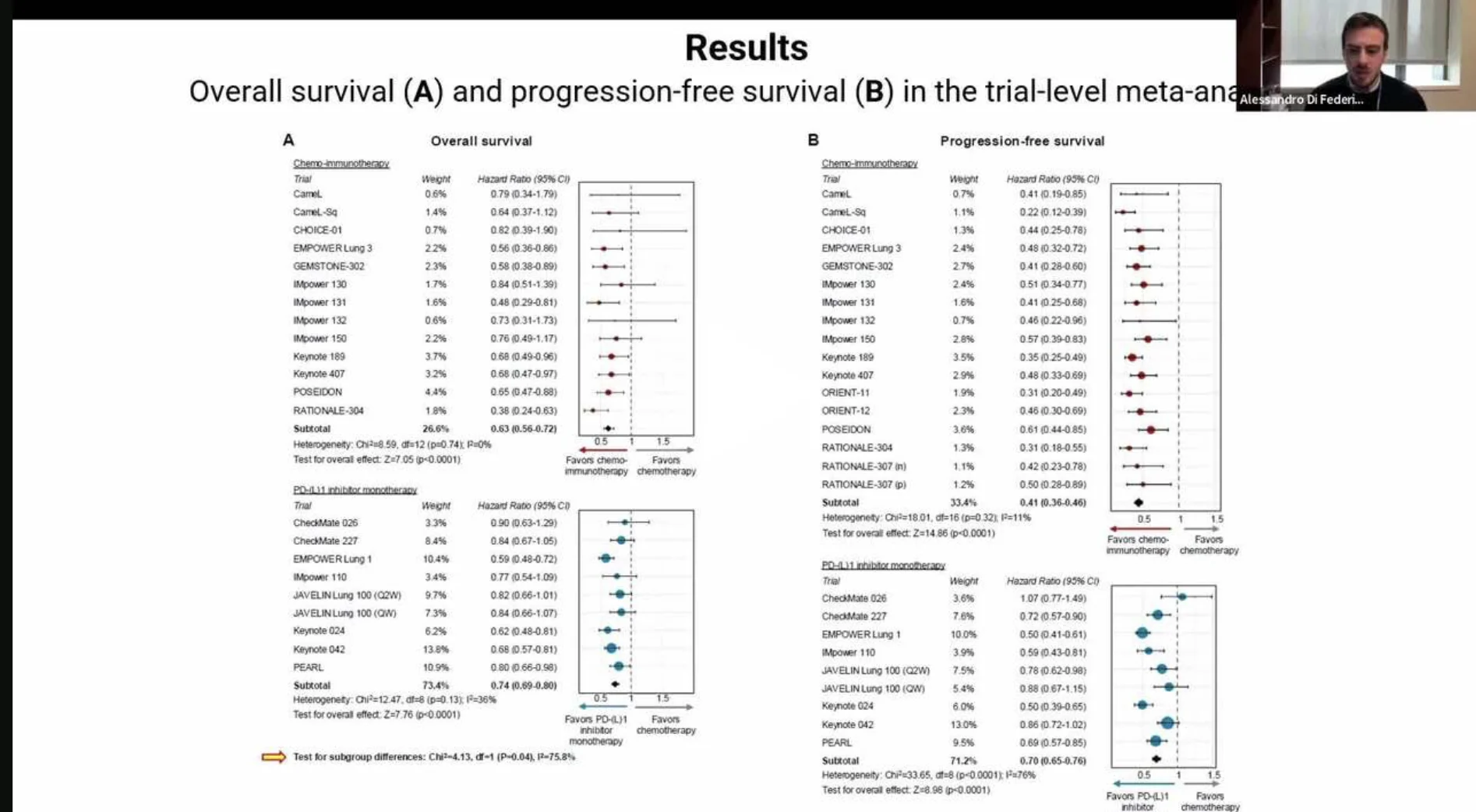
Alessandro Di Federico, (Medical Oncologist and Ph.D. student at the University of Bologna):
“Patients with advanced or metastatic non–small cell lung cancer (NSCLC) and high PD-L1 expression (≥50%) are commonly treated with PD-(L)1 inhibitor monotherapy, based on the assumption that it offers efficacy comparable to chemo-immunotherapy while avoiding chemotherapy-related toxicity. However, this assumption relies on indirect comparisons, as no head-to-head trials have directly evaluated these strategies in this population. In some countries, such as Italy, regulatory restrictions further limit the use of chemo-immunotherapy in patients with high PD-L1 expression.
In our study, presented at IASLC TTLC, we conducted a comprehensive meta-analysis to address this evidence gap. Using multiple complementary approaches—including trial-level meta-analyses of hazard ratios, meta-regression, network meta-analysis, and reconstructed individual patient data analyses—we consistently observed a significant benefit with chemo-immunotherapy over PD-(L)1 inhibitor monotherapy. This advantage was evident in both progression-free survival and, notably, overall survival.”
Adrien Rousseau (Medical Oncologist and PhD Student at Paris-Saclay University, France):
“Should we stop or continue immunotherapy after 2 years in advanced NSCLC?
Most landmark immunotherapy trials in advanced non–small cell lung cancer — such as KEYNOTE-024, KEYNOTE-189, and KEYNOTE-407 — used a fixed treatment duration of 2 years.
Yet in daily practice, many physicians continue treatment beyond 2 years, often driven by fear of relapse.So what does the evidence actually say?
Real-world data
A U.S. Flatiron database analysis (706 patients) led by Lova Sun showed that only 1 in 5 patients stopped at 2 years — with no difference in overall survival between stopping and continuing.
A French SNDS nationwide analysis (3,214 patients) found that only 1 in 3 patients stopped at 2 years — again with no survival difference.
A multicenter Italian study (173 patients) led by Maria Gemelli reported that only 1 in 6 patients discontinued at 2 years. No difference in overall survival was observed, although a signal for shorter PFS was suggested in specific subgroups (brain metastases, low PD-L1, KRAS mutations).
Prospective evidence: The recent DICIPLE trial led by Gérard Zalcman suggests that early discontinuation in patients with controlled disease may not be detrimental.
Our contribution
I am pleased to share our latest publication in JAMA Oncology, where we report outcomes of more than 1,400 patients who discontinued immunotherapy at 2 years.
Within 4 years after treatment discontinuation:
Overall survival reached 76.9%
Most patients did not require a subsequent systemic therapy
Very few patients had immunotherapy rechallenge
These findings contribute to the growing body of evidence supporting that stopping immunotherapy at 2 years may be a reasonable and safe strategy for selected patients.
Thank you to all teams and collaborators involved in this collective effort at Gustave Roussy and CESP – Centre de recherche en Epidémiologie et Santé des Populations.
Special gratitude to my mentors Benjamin Besse Stéphanie Foulon for their guidance and support throughout this work, and to all my co-authors.”

Esther A. Omilaju (Clinical Pharmacist in Training, Oncopharmacogenomics and Precision Oncology, AI-Enhanced Cancer Therapy Research):
“CAR T-Cell Therapy: A Promising Frontier in Cancer Treatment
CAR T-cell therapy represents a major advancement in cancer management, particularly in hematological malignancies. It is a form of immunotherapy where a patient’s own T lymphocytes are genetically engineered to recognize and eliminate cancer cells with high specificity.
This approach is closely linked to developments in Chimeric Antigen Receptor T-cell Therapy, where T cells are modified to express receptors that target tumor-associated antigens. Once reinfused into the patient, these cells actively seek out and destroy malignant cells.
Unlike conventional treatments such as chemotherapy, which affect both healthy and cancerous cells, CAR T-cell therapy offers a more targeted mechanism, reducing off-target toxicity and improving therapeutic outcomes in certain cancers like leukemia and lymphoma.
Although challenges such as cytokine release syndrome and high cost remain, ongoing research continues to refine its safety, accessibility, and broader clinical application. CAR T-cell therapy stands as a clear example of how genetic engineering and immunology are converging to reshape the future of oncology.”
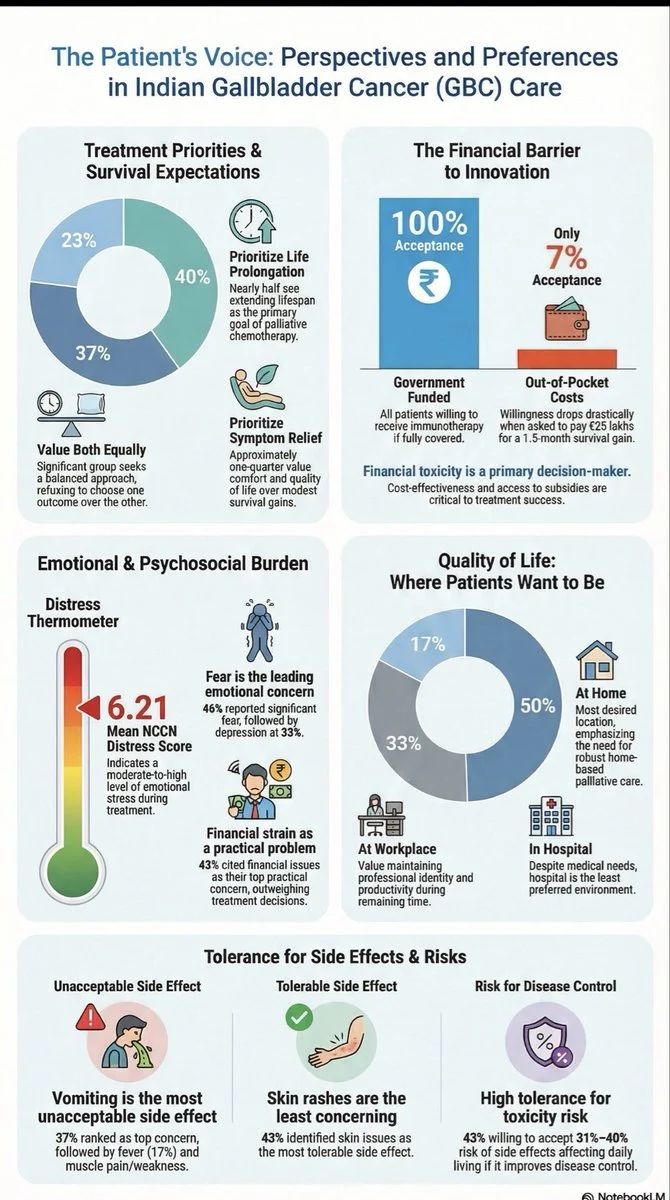
Amol Patel (Medical Oncologist, Advocate of “One Earth, One Family, One Future):
“In the saddest reality of gallbladder cancer, does patient voices matter? We tried to understand their views and preferences. 25% of patients do not want costly chemotherapy immunotherapy. More power to palliative care. Make gallbladder cancer history ! “
Guru Sonpavde (GU Oncology and Phase I Clinical Trials Director, Chair of Bladder Cancer Research at AdventHealth Central Florida, Professor of Medicine at the University of Central Florida, and at Loma Linda University Health):
“bladder cancer bladder-preservation clinical trial combining radiation + PD-L1 inhibitor immunotherapy for muscle-invasive bladder cancer (MIBC): In this phase II trial of cisplatin-ineligible patients with MIBC investigating radiation + concurrent + adjuvant avelumab, the majority (9/14, 64%) had a clinical complete remission (cCR). Although many patients experienced Grade 1-2 toxicities, only 3 of 14 patients (21%) experienced a Grade 3 event. Ongoing important trials in this context are:
1) Two randomized Phase III trials (SWOG/NRG 1806 and KEYNOTE-992 are investigating the addition of anti-PD1/PD-L1 therapy to concurrent chemoradiotherapy in MIBC
and
2) SWOG S2427, a Phase 2 trial of radiation plus pembrolizumab led by Dr Leslie Ballas SWOG Cancer Research Network in patients with MIBC who have experienced a good clinical response to neoadjuvant systemic therapy (chemo, chemo-IO or EV-pembro). Relevant data from other previously reported trials include:
The DUART trial- radiation + concurrent and adjuvant durvalumab (cCR was 62%,
IMMUNOPRESERVE trial tested concurrent durvalumab and tremelimumab (anti-CTLA4) with radiation (cCR of 81%), 3) NUTRA trial of radiation plus concurrent and adjuvant nivolumab (cCR not reported, median PFS 11.4 months).”
Dr. Howard Smith, MD (Harvard Medical School; AM in Immunology, Harvard; Medical Broadcaster with 40+ Years in Radio and Podcasts; Former Medical Editor at WBZ-AM, Boston):
“Empowering Ovarian Cancer Immunotherapy. Aggressive ovarian cancers are deadly because they block a patient’s immune system. A new study demonstrates how this shield can be removed. “
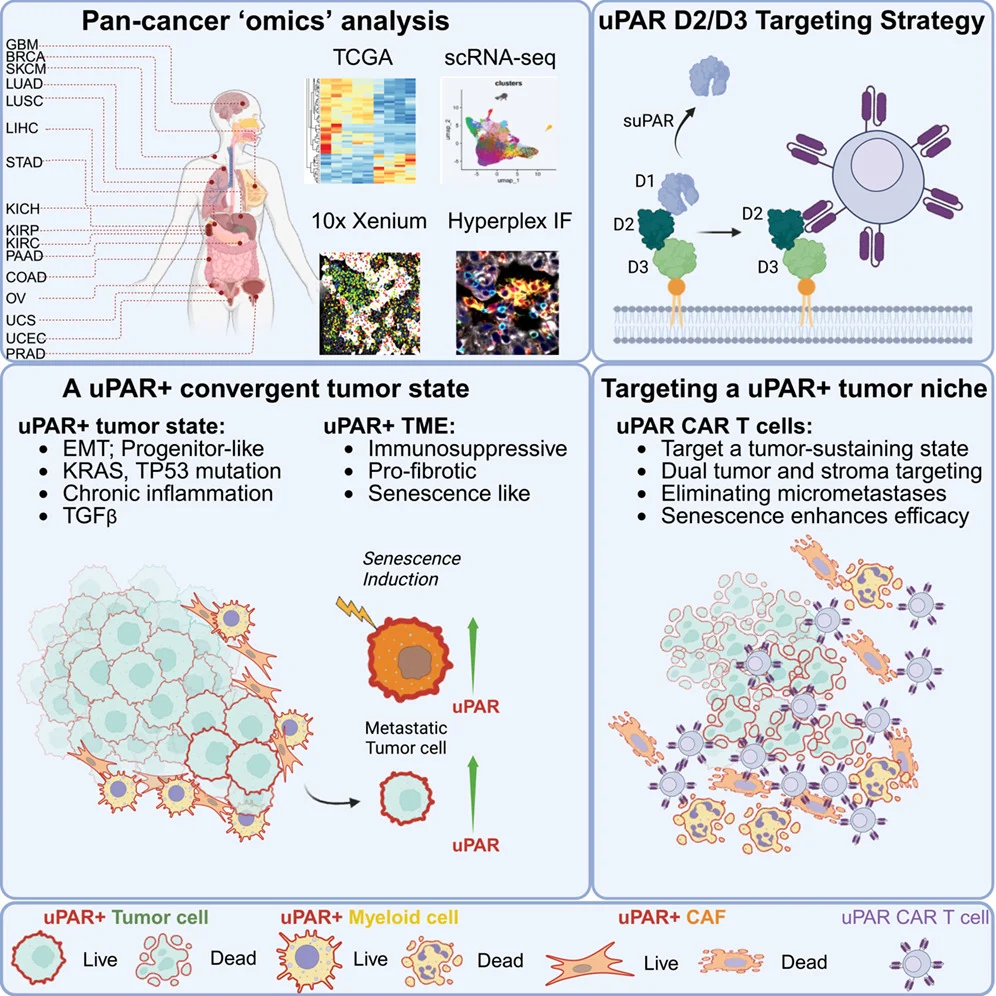
Jochen Reiser MD, PhD, (President of UTMB and CEO of the UTMB Health System at the University of Texas):
“Great new paper in Cell What if we’ve been targeting the wrong thing in solid tumors?
uPAR CAR T cells don’t just attack cancer cells but instead they dismantle the entire tumor ecosystem. Tumor + stroma + senescence. A potential paradigm shift.A convergent uPAR-positive tumor ecosystem creates broad vulnerability to CAR T cell therapy.”
Janne Ruotsalainen (Senior Research Scientist at CureVac):
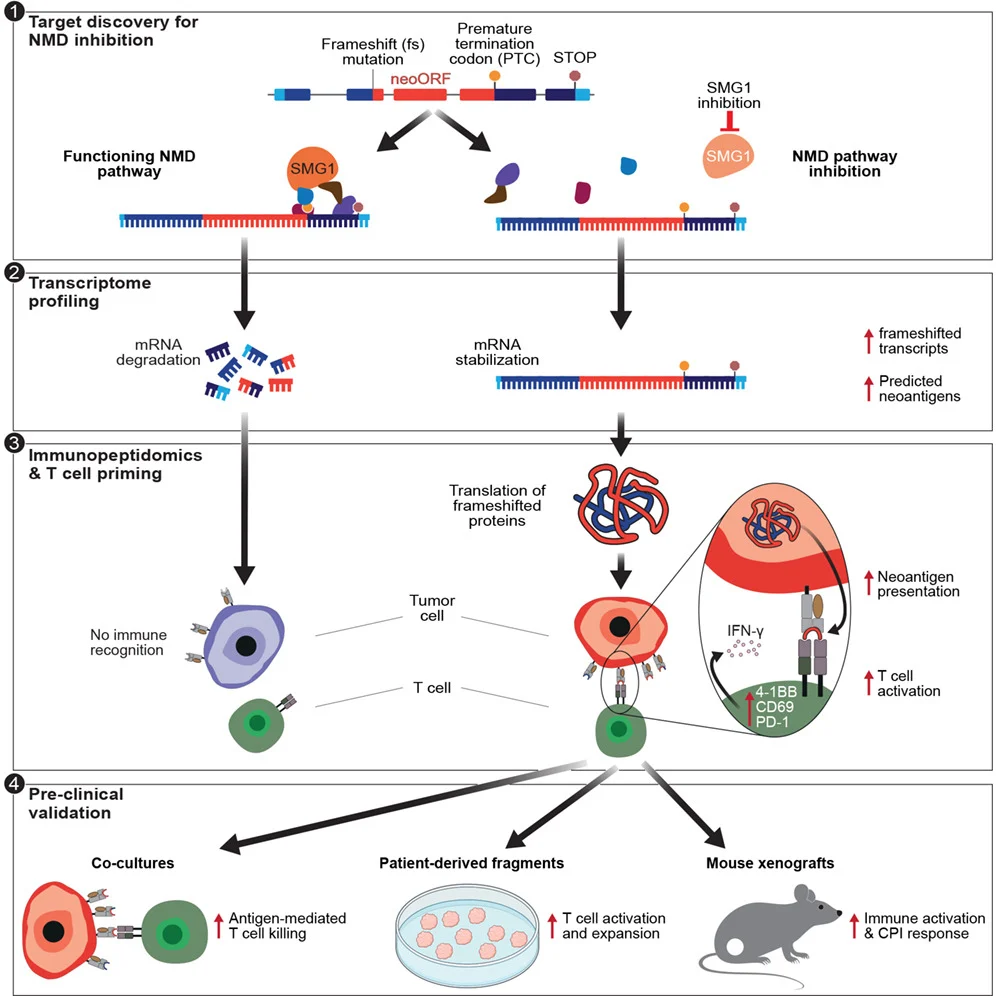
” 𝗡𝗲𝘄 𝗶𝗻 𝗜𝗺𝗺𝘂𝗻𝗶𝘁𝘆: 𝗧𝗮𝗿𝗴𝗲𝘁𝗶𝗻𝗴 𝗻𝗼𝗻𝘀𝗲𝗻𝘀𝗲‑𝗺𝗲𝗱𝗶𝗮𝘁𝗲𝗱 𝗺𝗥𝗡𝗔 𝗱𝗲𝗰𝗮𝘆 (𝗡𝗠𝗗) 𝘁𝗼 𝗲𝘅𝗽𝗮𝗻𝗱 𝘁𝘂𝗺𝗼𝗿 𝗻𝗲𝗼𝗮𝗻𝘁𝗶𝗴𝗲𝗻 𝘃𝗶𝘀𝗶𝗯𝗶𝗹𝗶𝘁𝘆
A recent study in Immunity (Vendramin et al., Nonsense‑mediated mRNA decay inhibition reshapes the cancer immunopeptidome, 2026) reports that inhibition of nonsense‑mediated mRNA decay (NMD) can reshape the tumor immunopeptidome and improve responsiveness to immune checkpoint blockade across cancer types.
𝗞𝗲𝘆 𝗳𝗶𝗻𝗱𝗶𝗻𝗴𝘀 𝗳𝗿𝗼𝗺 𝘁𝗵𝗲 𝗮𝘂𝘁𝗵𝗼𝗿𝘀:
The study identifies reduced activity of the NMD pathway kinase SMG1 as a correlate of improved clinical response to immune checkpoint blockade (ICB) in large patient cohorts (>1,000 cases).
Pharmacological or genetic NMD inhibition stabilizes transcripts containing premature termination codons, leading to presentation of previously inaccessible canonical and non‑canonical peptides on major histocompatibility complex class I (MHC class I) molecules.
As a result, neoantigen abundance increases to levels comparable to tumors with high mutational burden, despite many of these antigens being non‑mutational in origin.
𝗢𝗻 𝘁𝗿𝗮𝗻𝘀𝗹𝗮𝘁𝗶𝗼𝗻𝗮𝗹 𝗳𝗲𝗮𝘀𝗶𝗯𝗶𝗹𝗶𝘁𝘆 𝗮𝗻𝗱 𝗳𝘂𝘁𝘂𝗿𝗲 𝗮𝗽𝗽𝗹𝗶𝗰𝗮𝗯𝗶𝗹𝗶𝘁𝘆:
While clinical applicability and safety will ultimately need to be addressed in early‑phase patient trials, the study provides a strong preclinical rationale for pharmacological modulation of NMD. Beyond synergy with immune checkpoint blockade, the selective upregulation of otherwise hidden tumor peptides also raises the possibility that NMD inhibition could support additional immunotherapeutic strategies, including active mRNA‑ or peptide‑based immunization targeting these newly exposed antigens.𝗢𝗻 𝗺𝗶𝘀𝗺𝗮𝘁𝗰𝗵 𝗿𝗲𝗽𝗮𝗶𝗿 𝘀𝘁𝗮𝘁𝘂𝘀 𝗮𝗻𝗱 𝗺𝘂𝘁𝗮𝘁𝗶𝗼𝗻 𝗯𝘂𝗿𝗱𝗲𝗻:
Importantly, the data indicate that checkpoint blockade efficacy was enhanced independent of classical tumor mutation burden, suggesting potential relevance beyond microsatellite instability (MSI)‑driven, mismatch repair (MMR)‑deficient cancers. This is notable because microsatellite‑stable (MSS), MMR‑proficient tumors typically harbor fewer mutations and are therefore less responsive to immune checkpoint blockade. The work proposes that RNA‑level dysregulation can partially compensate for low DNA‑level neoantigen load, thereby broadening the spectrum of tumors that may become immunologically visible.Overall, this study highlights RNA surveillance pathways as tractable modulators of tumor immunogenicity, offering a mechanistically distinct route to increase immune recognition across cancers, while clearly motivating further translational evaluation.”
Robert J. Wassler (Healthcare IT Project Manager specializing in PMO, risk management, Agile methodologies, and ServiceNow implementation):
“Cancer treatment is being rewritten—right now.
A new wave of immunotherapy isn’t just slowing cancer down… it’s eliminating it in some patients.
Instead of attacking tumors directly like chemotherapy, these therapies train the immune system to recognize and destroy cancer cells—and the results are stunning.
But here’s the reality check:
- Not all cancers respond yet
- Access to these treatments is still uneven
- We’re just scratching the surface of what’s possible
The takeaway?
We’re entering a new era where cancer may increasingly become manageable—or even curable—for many.”