This week in OncoDaily Immuno-Oncology, emerging data challenge and refine how we deliver immunotherapy – from real-world evidence on safely stopping PD-1 therapy in advanced NSCLC and perioperative EV + pembrolizumab reshaping bladder cancer, to immune microenvironment biomarkers redefining risk in young TNBC. Highlights include advances in CAR-T engineering for solid tumors, fasting-enhanced immunotherapy responses, personalized neoantigen vaccines, and critical reflections on negative melanoma trials. Together, these insights underscore a maturing IO field focused not only on innovation, but on precision, patient selection, and smarter treatment strategies.
Jose Luis Portugal Del Pino (Clinical Practice Instructor (Physician) at Universidad Científica del Sur):
“Can we safely stop immunotherapy after 2 years in advanced NSCLC?
The final results of the I-STOP study (European Journal of Cancer) provide important real-world evidence on one of the most debated questions in thoracic oncology.
Study overview
173 patients with advanced NSCLC who had disease control after ≥24 months of single-agent PD-1/PD-L1 therapy were analyzed. Patients either discontinued or continued treatment.
Key findings
- No overall survival difference between stopping and continuing
(HR 1.22; 95% CI 0.50–2.95; p = 0.66) - Higher progression risk after discontinuation
(HR 2.16; 95% CI 1.04–4.48; p = 0.035) - Subgroups at higher risk if stopping: – PD-L1 ≤ 50%
– Brain metastases
– ECOG PS ≥ 1
– KRAS mutations - ICI rechallenge was effective
62.5% response rate in relapsing patients - Late grade 3–4 immune-related AEs were uncommon (3.5%)
Clinical takeaway
Stopping anti-PD-1/PD-L1 therapy at 24 months appears safe for many patients with durable disease control – without compromising overall survival.
However, selected high-risk subgroups may benefit from prolonged treatment.
The real challenge now is clear: Identifying who can safely stop – and who should continue.”
Adnan Khattak (Medical Oncologist at Hollywood Private Hospital):
“KeyVibe-010, Phase III trial, evaluating vibostolimab co-formulated with pembrolizumab as adjuvant therapy in resected stage IIB–IV melanoma has unfortunately not demonstrated additional clinical benefit compared to pembrolizumab alone.
Another difficult outcome, following earlier premature discontinuations or negative studies such as PIVOT-12 and RELATIVITY-098. While these results are disappointing, they reinforce an essential truth in oncology: clinical trials are the only way we truly discover what works — and what does not.
Negative trials are not failures. They are important data. They refine our understanding, redirect scientific effort, protect future patients from ineffective strategies, and shape the next generation of research questions. Timely and transparent reporting of such results is critical for scientific progress.
We are deeply grateful to the patients who participated in this study – your contribution advances knowledge for the global oncology community.
At One Clinical Research, we are proud to have been the lead recruiting site globally for this trial. The commitment of our patients, investigators Azim Khan,Omar Faruque and research team remains unwavering. We value our collaboration with MSD MSD Australia & New Zealand Moderna.
Now, we turn our focus with cautious optimism toward INTERPATH-001, the personalised cancer vaccine trial. After consecutive setbacks in the adjuvant melanoma space, we hope this next chapter brings meaningful progress for our patients.
The work continues. Always.”
Diego Gomez Abreo (Medical Director, Clinical Research Unit at Fundación Cardiovascular de Colombia):
“Can the Immune Microenvironment Redefine Risk in Young Triple-Negative Breast Cancer?
In oncology, we often move from the macro to the micro — from stage and tumor size to biology and immune contexture. This study challenges one of our most ingrained assumptions: that all young patients with early triple-negative breast cancer (TNBC) require chemotherapy.
In a population-based cohort of young (<40), node-negative TNBC patients who received no systemic therapy, stromal tumor-infiltrating lymphocytes (sTILs) emerged as a powerful independent prognostic biomarker .
The numbers are striking:
- Patients with high sTILs (≥75%) had a 10-year overall survival of ~95%
- Their 10-year risk of distant metastasis or death was only ~2%
- Each 10% increase in sTILs reduced mortality risk by ~19%
This is not predictive in the context of chemotherapy.
This is pure biology – immune surveillance translating into durable survival.
Why does this matter for breast cancer today?
Young women with TNBC routinely receive anthracycline- and taxane-based chemotherapy. While life-saving, this comes at the cost of infertility, cognitive impairment, cardiotoxicity, and long-term quality-of-life impact.
If a subgroup exists with an ultralow biological risk driven by a robust endogenous immune response, should we still treat them all the same?
sTILs offer:
- Standardized evaluation
- Low cost (H&E-based)
- High reproducibility
- Immediate global applicability
This could open the door to:
- Prospective chemotherapy de-escalation trials
- Integration with immune gene signatures
- Refinement of staging systems beyond TNM
Broader implications beyond breast cancer
The principle extends far beyond TNBC.
Across oncology, immune infiltration is emerging as a central determinant of outcome:
- Melanoma
- Non-small cell lung cancer
- Colorectal cancer (Immunoscore)
- Ovarian cancer
- HPV-related tumors
The concept is consistent: an immune-inflamed tumor microenvironment can convert an aggressive phenotype into a biologically favorable disease.
In the era of immunotherapy, this also raises critical questions:
- Could high baseline immune infiltration identify patients who need less cytotoxic therapy?
- Should immune contexture be incorporated into formal staging?
- Can we design biology-driven de-escalation strategies across tumor types?
This study reinforces a paradigm shift:
Cancer prognosis is no longer defined solely by tumor burden – but by the dynamic interaction between tumor and host immunity.
The future of oncology will not only be precision targeting of tumor mutations.
It will also be precision interpretation of immune architecture.”
Djordje Atanackovic (Professor at University of Maryland Greenebaum Comprehensive Cancer Center):
“Postdoc Position: CAR T Cell Therapy for Solid Tumors | Baltimore
The Atanackovic Lab at the University of Maryland Greenebaum Comprehensive Cancer Center (UMGCCC) is recruiting a junior Postdoctoral Fellow to join our translational CAR T cell immunotherapy program.
Our research has a growing emphasis on solid tumors, while continuing to maintain strong expertise in hematologic malignancies and clinically integrated CAR T cell platforms.
This position is ideal for recent PhD, MD or MD/PhD graduates with hands-on experience in:
- Engineering and characterization of next-generation CAR T cells (tri-specific and armored designs)
- Work with primary human T cells and patient-derived clinical trial samples
- Advanced flow cytometry and immune profiling
- Research conducted in a clinically integrated, regulatory-compliant environment
Candidates should demonstrate independent experimental productivity and strong scientific writing skills.
The successful candidate will work at the interface of preclinical modeling, GMP-adjacent workflows, and early-phase clinical immunotherapy studies within an NCI-designated Comprehensive Cancer Center, with strong mentorship and support for first-author publications.
Location: Baltimore
Apply here.
Rolling review – please share with interested trainees!”
Benjamin Louis (PhD Candidate at Universitätsspital Zürich):
“Very happy that the rest of the work I had the opportunity to be part of during my time at the Dana-Farber Cancer Institute has been published in the Journal For ImmunoTherapy of Cancer.
With this work, we provide robust pre-clinical data, with an improved version of the CAR-E able to drive robust expansion and memory formation in patient and humanized mice settings.
Congratulations to everyone that drove this project so far, exited to see the CAR-E move towards a Phase I clinical trial and eventually meaningfully impact patients lives!”
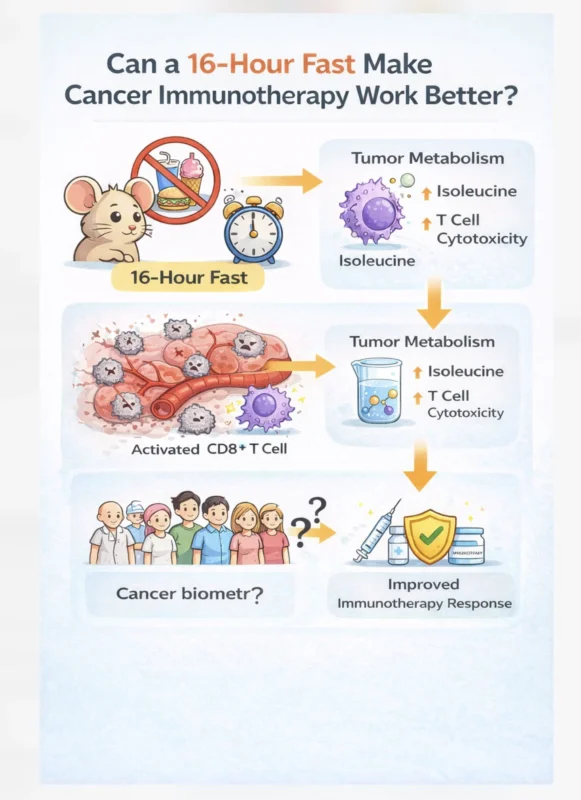
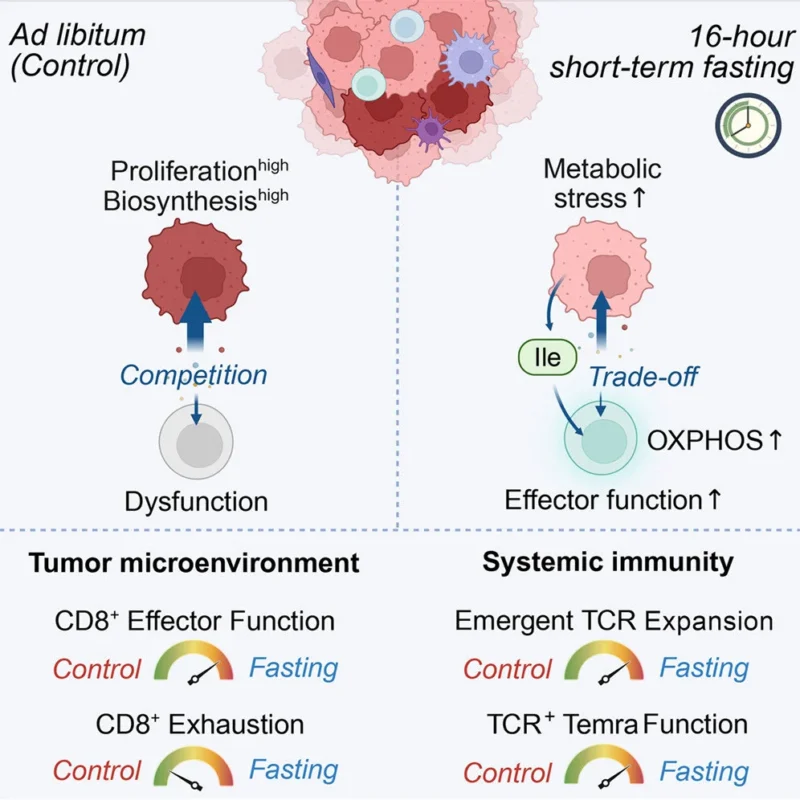
Luigi Fontana (Director, Charles Perkins Centre RPA ‘Health for Life’ Program at University of Sydney):
“Can a 16-hour fast make cancer immunotherapy work better? A new Cell Metabolism study (Chen et al., 2026) explores a simple idea: Short-term fasting may reshape the tumor microenvironment and enhance anti-tumor immunity.”
Yannick Buccella (Gastrointestinal and molecular oncologist):
“There is even data that the vaccine sensitizes tumor cells for the immunotherapy that we give to >50% of cancer patients nowadays. Do I sit here and propagate giving everybody „the jab“ to make the immunotherapy more effective? No, because it’s a hypothesis only. And medicine doesn’t work like that.”
Nurullah İlhan (Founder & Head of the Medical Oncology Clinic):
“Personalized cancer vaccines move forward. In early TNBC, an individualized neoantigen mRNA vaccine induced long-lasting T-cell immunity (up to 6 years). 11/14 relapse-free (~5y follow-up). Important proof-of-concept in adjuvant immunotherapy.”
Anirban Maitra (Director, Perlmutter Cancer Center at NYU Langone Health):
“How much of the time of day effects of immunotherapy are because of the favorable impacts of overnight fasting on T cell function (getting your infusion pre-breakfast)? Something to consider! 16-h fasting optimizes cancer immunotherapy in mice and humans”
Sumanta K. Pal (Vice Chair of Academic Affairs at City of Hope):
“Brilliant overview of studies to look out for as we all head into GU26.”
Written by Rima Grigoryan, MDc, Assistant Editor of OncoDaily IO.


