At OncoDaily Immuno-Oncology, we highlight the most impactful developments in cancer immunotherapy each week. This roundup covers groundbreaking research in regulatory T cells, novel CAR-T therapies, and the latest clinical trials. It provides key insights into advancements in lung cancer treatment, immunotherapy combinations, and global collaborations pushing the field forward.
John Gordon (co-Founder, Director, VP Scientific Affairs at Celentyx Ltd):
“Immunotherapy | ‘Morphing Tregs to OutFOX Tumours’ | Reprogramming Regulatory Tcells in the Tumor MicroEnviromment by Morpholino-Mediated Splicing of FOXP3 | Breaking Study at Science Magazine Immunology |
The master regulator of regulatory T (Treg) cells, FOXP3, is present in humans as two major variants, including a shorter isoform lacking exon 2 (FOXP3dE2). Here, Yujing Li, Naresh Singh, Chuanpeng Dong, Samantha Sharma, et al found that mice expressing the FOXP3dE2 isoform were resistant to the growth of multiple tumor types.
Tumor-infiltrating Treg cells from Foxp3dE2 mice were less suppressive of CD8 T cell responses and produced higher levels of effector cytokines, including IFN-γ. A morpholino oligo that induced the skipping of FOXP3 exon 2 slowed growth of tumors in mice and organoids derived from patients with breast or colorectal cancer. These findings suggest that targeting FOXP3 isoform usage could be an approach to alleviate Treg cell–mediated tumor immunosuppression.
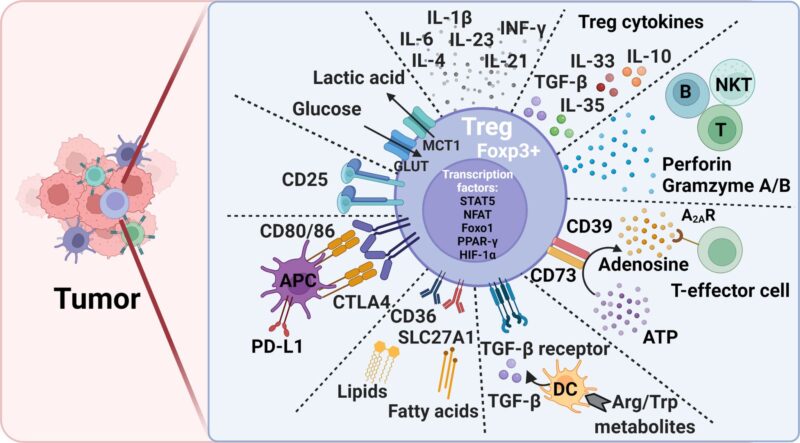
FIGURE | OVERVIEW of Treg-mediated immunosuppression mechanisms and potential therapeutic targets to alleviate immunosuppression in tumors | Taken from: Functional Diversity of Regulatory T Cells in the Context of Cancer Immunotherapy.”
Marco Donia (Professor at University of Copenhagen (Københavns Universitet)
Tumor-infiltrating lymphocyte Cell Therapy in melanoma: not as hard as we thought?
- More information (Annals of Oncology, Renske ten Ham et al., Aug 2025)
- Editorial (Javaid et al., 2025)
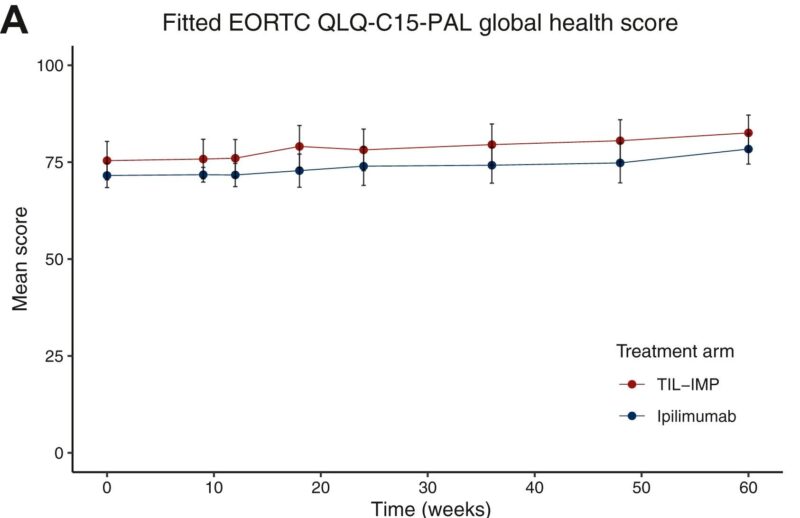
We analyzed health-related quality of life (HRQoL) in the phase III trial of TIL (tumor-infiltrating lymphocyte) therapy vs ipilimumab conducted at CCIT-DK – National Center for Cancer Immune Therapy of Denmark together with The Netherlands Cancer Institute. More info on the trial results here.
Key points from the phase III study
- TIL therapy has already shown improved efficacy (PFS) over ipilimumab in PD-1–refractory metastatic melanoma (full article)
- Patients treated with TIL therapy reported similar or better global health, emotional functioning, and lower fatigue and pain compared with ipilimumab.
- Slightly higher nausea/vomiting was seen, but not clinically meaningful.
- The expected negative impact of the more intensive TIL procedure was not observed at week +24 (see editorial for context).
Take-home message
TIL therapy improves efficacy outcomes and does not impair quality of life compared with ipilimumab in this difficult-to-treat population.
It remains an intensive treatment, requiring careful patient selection – in practice, probably <25% of patients reaching 2nd line are candidate.
Clinical perspective
TIL therapy is consolidating its role as a promising treatment option that combines progression-free survival benefit with preserved quality of life.
Limitations
Short follow-up, reduced patient numbers after 24 weeks, assessment timings not capturing the highest toxicity period, and HRQoL tools not fully adapted to melanoma or cellular therapies.
Congratulations to the brilliants corresponding author and first author, respectively Valesca Retèl and Renske ten Ham, and all the collaborators from The Netherlands Cancer Institute and CCIT-DK – National Center for Cancer Immune Therapy of Denmark, including Maartje Rohaan, Mees Egeler, Renee Menezes, Troels Holz Borch, Inge Marie Svane, John Haanen and many others
Thanks to all patients and families who participated in the study, Healthcare personnel, funding agencies Kræftens Bekæmpelse (Danish Cancer Society), Region Hovedstaden, KWF Kankerbestrijding and others; A. Javaid, Sapna Patel and James Larkin for the Editorial”
Riccardo Ray Colciago (Researcher at University of Milano-Bicocca):
“Publication Alert!
I am delighted to share that our latest article
‘Real-world feasibility of combining pembrolizumab with radiotherapy in early triple-negative breast cancer’
In this prospective study, we investigated the safety and feasibility of administering adjuvant pembrolizumab concurrently with radiotherapy in patients with operable triple-negative breast cancer (TNBC).
Key findings:
Manageable acute toxicities: All patients experienced only mild (grade 1) adverse events, primarily radiodermatitis. A few developed moderate (grade 2) effects such as fatigue, nausea, or skin erythema, with no interruptions to therapy.
Conclusion: Concurrent radiotherapy and pembrolizumab appears to be feasible and well-tolerated in everyday clinical practice, offering valuable insights beyond controlled clinical trials.
This achievement is entirely the result of great teamwork!
I am grateful to my colleagues and collaborators for their dedication and commitment throughout this project.”
Scott Kopetz (Associate Vice President, Translational Integration at MD Anderson Cancer Center):
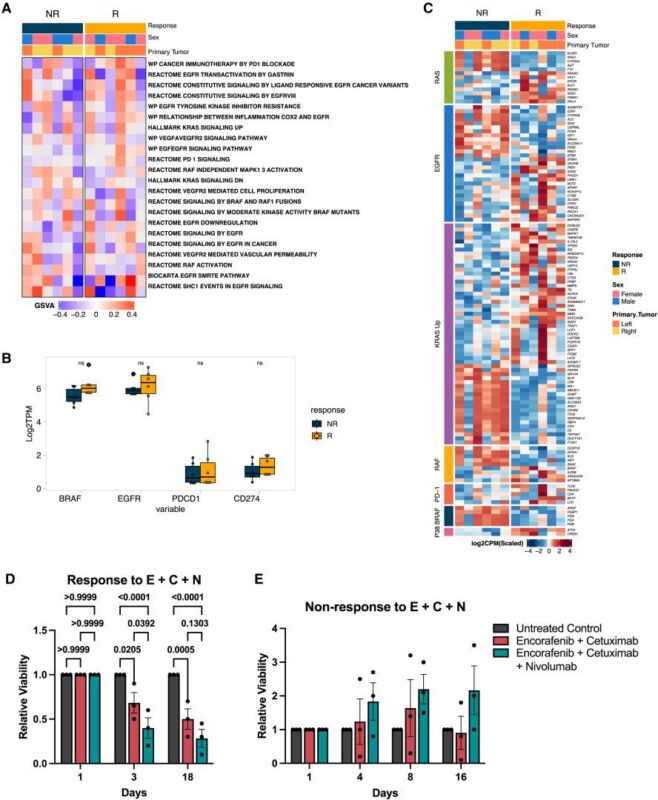
“Exciting science and clinical efficacy in this MDAnderson team science study led by Van Morris, MD In Cancer Cell.
Triple therapy with IO led to durable responses in BRAF V600E MSS colorectal cancer, showing promise. Randomized study ongoing.”
Patrick Hwu (President and CEO at Moffitt Cancer Center):
“Why do some patients respond better to immunotherapy?
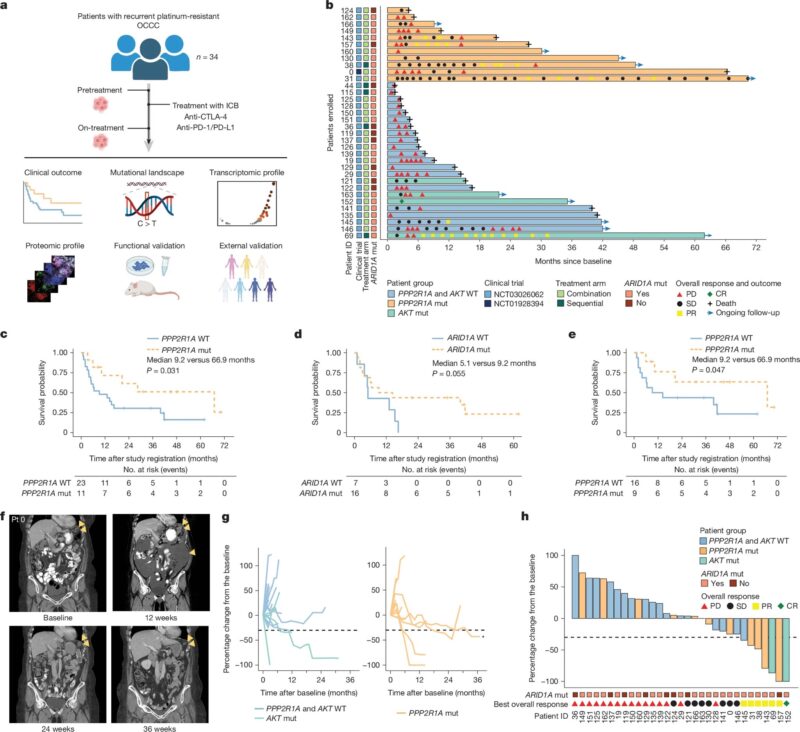
A new study in Nature found that tumors with mutations in PPP2R1A – a gene encoding a scaffold protein in the PP2A complex — were linked to much longer survival after immune checkpoint blockade (ICB).
In ovarian clear cell carcinoma and other cancers, PPP2R1A mutations were tied to stronger immune signals, more tumor-infiltrating T cells, and better tumor control. Preclinical models confirmed that altering or inhibiting PPP2R1A boosted the effectiveness of ICB and CAR-T therapies.
These findings suggest PPP2R1A mutations could serve as a predictive biomarker for immunotherapy success and highlight PP2A as a potential therapeutic target to make treatments more effective.
Thank you, Amir Jazaeri for your visit and lecture Moffitt Cancer Center this week!”
Fred R. Hirsch (Executive Director at the Center for Thoracic Oncology at Mount Sinai Hospital):
“Finally, new guidelines for immunotherapy for lung cancer and mesotheliomas! However, treatment is evolving very fast!”
Filipe Pereira (Professor, Wallenberg Fellow in Molecular Medicine at Lund University):
“New in Immunity: We have mapped the transcription factor ‘routes’ that program two dendritic cell subsets opening paths for personalized cancer immunotherapy.
Here is a summary of what we discovered. Illustration by Azuravesta.”

Saul Priceman (Associate Professor at Keck School of Medicine of USC):
“Excited to share our new publication in JITC with a universal immunotherapy combination using oncolytic virus delivery of antigens (CD19t or BCMAt) to redirect T cell engagers (TCEs) to target solid tumors.
This study expands on our foundational work published in hashtag#ScienceTranslationalMedicine in 2020 showing effective redirection of CD19-CAR T cells to solid tumors using oncolytic viruses carrying CD19t. Congrats Anthony K Park, and all our collaborators! This work is now in clinical trial and is the basis for other groups who are entering this field of combining oncolytic viruses with cell therapies to eradicate solid tumors.”
Ulrike Harjes (Senior Editor at Nature Medicine):
“New out in NatureMedicine Nature Portfolio
Immunotherapy and senolytics in head and neck squamous cell carcinoma: phase 2 trial results by Song Fan.
Two phase 2 trials, along with translational analysis of prospective cohorts and experimental analysis, indicate that immunosenescence as a mechanism of resistance to =immunotherapy can be overcome with the senolytics hashtag#dasatinib and quercetin in patients with head and neck squamous cell carcinoma.”
John Gordon (co-Founder, Director, VP Scientific Affairs at Celentyx Ltd):
“Cancer | Immunotherapy | Constructing the Cure: Engineering the Next Wave of Antibody and Cellular Immune Therapies | A Roadmap from the Society for Immunotherapy of Cancer (SITC) | Open Access Article Now Breaking at JITC
Immuno-oncology has revolutionized cancer treatment by mobilizing the immune system to eliminate tumors. Although immune checkpoint inhibitors and T cell therapies have mediated durable responses in hematologic malignancies and select solid tumors, most patients still relapse or fail to respond.
To overcome these limitations, novel, next-generation immune constructs, including bispecific-cell therapy combinations, armored cells, tethered cytokines, immune-stimulatory antibody conjugates, and in vivo gene editing, are being developed to enhance specificity, persistence, and immune activation.
This Society for Immunotherapy of Cancer roadmap highlights emerging technologies that integrate, redirect, or potentiate immune response.
Here, Stefanie Bailey, Eric Bartee, Kyle Daniels, Christopher Heery, Pravin Kaumaya, Gregory Lesinski, et al (corresponding authors Chrystal Paulos and Avery Posey, Jr) examine advances in construct design, strategies for clinical translation, and opportunities for combinatorial approaches.
By addressing translational barriers and real-world challenges, we outline how innovative engineering can unlock a new era of safe, durable, and accessible immunotherapies. Realizing this potential will require coordinated efforts from researchers, clinicians, industry leaders, and policymakers to deliver curative outcomes to patients worldwide.”
Written by Rima Grigoryan, MDc, Assistant Editor of OncoDaily IO.


