Sintilimab plus lenvatinib was evaluated as conversion therapy followed by sequential surgery (SILENSES ) for patients with advanced unresectable hepatocellular carcinoma. The study explored whether this combination of a PD-1 inhibitor and a multikinase inhibitor could help patients with initially unresectable HCC become eligible for surgery. This question is clinically important because many patients with HCC are diagnosed at an advanced stage, when curative-intent resection is not possible and systemic therapy is usually the main treatment option.
In this study, 120 treatment-naïve patients received sintilimab plus lenvatinib as conversion therapy, and tumor resectability was reassessed during treatment by a multidisciplinary team. Successful conversion was achieved in 56% of patients, and 60 patients underwent curative-intent surgery. With a median follow-up of 41 months, the overall cohort had a median overall survival of 36 months, while patients who proceeded to surgery achieved a 5-year overall survival rate of 73.9%.
These findings suggest that sintilimab plus lenvatinib may create a surgical opportunity for selected patients with unresectable HCC, although the single-arm design means the results need confirmation in larger comparative studies.
Title: Sintilimab (PD-1 inhibitor) plus lenvatinib as conversion therapy followed by sequential surgery (SILENSES) for advanced unresectable hepatocellular carcinoma: a phase II, expansion trial
Authors: Shichun Lu, Wenwen Zhang, Junfeng Li, Ze Zhang, Bingyang Hu, Xuerui Li, Yinbiao Cao, Zhijun Wang, Zhanbo Wang, Huiyi Ye, Baolin Qu, Yu Li, Guangyu Ma, Tao Wan, Zhe Liu, Bing Liu, Yinzhe Xu, Haowen Tang, Xiao Zhao, Liru Pan, Li Yang, Jing Yuan, Ruiqing Chen & Xiangbing Bian
Background
Hepatocellular carcinoma remains one of the most challenging solid tumors, especially when diagnosed at an advanced stage. Although curative-intent surgery offers the best chance for long-term survival in early-stage disease, more than half of patients with HCC present with advanced disease, where upfront resection is often not possible. The SILENSES phase II expansion trial explored an important clinical question: can systemic therapy with the PD-1 inhibitor sintilimab plus the multikinase inhibitor lenvatinib shrink or control initially unresectable HCC enough to allow surgery, and can this strategy translate into durable survival benefit?
The study, published in Signal Transduction and Targeted Therapy, evaluated sintilimab plus lenvatinib as conversion therapy followed by sequential surgery in patients with advanced unresectable HCC.
Methods
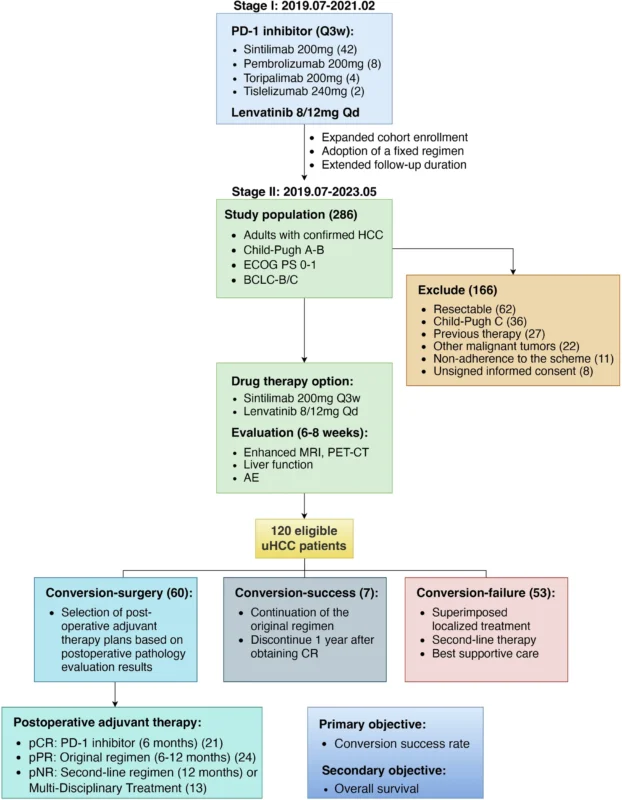
SILENSES was a prospective, single-arm, expansion phase II trial conducted at the Chinese PLA General Hospital. Between July 2019 and May 2023, 286 patients with HCC were screened, and 120 treatment-naïve patients with radiologically confirmed unresectable HCC were enrolled. All patients received sintilimab 200 mg intravenously every 3 weeks plus oral lenvatinib once daily, dosed by body weight: 12 mg for patients weighing at least 60 kg and 8 mg for those below 60 kg. Tumor resectability was reassessed every 6 to 8 weeks, beginning before the fourth treatment cycle.
Successful conversion required preserved performance status and liver function, adequate future liver remnant, controlled or resectable extrahepatic disease, and intact vascular inflow and outflow of the remaining liver. Surgery was offered after multidisciplinary team evaluation and patient consent. The primary endpoint was conversion success rate. Secondary endpoints included overall survival, recurrence-free survival, objective response rate by independent imaging review, R0 resection rate, pathological response, and safety.
Study Design
The trial population represented a high-risk HCC group. More than 80% of patients had macrovascular invasion, over 90% had BCLC stage C disease, approximately half had alpha-fetoprotein levels above 400 ng/mL, and 33% had extrahepatic metastases. Most patients were male, had hepatitis B virus infection, and had Child-Pugh class A liver function. This is important because the study did not focus on borderline resectable early tumors, but rather on patients with clearly advanced disease features.
The treatment strategy was built around two steps. First, patients received systemic conversion therapy with sintilimab plus lenvatinib. Second, patients who met predefined conversion criteria could proceed to curative-intent hepatectomy. Postoperative therapy was adapted according to pathological response: patients with pathological complete response received sintilimab monotherapy for 6 months, while those with residual viable tumor continued sintilimab-lenvatinib for longer durations.
Results
Sintilimab plus lenvatinib demonstrated meaningful antitumor activity. Successful conversion was achieved in 67 of 120 patients, giving a conversion rate of 56%. Among these 67 patients, 60 underwent curative-intent surgical resection after multidisciplinary evaluation and consideration of patient preference. Notably, 51 of 60 surgical patients, or 85%, reached curative resection criteria within 2 to 5 months of conversion therapy, with a median time of 3.2 months.
Radiologic response was also strong. By independent imaging review, the objective response rate was 58.3% by mRECIST and 45.8% by RECIST v1.1. Target lesion reduction was observed in 93% of evaluable patients by mRECIST and 90% by RECIST v1.1. Imaging response was significantly associated with successful conversion under both response criteria, with P < 0.001.
Among the 60 patients who underwent surgery, pathological outcomes were clinically notable. Pathological complete response was observed in 21 patients, representing 35% of the surgical cohort. Another 25 patients, or 42%, had pathological partial response, while 14 patients, or 23%, had pathological nonresponse. Surgical procedures included major hepatectomy in 34 patients, portal vein tumor thrombectomy in 21 patients, hepatic vein tumor thrombectomy in 3 patients, and regional lymphadenectomy in 14 patients.
With a median follow-up of 41 months, the median overall survival in the full cohort was 36 months. The estimated overall survival rates at 12, 36, and 60 months were 70.8%, 49.1%, and 42.6%, respectively. The survival difference between patients who underwent surgery and those who did not was substantial. In the surgical group, the estimated 12-, 36-, and 60-month overall survival rates were 93.3%, 79.2%, and 73.9%. In the nonsurgical group, the corresponding rates were 48.3%, 20.0%, and not estimable at 60 months, with a median overall survival of 12 months. The stratified hazard ratio for death was 0.15, with a 95% CI of 0.08–0.28 and P < 0.001.
The investigators also performed a landmark analysis to reduce immortal time bias. This analysis continued to show significantly longer survival among patients who underwent sequential surgery after conversion, with a stratified hazard ratio for death of 0.23 and P < 0.001. In multivariable analysis, decreased AFP during conversion therapy and surgical intervention after successful conversion remained independent protective factors for overall survival.
Recurrence-free survival was evaluated in patients who achieved curative-intent resection. Excluding two patients with R1 resection, the median postoperative follow-up was 38 months. Recurrence occurred in 26 of 58 patients, or 45%. The median recurrence-free survival was 40 months, with estimated RFS rates of 72.0% at 12 months, 53.0% at 36 months, and 45.4% at 48 months. Depth of pathological response was the only identified risk factor for both overall survival and recurrence-free survival in the surgical group.
Key Findings
The most important finding from SILENSES is that more than half of patients with initially unresectable HCC achieved successful conversion with sintilimab plus lenvatinib. This is clinically relevant because conversion created a pathway to potentially curative surgery in a population with advanced disease biology.
The second key finding is the survival difference after surgery. Patients who underwent resection after successful conversion achieved a 5-year overall survival rate of 73.9%, compared with poor outcomes in the nonsurgical group. Although this was not a randomized comparison, the magnitude of difference supports the importance of reassessing surgical candidacy during effective systemic therapy.
The third finding is that pathological response matters. Patients with deeper pathological response had better survival outcomes, suggesting that radiologic shrinkage alone may not fully capture the biological effect of therapy. Pathological complete response occurred in 35% of resected patients, showing that sintilimab plus lenvatinib can induce deep tumor regression in selected patients.
Safety was manageable but not negligible. Treatment-related adverse events occurred in 92% of patients, and grade 3–5 treatment-related adverse events occurred in 31%. The most common adverse events were hypertension, rash, hand-foot syndrome, pyrexia, and nausea. Four fatal treatment-related events were reported, including immune-related myocarditis and gastrointestinal hemorrhage. Among surgical patients, grade III postoperative complications occurred in 7%, grade IV complications in 17%, and no deaths were attributed to surgical complications.
Key Takeaway Messages
Sintilimab plus lenvatinib achieved a 56% conversion rate in initially unresectable HCC.
Among conversion responders, 60 patients proceeded to curative-intent surgery.The full cohort had a median overall survival of 36 months and a 5-year overall survival rate of 42.6%.Patients who underwent surgery after successful conversion achieved a 5-year overall survival rate of 73.9%. Median recurrence-free survival after curative-intent surgery was 40 months.Pathological response emerged as an important prognostic marker for both recurrence-free and overall survival.
The study supports multidisciplinary reassessment of resectability during systemic therapy, but randomized trials are needed before this approach can be considered definitive.
Conclusion
The SILENSES phase II expansion trial suggests that sintilimab plus lenvatinib may be an effective conversion strategy for selected patients with initially unresectable hepatocellular carcinoma. The regimen achieved a 56% conversion success rate, produced objective responses in nearly half to more than half of patients depending on response criteria, and allowed 60 patients to undergo curative-intent resection. The most striking result was the 73.9% 5-year overall survival rate among surgical patients, compared with markedly poorer survival in those who did not undergo surgery.
These findings strengthen the concept that effective systemic therapy can create a surgical window in advanced HCC. However, the study’s single-arm, single-center design, limited sample size, and absence of a randomized control group mean that the results need careful interpretation. The data are promising, especially for patients with good liver function and measurable treatment response, but further prospective multicenter studies are needed to define optimal patient selection, timing of surgery, and postoperative treatment strategies.