A new preclinical study published in JCI Insight explores a novel immunotherapy strategy based on noncoding double-stranded DNA delivered by lipid nanoparticles (DNA-LNPs). The central idea is biologically compelling: instead of relying solely on checkpoint inhibition to release existing antitumor immunity, DNA-LNPs may actively generate or amplify immune activation by engaging cytosolic DNA-sensing pathways, thereby converting poorly inflamed tumors into more immunologically responsive ones.
The investigators show that intracellular delivery of noncoding dsDNA by LNPs induced robust cytokine release, CD8+ T cell recruitment, and meaningful antitumor activity across several murine cancer models, including hepatocellular carcinoma, acute myeloid leukemia, melanoma, and melanoma lung metastasis. Importantly, this effect was not reproduced by empty LNPs, supporting the conclusion that the immune activation was driven by the DNA cargo itself, rather than by the lipid carrier alone.
Background
Immune checkpoint inhibitors have transformed oncology, but durable benefit remains limited to a subset of patients, particularly those with preexisting T cell–inflamed or “hot” tumors. One major challenge in immuno-oncology is therefore how to induce effective immunity in tumors that lack sufficient baseline immune activation.
The rationale for DNA-LNP therapy is based on activation of innate immune sensors that detect cytosolic DNA, particularly STING and AIM2. These pathways can trigger inflammatory cytokines, type I interferons, and downstream adaptive immune responses. The authors hypothesized that noncoding DNA-LNPs could serve as a potent immune stimulant capable of enhancing endogenous antitumor immunity and sensitizing tumors to PD-L1 blockade.
Study Design and Experimental Models
The study evaluated DNA-LNPs in multiple mouse cancer models:
- genetically induced hepatocellular carcinoma
- chemically induced hepatocellular carcinoma
- acute myeloid leukemia
- subcutaneous B16-F10 melanoma
- B16-F10 melanoma lung metastasis
The investigators tested DNA-LNPs both as monotherapy and in combination with anti–PD-L1 therapy. Mechanistic studies included cytokine profiling, immune cell infiltration analyses, CD8 and NK depletion experiments, NanoString transcriptomics, qRT-PCR, immunohistochemistry, and flow cytometry.
Key Results
DNA-LNPs induced potent innate immune activation
Across models, DNA-LNP treatment caused rapid induction of inflammatory cytokines and interferon-related signals, including:
- IFN-β
- TNF-α
- IL-6
- IL-27
- CCL2
- IFN-γ
- in some settings, IL-1β and IL-18
This pattern is consistent with activation of cGAS-STING and AIM2 inflammasome pathways. Notably, empty LNPs did not induce comparable cytokine responses, particularly after intravenous dosing, indicating that the immunostimulatory effect required DNA delivery into cells.
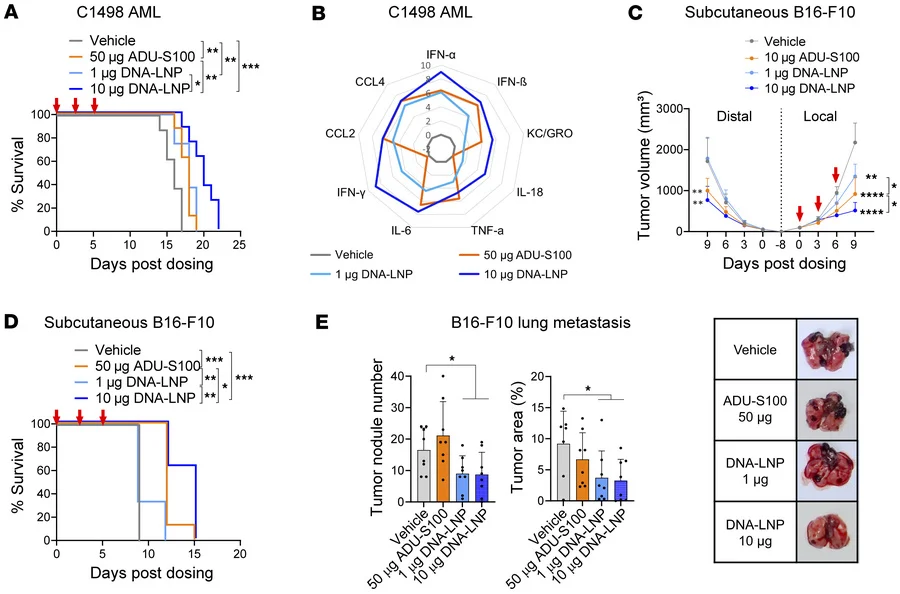
Strong antitumor activity was observed across multiple tumor types
In the hepatocellular carcinoma models, even a single dose of DNA-LNP produced marked tumor control and prolonged survival. In the genetically induced HCC model, treated animals survived far longer than vehicle-treated controls, and their livers showed substantially lower tumor burden at endpoint. Similar antitumor activity was confirmed in the chemically induced HCC model, where DNA-LNP reduced tumor nodule counts.
In nonhepatic models, DNA-LNPs also demonstrated broad antitumor activity:
- In acute myeloid leukemia, DNA-LNP prolonged survival in a dose-dependent manner and outperformed the STING agonist ADU-S100 in this model.
- In subcutaneous melanoma, DNA-LNP reduced growth of both treated tumors and, at higher dose, contralateral untreated tumors, suggesting a broader immune-mediated effect.
- In melanoma lung metastasis, DNA-LNP reduced lung tumor burden, whereas ADU-S100 showed no meaningful antitumor effect under the tested conditions.
Together, these results support DNA-LNPs as a generalizable immune-activating strategy, rather than one restricted to a single histology or route of administration.
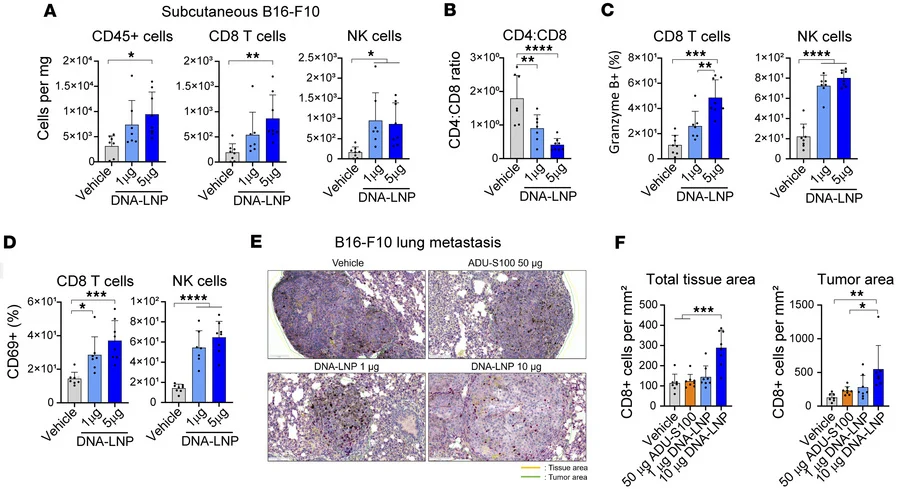
The antitumor effect was CD8+ T cell–dependent
One of the most important mechanistic findings was that DNA-LNP treatment increased CD8+ T cell infiltration into tumor sites and enhanced markers of immune activation.
In liver tumors, transcriptomic and RT-PCR analyses showed enrichment of cytotoxic cell and T cell signatures shortly after treatment. In melanoma, DNA-LNP increased:
- total immune cell infiltration
- CD8+ T cell infiltration
- NK cell infiltration
- granzyme B expression
- CD69 expression on CD8+ T cells and NK cells
However, depletion studies showed that the therapeutic effect depended primarily on CD8+ T cells:
- In HCC and AML, CD8 depletion essentially abolished the antitumor benefit.
- In melanoma, CD8 depletion substantially reduced efficacy, although not completely, suggesting that additional immune populations may contribute in some settings.
This establishes DNA-LNPs as a strategy capable of promoting functionally relevant CD8-dependent antitumor immunity.
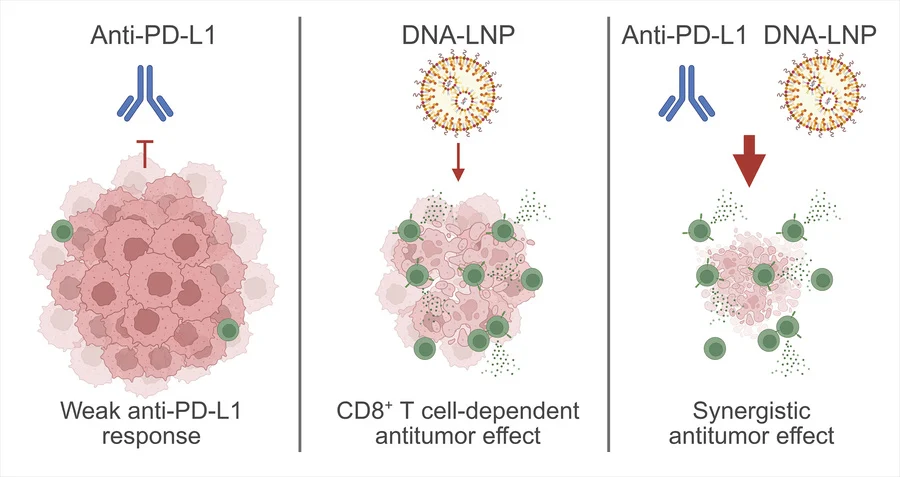
DNA-LNPs enhanced anti–PD-L1 activity in both responsive and resistant settings
A particularly important translational finding was the synergy between DNA-LNPs and anti–PD-L1 therapy.
In the HCC model, when therapy was initiated under more advanced disease conditions, neither anti–PD-L1 alone nor low-dose DNA-LNP alone was sufficient for durable long-term control. However, the combination produced a striking therapeutic effect. At higher DNA-LNP dose plus anti–PD-L1, most animals remained disease-free at study endpoint; even low-dose DNA-LNP combined with anti–PD-L1 achieved durable disease control in a substantial proportion of mice.
In the B16-F10 melanoma model, which is resistant to anti–PD-L1 monotherapy, anti–PD-L1 alone had no significant effect. Yet when combined with DNA-LNP, tumor growth control improved and survival was prolonged.
This is one of the most relevant observations in the paper: DNA-LNPs were able to sensitize or resensitize tumors to PD-L1 blockade, including a model intrinsically resistant to checkpoint inhibition.
Mechanistic Interpretation
The data support a coherent immunologic model. DNA-LNPs deliver noncoding dsDNA into cells, where it activates cytosolic DNA sensors including STING and AIM2. This leads to induction of inflammatory cytokines and interferon signaling, which in turn promotes recruitment and activation of cytotoxic lymphocytes, especially CD8+ T cells. Once a more inflamed tumor microenvironment is established, PD-L1 blockade becomes more effective.
In other words, DNA-LNPs appear to function as an innate immune primer that may convert “cold” tumors into more “hot,” checkpoint-responsive tumors.
Why this study matters
This work is notable for several reasons.
First, it moves beyond the idea of checkpoint inhibition as a standalone therapy and instead addresses one of the field’s biggest biological limitations: the absence of preexisting antitumor immunity.
Second, the approach appears versatile. Activity was demonstrated across multiple tumor models and with both local and systemic delivery, suggesting that the platform may be adaptable.
Third, the synergy with anti–PD-L1 in an ICI-resistant melanoma model is especially important, because it suggests a route to overcome primary resistance to checkpoint blockade.
Fourth, unlike some prior innate immune agonist approaches, the study suggests that DNA-LNPs may engage multiple innate immune pathways simultaneously, potentially producing broader immune activation than single-pathway agonists.
Limitations
Despite its promise, this remains a preclinical study and should be interpreted accordingly.
Several important limitations apply:
- all efficacy data are from mouse models
- toxicity and cytokine-related tolerability in humans remain unknown
- biodistribution of standard LNP formulations may favor the liver, which may be advantageous for hepatic
- tumors but less optimal for other sites
- the precise contribution of STING versus AIM2 and other immune populations still requires deeper mechanistic dissection
- optimal dosing, schedule, and tumor-targeting strategies remain to be defined
The authors also note that human immune systems may be more sensitive to LNP-associated inflammation than murine systems, which makes careful translational development essential.
Key Takeaway Messages
- Noncoding DNA delivered by lipid nanoparticles induced strong innate immune activation and meaningful antitumor effects across multiple mouse cancer models.
- The therapeutic effect was largely CD8+ T cell–dependent, with increased tumor infiltration and activation of cytotoxic lymphocytes.
- DNA-LNPs synergized with anti–PD-L1 therapy, including in a checkpoint-resistant melanoma model, supporting their potential as a sensitizing strategy for immunotherapy.
- The study positions DNA-LNPs as a promising innate immune–priming platform that may broaden the population of patients who benefit from checkpoint blockade
You Can Read All Article Here