Metastatic breast cancer is still the main cause of breast cancer–related mortality, despite major progress in endocrine therapy, targeted agents, antibody-drug conjugates, and immunotherapy. Much of metastatic breast cancer research has focused on tumor-intrinsic biology, including receptor status, genomic alterations, and mechanisms of treatment resistance. Yet metastasis is not sustained by cancer cells alone. It also depends on the host environment, particularly the systemic immune and vascular landscape that can either restrain or enable tumor dissemination. A new prospective study brings this idea into sharper focus by showing that metastatic breast cancer is associated with a coordinated circulating program of immune exhaustion, regulatory expansion, myeloid suppression, inflammatory activation, and angiogenic remodeling, all captured through liquid biopsy (Kurma et al., 2026).
This is what makes the study especially interesting. It does not look only at circulating tumor cells, and it does not treat liquid biopsy as a narrow tool for counting tumor-derived material in blood. Instead, it uses a multilayered strategy to examine three interrelated dimensions of metastatic disease at the time of stage IV diagnosis and before first-line metastatic treatment: circulating tumor cells, immune cell phenotypes, and plasma protein signals. In doing so, it presents metastatic breast cancer not simply as disseminated tumor burden, but as a systemic immunovascular disease with clinically relevant blood-based markers of progression (Kurma et al., 2026).
© The Author(s) 2026.
Why This Question Matters
The idea that the metastatic process is shaped by the host immune environment is not new, but it has been difficult to translate into clinically useful tools. Tumor-infiltrating lymphocytes, immune checkpoints, and microenvironmental signals have all been studied in primary and metastatic tissue. However, tissue biopsies remain spatially limited and cannot easily capture the broader systemic state. Blood, by contrast, offers a dynamic and accessible window into how tumor cells interact with immune cells, inflammatory mediators, and vascular remodeling pathways in real time. That is particularly relevant in metastatic breast cancer, where dissemination, immune escape, and colonization of distant organs are not local events but systemic processes (Kurma et al., 2026).
Kurma and colleagues built their study around this concept. They hypothesized that integrating immunophenotypic and proteomic information from peripheral blood with clinical features could reveal mechanisms of progression and identify clinically actionable markers. Their final conclusion is ambitious but well supported by the data they generated: metastatic breast cancer should be understood as a systemic immunovascular disorder, and circulating IL-8 and NOS3 may serve as important prognostic biomarkers within that framework (Kurma et al., 2026).
Study Design and Patient Population
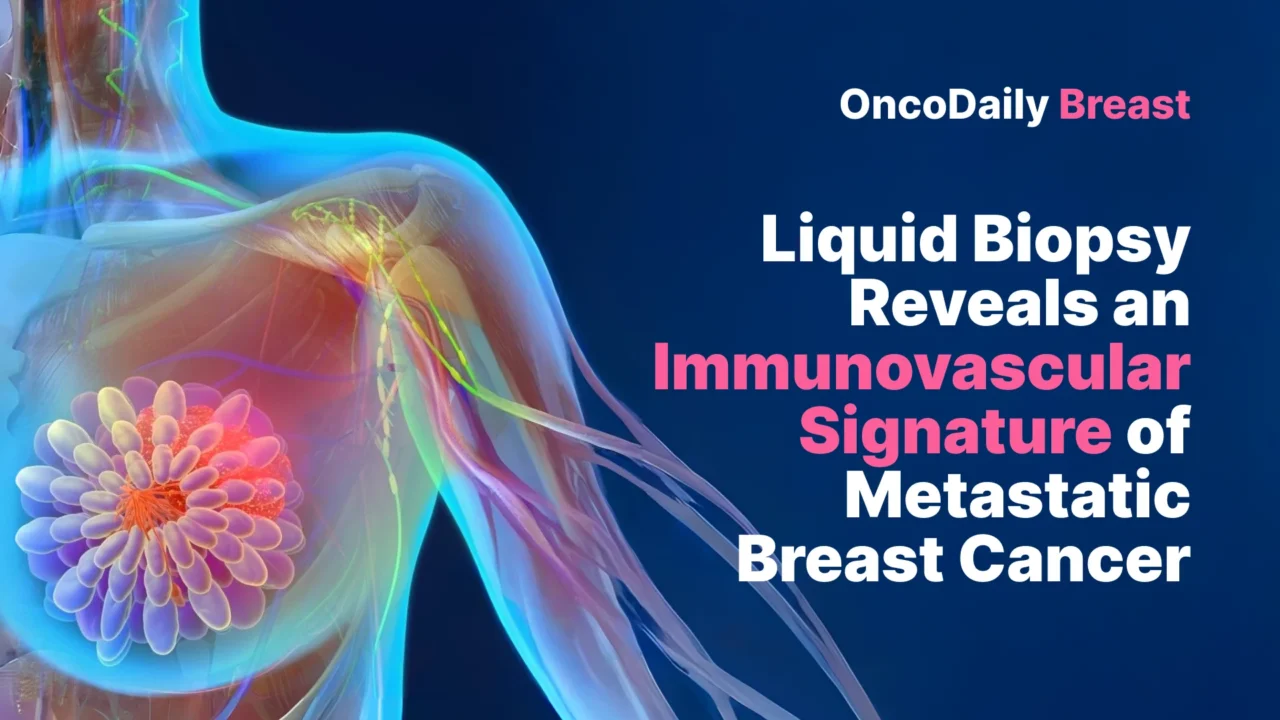
The study prospectively enrolled 60 patients with histologically confirmed stage IV breast cancer in the ALCINA 2 clinical trial. Blood samples were collected before treatment initiation for metastatic disease. For comparison, the investigators also recruited 21 age-matched healthy female donors. This design is important because it avoids the confounding effect of early metastatic therapy on circulating immune cells and plasma proteins, allowing a cleaner view of the baseline metastatic state (Kurma et al., 2026).
The cohort mainly reflected luminal metastatic breast cancer. Most patients, 83.3%, had hormone receptor–positive and HER2-negative disease, while smaller groups had HR-positive/HER2-positive, HR-negative/HER2-positive, or triple-negative tumors. Bone was the most frequent site of metastasis, followed by liver, lung, and lymph nodes, and more than half of patients had metastases in multiple organs. Median follow-up was 31.5 months, and median progression-free survival was 19.9 months. Circulating tumor cells were detected in 58.3% of patients, with counts ranging from 1 to 962 CTCs per 7.5 mL of blood (Kurma et al., 2026).
That CTC range alone highlights how heterogeneous metastatic breast cancer remains, even before therapy begins. But the study moves beyond this heterogeneity by asking what kind of systemic environment is associated with CTC positivity and poorer outcome.
A Peripheral Blood Landscape Defined by T-Cell Exhaustion
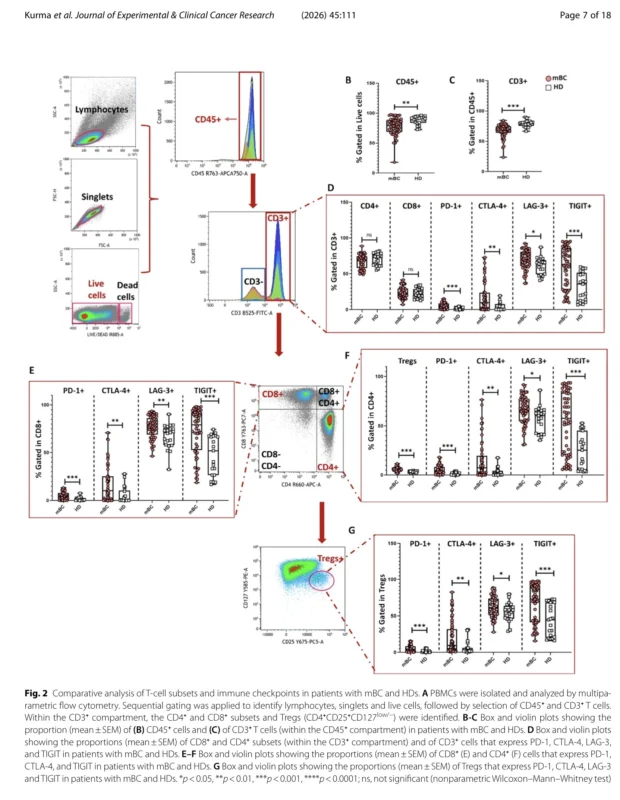
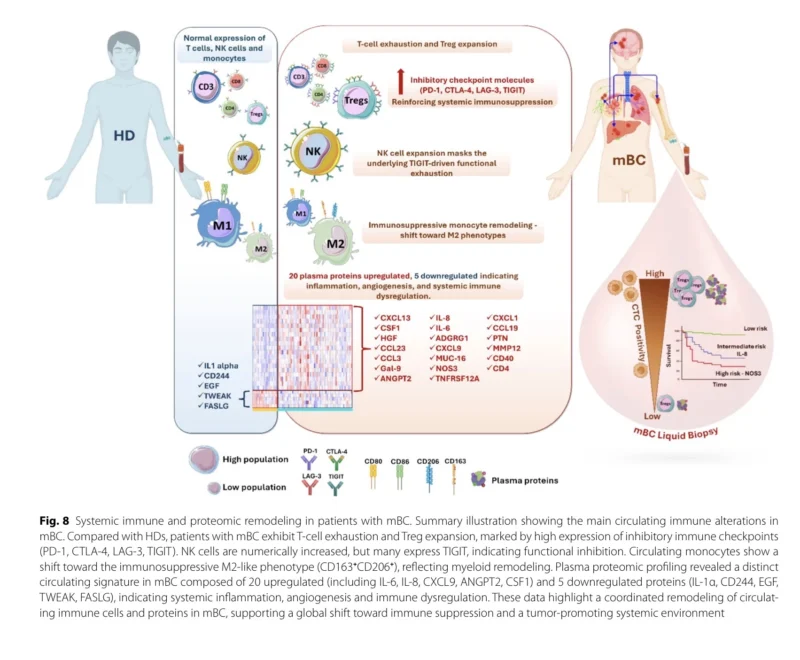
One of the clearest findings was the marked disruption of circulating T-cell populations. Compared with healthy donors, patients with metastatic breast cancer had significantly lower proportions of total leukocytes and lower proportions of CD3-positive T cells. This suggests not just immune activation, but a degree of systemic immune attrition. At the same time, CD4-positive and CD8-positive proportions within the T-cell compartment were relatively preserved, indicating that the key issue may be the global erosion and dysfunction of circulating T cells rather than a simple shift in lineage distribution (Kurma et al., 2026).
The functional phenotype of these cells was even more revealing. Patients with metastatic breast cancer had higher proportions of T cells expressing PD-1, CTLA-4, LAG-3, and TIGIT. This was seen across the broader CD3-positive population as well as within CD4-positive and CD8-positive subsets. Such co-expression of inhibitory receptors is strongly consistent with an exhausted immune phenotype. These are not immune cells that are absent from the metastatic process. They are immune cells that appear chronically engaged but functionally restrained (Kurma et al., 2026).
That distinction matters. Breast cancer, especially hormone receptor–positive disease, has often been described as immunologically “cold.” This study argues for a more nuanced interpretation. The peripheral immune system in metastatic breast cancer may not be uninvolved. Instead, it may be deeply involved but organized around inhibition rather than effective anti-tumor activity.
Regulatory T Cells Expand While Effector Immunity Contracts
The suppressive nature of this systemic immune state becomes even clearer when looking at regulatory T cells. The study found that Tregs were significantly increased in patients with metastatic breast cancer compared with healthy donors. Moreover, these Tregs expressed high levels of inhibitory checkpoint molecules, including PD-1, CTLA-4, and TIGIT. This suggests not merely a numerical expansion, but a functionally potent regulatory compartment capable of dampening immune surveillance and supporting tumor persistence (Kurma et al., 2026).
These findings are important because they frame metastatic breast cancer as an immune ecosystem in which suppressive populations are actively maintained. It is not simply that effector immunity becomes weak. It is that regulatory mechanisms become dominant. In practical terms, that may help explain why metastatic disease often progresses despite the presence of immune cells and why response to immune-based strategies remains inconsistent across breast cancer subtypes.
NK Cells and Monocytes Show a Similar Pattern of Dysfunction
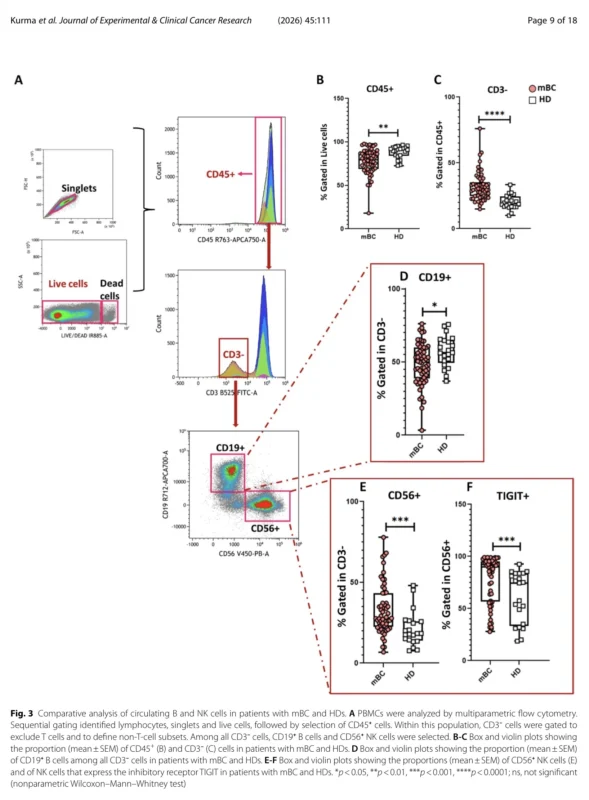
The study also found changes outside the T-cell compartment that support the same broader story. Natural killer cells were increased in circulation, but many of these NK cells expressed TIGIT, an inhibitory receptor associated with reduced cytotoxicity. This creates a paradoxical picture: NK cells are present in greater numbers, yet appear functionally constrained. Since NK cells are central to the surveillance of circulating tumor cells in the bloodstream, TIGIT-mediated restraint may be one mechanism by which CTCs survive and seed distant organs (Kurma et al., 2026).
The monocyte compartment showed another layer of systemic remodeling. Patients with metastatic breast cancer had an increased proportion of CD163-positive, CD206-positive monocytes, a phenotype consistent with alternatively activated, immunosuppressive M2-like cells. These cells are associated with tissue repair, immune dampening, and tumor-supportive behavior. By contrast, monocytes expressing CD80 and CD86, markers more aligned with immune stimulation and antigen presentation, tended to be lower. Together, these findings suggest a peripheral myeloid environment that is shifted toward tolerance, remodeling, and tumor support rather than immune activation (Kurma et al., 2026).
Plasma Proteomics Show Inflammation and Angiogenesis at the Core
Perhaps the most translationally compelling part of the study was the plasma proteomic profiling. Using the Olink 96 Immuno-Oncology panel, the investigators identified a distinct circulating signature in metastatic breast cancer composed of inflammatory cytokines, chemokines, angiogenic mediators, immune regulators, and tumor-associated proteins. Among the proteins increased in patients with metastatic disease were IL-6, IL-8, HGF, ANGPT2, NOS3, CSF1, and galectin-9. Several chemokines linked to trafficking and inflammation were also elevated, while proteins such as FASLG and TWEAK were reduced (Kurma et al., 2026).
This pattern is biologically coherent. IL-8 is widely linked to inflammation, invasion, and metastatic behavior. HGF and ANGPT2 support motility, endothelial activation, and vascular remodeling. NOS3 is closely tied to nitric oxide production and vascular function. CSF1 can help sustain immunosuppressive macrophage and monocyte programs. Galectin-9 contributes to immune exhaustion and regulatory signaling. Meanwhile, the reduction in FASLG and TWEAK suggests attenuation of apoptosis-related and immune-activating pathways. Altogether, the plasma data describe a host environment that is chronically inflamed, pro-angiogenic, and immunologically permissive (Kurma et al., 2026).
CTC Positivity Reflects More Than Tumor Burden
One of the most valuable insights from the study is that circulating tumor cells were not isolated from the rest of the biology. CTC positivity correlated with a higher proportion of regulatory T cells and with increased levels of IL-8, HGF, galectin-9, and TNFRSF12A. In patients with at least 5 CTCs, IL-8, galectin-9, CD4, and TNFRSF12A were even more strongly elevated. This suggests that CTCs are not just markers of dissemination. They are also markers of the biologic conditions that allow dissemination to succeed (Kurma et al., 2026).
That is a meaningful conceptual shift. CTC burden may reflect not only how many cells have entered the bloodstream, but how permissive the immune and vascular environment has become for their survival. In this model, circulating tumor cells are both a tumor readout and a host-environment readout.
© The Author(s) 2026.
IL-8 and NOS3 Emerge as Independent Prognostic Markers
The most clinically actionable finding may be the survival analysis. In univariate analyses, liver metastases, hormone receptor loss, triple-negative phenotype, and higher CTC burden were associated with shorter progression-free survival. But when the investigators moved to multivariable Cox regression, two soluble factors retained independent prognostic significance: IL-8, with a hazard ratio of 1.49, and NOS3, with a hazard ratio of 4.72. These markers outperformed many conventional variables in the final model (Kurma et al., 2026).
This does not mean IL-8 and NOS3 are ready for immediate routine use in clinic. The cohort is relatively small, and the findings need validation in larger independent populations. But it does suggest that blood-based immune and vascular markers may eventually refine how clinicians stratify risk in metastatic breast cancer. If confirmed, these markers could help identify patients with a more aggressive systemic phenotype, even within conventional receptor-defined groups.
Clinical Meaning and Future Direction
The broader significance of this study is not only the identification of two biomarkers. It is the framework it proposes. Metastatic breast cancer is presented here as a disease sustained by coordinated immune exhaustion, regulatory dominance, NK-cell inhibition, myeloid polarization, inflammatory signaling, and vascular remodeling. That framework has therapeutic implications. It points toward a future in which liquid biopsy may help guide not just tumor monitoring, but immunologic and stromal risk assessment as well (Kurma et al., 2026).
The authors appropriately stress that the data are correlative and hypothesis-generating. The cohort was modest, overall survival was not mature, and treatment heterogeneity limits broader conclusions. Yet the work is still important because it provides a clinically accessible portrait of metastatic biology at the time treatment begins. It shows that blood can capture far more than circulating tumor cells alone. It can reveal the ecosystem that allows metastasis to persist.