Neoadjuvant therapy has become a central part of treatment for HER2-positive breast cancer, not only because it increases the likelihood of breast conservation, but also because it offers an early readout of treatment sensitivity through pathologic complete response (pCR). In this disease subtype, pCR is more than a surrogate curiosity. It is closely linked to long-term outcomes and has helped shape modern HER2-directed treatment strategies (Cortazar et al., 2014; van Mackelenbergh et al., 2023).
For years, dual HER2 blockade with trastuzumab and pertuzumab, combined with chemotherapy, has defined the neoadjuvant standard. Yet one practical question continues to divide clinicians: how much chemotherapy is truly necessary once potent HER2 blockade is in place? More specifically, does carboplatin meaningfully improve outcomes when added to a taxane plus trastuzumab and pertuzumab, or does it mainly add toxicity?
A multicenter real-world study from China by Wu and colleagues, published in the World Journal of Surgical Oncology, addresses this question in a way that feels highly relevant to everyday practice. Rather than asking whether one regimen is universally better, the study asks something more clinically useful: which patients may still benefit from carboplatin, and which patients may reasonably receive a less toxic de-escalated regimen (Wu et al., 2024)
That question has become even more important in light of recent prospective data presented at the 2025 ASCO Annual Meeting from the phase 3 neoCARHP trial. As Paolo Tarantino, MD, PhD, commented, “These data were impressive because we would have expected the carboplatin to add to the efficacy of THP. Instead, in the setting of HER2 blockade, it doesn’t seem to add much.” In neoCARHP, the pCR rate was 64.1% with THP alone versus 65.9% with TCHP, a difference of only -1.8% (95% CI, -8.5% to 5.0%; P = .0089), suggesting that in at least some patients, carboplatin may contribute little additional efficacy while clearly increasing hematologic toxicity (Tarantino et al., 2026).
Against that backdrop, the Chinese real-world data become especially interesting, because they suggest the answer may not be binary.
Why This Question Matters
HER2-positive breast cancer accounts for roughly 15% to 20% of invasive breast cancers and is biologically aggressive, with increased proliferation and invasiveness compared with HER2-negative disease (Oh & Bang, 2020). The introduction of trastuzumab transformed prognosis, and subsequent trials established the importance of dual HER2 blockade with trastuzumab and pertuzumab in the neoadjuvant setting (Gianni et al., 2012; Shao et al., 2020).
Yet the chemotherapy backbone remains an area of active refinement. Carboplatin-containing regimens such as TCbHP or TCHP are widely used because they often produce high pCR rates. But they also bring greater hematologic and gastrointestinal toxicity, and not every patient is equally fit for such treatment. This is particularly relevant in real-world settings, where age, comorbidity, frailty, and treatment tolerance often influence regimen choice as much as trial-based efficacy data.
The key clinical challenge is not simply whether de-escalation is possible. It is whether de-escalation can be done intelligently, without sacrificing the chance of cure.
The Study Design: Real-World, Pragmatic, and Clinically Useful
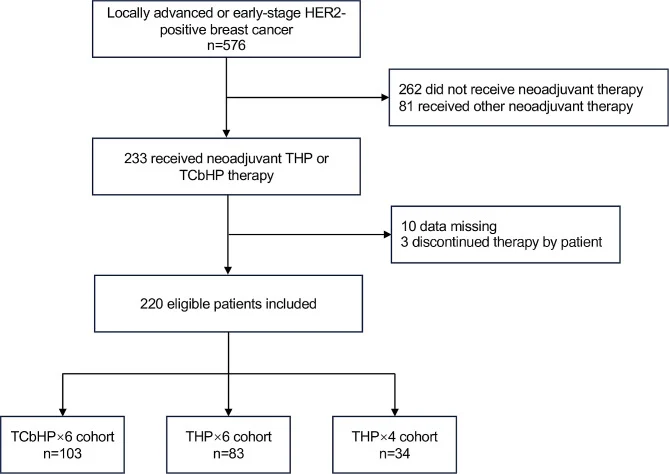
Wu and colleagues conducted a multicenter retrospective real-world study in China, including women with HER2-positive breast cancer treated with neoadjuvant therapy between March 2019 and February 2023 (Wu et al., 2024). Eligible patients received one of three regimens:
- 6 cycles of TCbHP
- 6 cycles of THP
- 4 cycles of THP
The main endpoint was pCR, defined as ypT0/is ypN0. Safety was assessed through treatment-emergent adverse events.
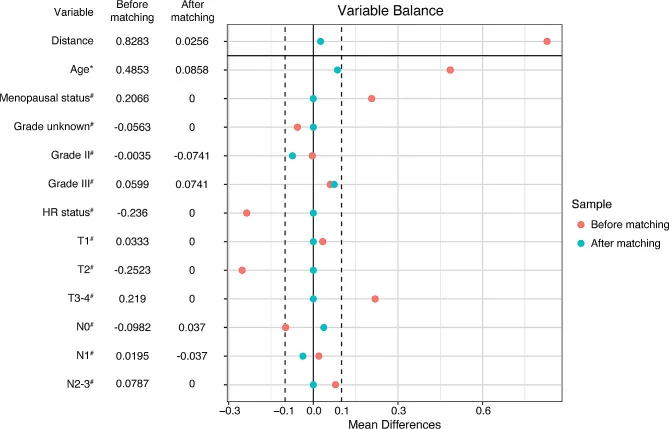
In total, 220 patients were included. Of these, 103 received TCbHP for 6 cycles, 83 received THP for 6 cycles, and 34 received THP for 4 cycles. The investigators also performed propensity score matching between the THPx6 and THPx4 groups, which is important because treatment allocation was not randomized and likely reflected patient condition and physician preference.
This is not a randomized phase 3 trial, and that limitation matters. But real-world analyses can be especially valuable when the question is not purely efficacy under ideal conditions, but regimen selection in the messy context of routine practice.
The Main Efficacy Signal: TCbHP Numerically Higher, But Not Dramatically So
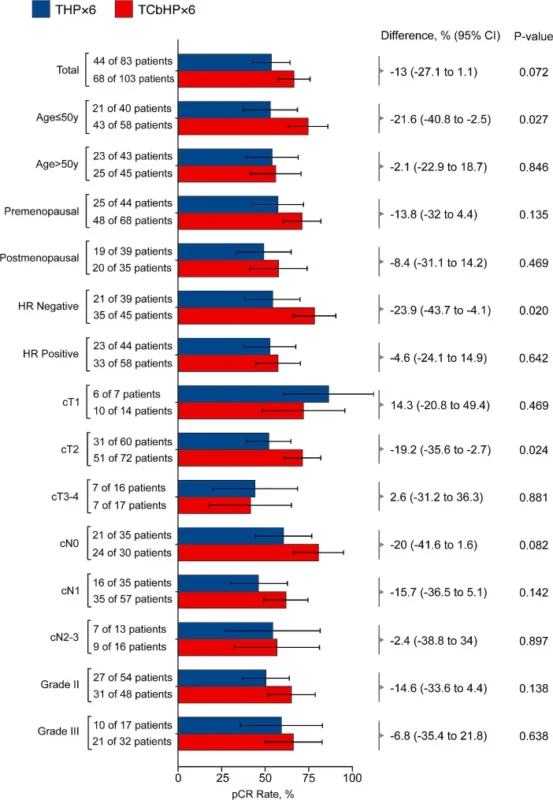
In the overall population, the pCR rate was 66% with TCbHPx6 and 53% with THPx6, with a P value of 0.072 (Wu et al., 2024). Statistically, that did not meet conventional significance, but clinically it remains notable. A 13-point absolute difference in pCR cannot simply be dismissed, especially in a curative-intent setting. At the same time, the lack of formal statistical significance suggests that the benefit of carboplatin may be more selective than universal.
This is precisely where the study becomes more useful than a simple regimen-versus-regimen comparison.
Rather than concluding that carboplatin should always be omitted or always retained, the investigators examined which subgroups appeared to derive greater benefit. They found that TCbHPx6 significantly outperformed THPx6 in patients aged 50 years or younger, in those with hormone receptor-negative disease, and in those with clinical T2 tumors (Wu et al., 2024).
That pattern makes biological and clinical sense. Younger patients are often more chemotherapy-sensitive and can better tolerate intensive treatment. Hormone receptor-negative HER2-positive tumors tend to achieve higher pCR rates overall and may be more chemosensitive than HR-positive disease. Clinical T2 tumors occupy an intermediate-risk space where intensification may still move the needle meaningfully, unlike very small tumors where dual blockade may already be enough, or very large tumors where even intensification may not fully overcome adverse biology.
The De-Escalation Story: THP May Be Enough for Some Patients
The study’s most practice-relevant message may be what it suggests about THP.
The THPx6 regimen produced a pCR rate of 53% in the overall group. That is not trivial. It means more than half of patients achieved pCR without carboplatin. In a disease where dual HER2 blockade is highly active, that level of efficacy matters, especially if it comes with substantially lower toxicity.
This finding becomes even more compelling when placed beside the emerging prospective neoCARHP data discussed by Tarantino. In neoCARHP, THP alone achieved a pCR rate of 64.1%, compared with 65.9% for TCHP. The very small difference surprised many investigators because the expectation had been that carboplatin would meaningfully increase efficacy. As Tarantino noted, in the setting of modern HER2 blockade, that added value may be much smaller than previously assumed.
He further emphasized the safety advantage of omitting carboplatin. THP was well tolerated, and while peripheral neuropathy remained a concern because taxanes were still used, hematologic toxicity was substantially lower without carboplatin. He also noted that the neoCARHP findings, together with phase 2 CompassHER2 pCR data, support carboplatin omission for at least some patients with stage II HER2-positive breast cancer, while acknowledging that this has not yet been confirmed for stage III disease because recurrence-free survival and overall survival data are still immature.
This is the key nuance. The de-escalation argument is no longer speculative. It is increasingly data-supported. But it is not yet fully settled across all risk groups.
Four Cycles Are Probably Not Enough
If the Chinese study supports de-escalation in selected patients, it also sends a strong warning against undertreatment.
One of the most important findings was the comparison between 6 cycles and 4 cycles of THP. After propensity score matching, the pCR rate was 63% with THPx6 versus only 29.6% with THPx4 (P = 0.029) (Wu et al., 2024). That is a major difference and likely the most clinically actionable result in the paper.
This means that if clinicians choose to omit carboplatin, they should not assume a shorter course of THP will preserve efficacy. In this study, duration mattered. The message is clear: de-escalation of intensity does not justify de-escalation of adequate treatment exposure.
This finding also fits with what has been observed across earlier neoadjuvant HER2-directed trials. NeoSphere and PEONY used shorter regimens and reported pCR rates in the range of 39% to 46% for trastuzumab-pertuzumab-taxane combinations, while longer exposure has often yielded higher pCR rates depending on patient mix and regimen design (Gianni et al., 2012; Shao et al., 2020; Hatschek et al., 2021).
So if THP is chosen as a lower-toxicity alternative, it appears that 6 cycles should be regarded as the minimum effective course rather than an optional intensification.
Toxicity: The Real Price of Carboplatin
The safety findings strongly favored THPx6 over TCbHPx6. Almost all patients receiving TCbHP experienced adverse events of any grade, and nearly half had grade 3 to 4 toxicity. By contrast, grade 3 to 4 events occurred in only 12% of patients receiving THPx6 (Wu et al., 2024).
The difference was particularly striking for hematologic toxicity. In the TCbHP group, grade 3 to 4 leukopenia and neutropenia each occurred in 23.3% of patients, and febrile neutropenia occurred in 19.4%. Gastrointestinal toxicity, including nausea and vomiting, was also clearly more frequent. Nearly one-third of patients receiving TCbHP required dose reduction or regimen adjustment, and carboplatin discontinuation occurred in 19 patients.
In contrast, no patients in the THPx6 cohort required dose reduction or regimen adjustment.
This is not a minor point. In a curative setting, clinicians often accept substantial toxicity to maximize efficacy. But the justification for doing so weakens when the incremental benefit becomes small or limited to certain subgroups. If a regimen adds a great deal of toxicity while only marginally improving pCR in many patients, then careful patient selection becomes essential.
How Should These Findings Be Interpreted Alongside neoCARHP?
This is where the article becomes most interesting. The Chinese real-world data and the neoCARHP discussion are not contradictory. They are complementary.
The real-world study suggests that carboplatin still has a role in some patients, particularly younger individuals, those with HR-negative disease, and those with cT2 to higher-stage tumors. The neoCARHP discussion suggests that in the broader context of strong HER2 blockade, carboplatin may not meaningfully improve pCR for many patients, especially in stage II disease.
Taken together, these data suggest a more personalized approach:
- For lower-risk or more chemotherapy-sensitive patients where dual HER2 blockade already delivers strong pCR rates, THP may be enough.
- For biologically higher-risk or clinically more advanced patients, carboplatin may still add value.
- For patients who are frail, older, or unable to tolerate hematologic toxicity, THPx6 appears to offer a reasonable lower-toxicity alternative.
- For patients in whom THP is chosen, shortening to 4 cycles should probably be avoided.
That is a more mature treatment philosophy than the old question of whether everyone needs TCHP. The answer increasingly appears to be no.
What This Means for Clinical Practice in 2026
The broader field is clearly moving toward risk-adapted neoadjuvant therapy in HER2-positive breast cancer. That means not every patient needs the same amount of chemotherapy, and not every regimen should be chosen purely on the basis of maximal pCR at any cost.
The Chinese study offers a practical framework. Wu and colleagues suggest favoring TCbHPx6 in patients aged 50 years or younger and in those aged above 50 but not older than 60 if they have HR-negative disease or cT2-4 tumors. They suggest THPx6 as a lower-toxicity alternative in patients with poorer general condition or without those higher-risk features (Wu et al., 2024).
That recommendation feels increasingly aligned with where the field is going. It also matches Tarantino’s interpretation of neoCARHP and CompassHER2 pCR, where carboplatin omission may be reasonable in selected stage II patients but remains less secure for stage III disease until recurrence-free survival and overall survival data mature.
The most important point is that de-escalation should not be equated with undertreatment. It should be deliberate, biologically informed, and based on the best available efficacy and toxicity data.
Limitations That Should Not Be Ignored
This study has real strengths, but it also has important limitations. It is retrospective and nonrandomized. Treatment allocation was influenced by physician judgment and patient preference. The comparison between TCbHP and THP therefore remains vulnerable to selection bias. In addition, the study focuses on pCR rather than long-term outcomes, and event-free survival data were not yet available (Wu et al., 2024).
That matters because pCR is strongly associated with prognosis, but it is not identical to event-free survival or overall survival. A regimen that modestly increases pCR at the cost of major toxicity may not always improve what ultimately matters most to patients.
That is why neoCARHP and CompassHER2 pCR are so important. They push the conversation beyond retrospective observation toward prospective de-escalation evidence. But even there, long-term survival endpoints are still needed.
Conclusion
The de-escalation debate in HER2-positive breast cancer is no longer theoretical. It is now supported by emerging prospective data and strengthened by real-world evidence.
Wu and colleagues show that six cycles of THP can provide meaningful efficacy with substantially less toxicity than TCbHP, while also making clear that four cycles are not enough. Their subgroup findings suggest carboplatin still matters in selected patients, particularly younger women, those with HR-negative disease, and those with clinically more significant tumors (Wu et al., 2024).
Placed in context with neoCARHP, the message becomes even sharper. As Paolo Tarantino observed, the absence of a major efficacy gain from carboplatin in the setting of dual HER2 blockade is striking. The field expected carboplatin to add more. Instead, its benefit may be narrower than assumed, while its toxicity remains very real.
That does not mean carboplatin should disappear. It means HER2-positive neoadjuvant therapy is entering a more individualized era. The next important step will be long-term recurrence and survival data. Until then, the best interpretation is probably this: THPx6 is a valid lower-toxicity option for selected patients, TCbHPx6 remains appropriate for higher-risk subgroups, and shorter THP should be avoided.
Read full article here