A new large retrospective study has added an unexpected and provocative layer to the conversation around metabolic health and breast cancer. In an analysis published in JAMA Network Open, GLP-1 receptor agonist use was associated with improved survival and recurrence outcomes in women with breast cancer who also had obesity or type 2 diabetes. The findings are attention-grabbing not only because of the size of the dataset, but because they suggest these agents may have effects that extend beyond glycemic control and weight loss. At the same time, the study is observational, not randomized, and its effect sizes are strong enough that they demand careful interpretation rather than immediate clinical enthusiasm.
Why This Question Matters
Patients with breast cancer who also have obesity or type 2 diabetes often face worse outcomes than those without these metabolic conditions. GLP-1 receptor agonists are already widely used for diabetes and obesity management, and their use has increased rapidly in recent years. That made them a logical group of drugs to study in breast cancer populations, especially because preclinical work has suggested possible anticancer effects and because weight reduction itself may influence long-term outcomes. The central clinical question was whether GLP-1 receptor agonist use around and after breast cancer diagnosis might be associated with lower all-cause mortality and improved recurrence-free survival.
How The Study Was Built
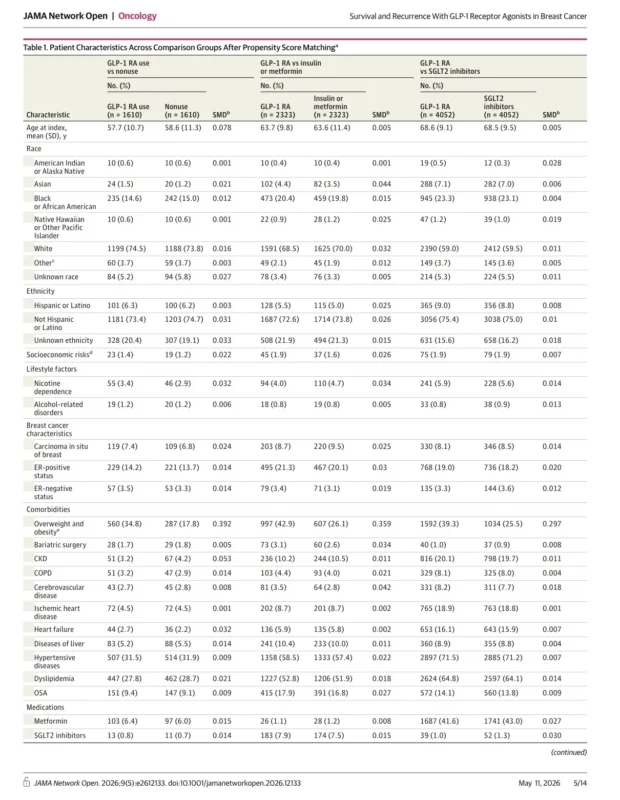
The investigators used the TriNetX US Collaborative Network and identified 841,831 eligible adult women with breast cancer across 68 healthcare organizations. They then performed 1:1 propensity score matching to create clinically comparable groups in three key analyses. The first compared GLP-1 receptor agonist users with nonusers among patients with obesity but without type 2 diabetes. The second compared GLP-1 receptor agonist users with patients receiving insulin or metformin among those with type 2 diabetes. The third compared GLP-1 receptor agonist users with patients receiving SGLT2 inhibitors among those with type 2 diabetes. The primary outcome was all-cause mortality, and the secondary outcome was recurrence-free survival over a 10-year follow-up window.
This design is important because it tries to answer a more nuanced question than a simple exposed-versus-unexposed comparison. By separating obesity and diabetes contexts, and by using active comparators like insulin, metformin, and SGLT2 inhibitors, the study attempted to reduce some of the obvious biases that often weaken retrospective analyses. Still, as the authors themselves note, causality cannot be established from this type of dataset.
The Obesity Cohort Produced One of the Strongest Signals
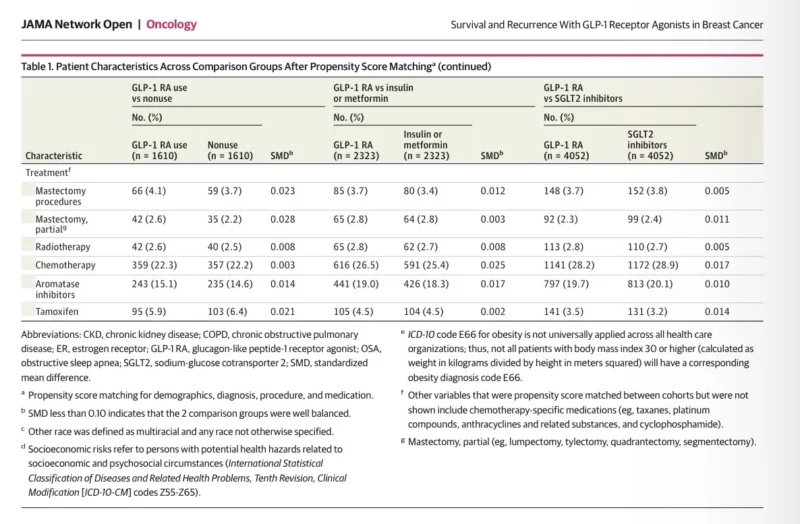
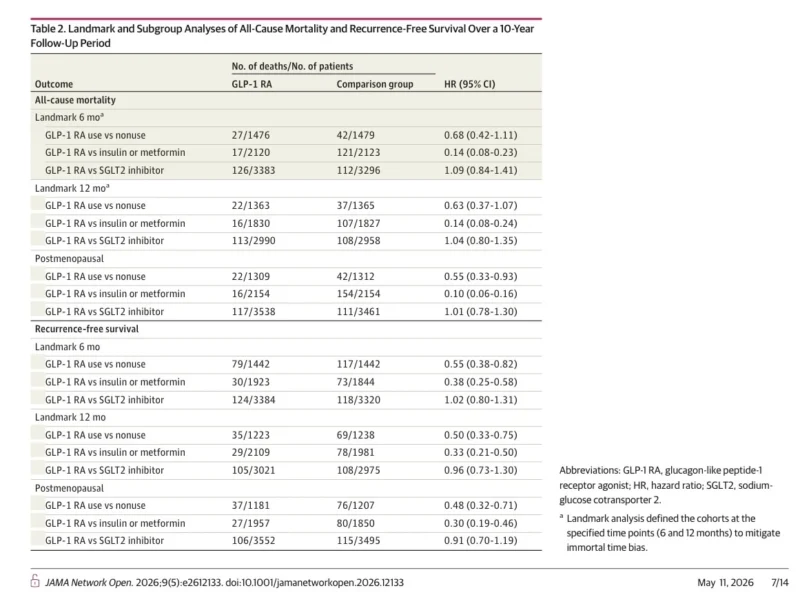
Among patients with breast cancer and obesity, GLP-1 receptor agonist use was associated with a substantially lower hazard of all-cause mortality compared with nonuse. The reported hazard ratio was 0.35 in unadjusted analyses and remained significant after adjustment. The recurrence-free survival result was also notable, with an unadjusted hazard ratio of 0.44 and a significant adjusted association as well. The 5- and 10-year survival probabilities in this matched obesity cohort favored GLP-1 receptor agonist users, and the authors interpreted these data as suggesting a potentially meaningful long-term association between GLP-1 receptor agonist exposure and improved outcomes in this group.
This is the result that will likely attract the most attention, because it points toward a possible therapeutic overlap between metabolic intervention and cancer outcomes. But it is also the finding that requires the most discipline in interpretation. Retrospective EHR-based studies can capture real-world associations, yet they are especially vulnerable to residual confounding, treatment-selection bias, differences in healthcare engagement, and unmeasured disease biology.
The Type 2 Diabetes Cohort Also Favored GLP-1 RAs Against Insulin or Metformin
The signal became even more dramatic in the comparison between GLP-1 receptor agonists and insulin or metformin among patients with type 2 diabetes. In that matched cohort, GLP-1 receptor agonist use was associated with a lower hazard of all-cause mortality, with a reported hazard ratio of 0.09, and a lower hazard of recurrence-free survival events, with a hazard ratio of 0.33 in unadjusted analyses. Adjusted analyses also remained significant. These numbers are unusually strong for a nonrandomized oncology-adjacent study, which is exactly why they are both intriguing and difficult to accept at face value.
There are at least two ways to read this. The optimistic interpretation is that GLP-1 receptor agonists could be contributing to a biologically meaningful improvement in breast cancer outcomes through weight loss, insulin regulation, cardiovascular benefit, inflammatory modulation, or direct tumor-related effects. The more cautious interpretation is that patients who receive GLP-1 receptor agonists may differ in important but incompletely measured ways from those treated with insulin or metformin, including overall health, treatment access, healthcare follow-up, or cardiometabolic care intensity. The current study cannot fully separate those possibilities.
The SGLT2 Comparison Was More Complicated
The comparison against SGLT2 inhibitors is especially valuable because it places GLP-1 receptor agonists against another modern metabolic drug class rather than older diabetic therapies. In this analysis, the unadjusted comparison did not show a significant difference in all-cause mortality or recurrence-free survival between GLP-1 receptor agonists and SGLT2 inhibitors. In adjusted analyses, a lower hazard was seen for GLP-1 receptor agonists, but the overall signal was far less dramatic than in the insulin/metformin comparison. In postmenopausal subgroup and 12-month landmark analyses, protective associations versus SGLT2 inhibitors were not sustained.
This part of the study may be the most clinically informative. If the observed benefit of GLP-1 receptor agonists disappears or weakens meaningfully when compared with another contemporary metabolic therapy, then the story becomes less about a unique anticancer effect and more about broader metabolic optimization, comparator selection, or residual differences between treatment populations. That does not weaken the importance of the findings, but it does shift how confidently they can be framed.
Why Oncologists Will Pay Attention
Even with its limitations, this study lands at a moment when breast oncology is becoming increasingly interested in the role of body composition, insulin resistance, and metabolic intervention. Weight change after diagnosis, obesity at diagnosis, and diabetes have all been linked to worse cancer outcomes in earlier work. If a widely used metabolic drug class is now associated with better long-term survival and lower recurrence in selected breast cancer populations, that naturally raises questions about whether these agents could eventually become part of supportive or even adjunct oncologic care.
It also matters that these findings did not come from a tiny exploratory dataset. The starting population exceeded 841,000 patients, even though the matched analytic cohorts were much smaller. That scale gives the paper visibility and makes it harder to dismiss, even if the final message remains cautious.
The Study’s Limitations Are Not Minor
The authors are appropriately careful in their conclusions. The study was retrospective, relied on structured EHR data, and used coded definitions for recurrence rather than centrally adjudicated oncologic outcomes. Exposure was defined through prescriptions rather than confirmed adherence. Patient-level weight change data were unavailable, so the analysis cannot explain whether the observed differences were driven by weight loss, metabolic control, cardiovascular effects, direct tumor biology, or differences in patient selection. The paper also notes substantial administrative censoring around five years, which limits the precision of later survival estimates even though the models extended to ten years.
Those limitations are not technical footnotes. They are central to interpretation. A result can be statistically compelling and still not be practice-changing. In this case, the study raises an important hypothesis, but it does not prove that GLP-1 receptor agonists improve breast cancer survival.
What Comes Next
The most appropriate next step is not immediate routine use of GLP-1 receptor agonists as a breast cancer outcome intervention. It is prospective testing. The authors explicitly call for randomized clinical trials, and that is the correct conclusion. Future studies will need to clarify whether any signal differs by menopausal status, endocrine therapy exposure, obesity phenotype, tumor subtype, or duration and timing of GLP-1 receptor agonist use. They will also need to determine whether the apparent benefit is unique to GLP-1 receptor agonists or reflects a broader effect of improved metabolic control.
Bottom Line
This study is one of the more provocative breast oncology signals of the year. In women with breast cancer and obesity or type 2 diabetes, GLP-1 receptor agonist use was associated with improved survival and lower recurrence risk in several matched comparisons. But the strongest results came from retrospective analyses with major potential for residual confounding, and the signal weakened in the comparison against SGLT2 inhibitors. The clinical message is not that GLP-1 receptor agonists should now be viewed as anticancer therapy. It is that metabolic therapy may matter more to breast cancer outcomes than many clinicians have assumed, and that this question is now important enough to deserve serious prospective study.


