Patients with stage III colon cancer are typically treated with surgical resection followed by adjuvant chemotherapy with a fluoropyrimidine-plus-oxaliplatin regimen. However, approximately 30% of patients with stage III disease experience recurrence despite such treatment.
Deficient mismatch repair (dMMR) colon cancers represent a biologically distinct subgroup, characterized by microsatellite instability-high status, hypermutation, immune checkpoint up-regulation, and abundant predicted neoantigens. Although dMMR colorectal cancer has shown responsiveness to immune checkpoint blockade in the metastatic setting, whether immunotherapy could improve outcomes in the adjuvant treatment of resected stage III dMMR colon cancer had remained unclear.
In this context, Sinicrope and colleagues conducted the phase 3 ATOMIC trial to evaluate whether the addition of atezolizumab, an anti–programmed death ligand 1 (PD-L1) antibody, to modified FOLFOX6 (mFOLFOX6) could improve outcomes in patients with resected stage III dMMR colon cancer.
The article was published in The New England Journal of Medicine on March 25, 2026.
Title: Atezolizumab plus FOLFOX for Stage III Mismatch Repair–Deficient Colon Cancer
Authors: Frank A. Sinicrope, Fang-Shu Ou, Dirk Arnold, Walter R. Peters, Robert J. Behrens, Christopher H. Lieu, Khalid Matin, Deirdre J. Cohen, Samara L. Potter, Andrew B. Nixon, Lisa A. Kottschade, Emily Kathol, Wendy L. Frankel, Ardaman Shergill, Dennis Hsu, Anke Reinacher-Schick, Paul Mehan, Philip J. Gold, Maged F. Khalil, Tyler Zemla, Clare Gatten, Eileen M. O’Reilly, and Jeffrey A. Meyerhardt
Methods
ATOMIC was an international, randomized, phase 3 trial conducted through the Alliance for Clinical Trials in Oncology and the National Clinical Trials Network, with participation from sites in the United States and Germany. Eligible patients were 12 years of age or older and had completely resected stage III colon adenocarcinoma with dMMR status confirmed locally, with retrospective central confirmation also performed.
Patients with known Lynch syndrome were eligible. Prior chemotherapy or radiotherapy was not allowed, except for one cycle of mFOLFOX6 when needed while awaiting MMR results.
Patients were randomly assigned in a 1:1 ratio to one of two treatment strategies:
- Atezolizumab plus mFOLFOX6 for 6 months, followed by atezolizumab monotherapy to complete 12 months of total therapy
- mFOLFOX6 alone for 6 months
The primary endpoint was disease-free survival (DFS), defined as the time from randomization to recurrence or death from any cause. Secondary endpoints included overall survival (OS) and safety. The trial was powered to detect a hazard ratio of 0.6 for DFS, and interim analyses were prespecified. At the second interim analysis, the data and safety monitoring board determined that the primary endpoint had been met.
Previous results from ASCO 2025
The results were previously presented at ASCO 2025, where the ATOMIC trial reported improved disease-free survival with the addition of atezolizumab to adjuvant mFOLFOX6 in patients with resected stage III dMMR colon cancer.
At that analysis, with a median follow-up of 37.2 months and 124 disease-free survival events, the 3-year disease-free survival was 86.4% in the atezolizumab plus mFOLFOX6 group versus 76.6% in the mFOLFOX6-alone group, corresponding to a hazard ratio for recurrence or death of 0.50 (95% CI, 0.35–0.72; p<0.0001).
The benefit was reported across analyzed subgroups, including older patients and both low- and high-risk disease.
Results from 2026
Between September 2017 and January 2023, 712 patients with stage III dMMR colon cancer were enrolled across 312 sites. A total of 355 patients were assigned to receive atezolizumab plus mFOLFOX6 and 357 patients to receive mFOLFOX6 alone. The median age was 64 years, 55.1% of the patients were women, and 83.7% had proximal tumors. Overall, 46.1% had clinically low-risk tumors (Tx, T1, T2, or T3; N1 or N1c), and 53.9% had high-risk tumors (T4, N2, or both). Baseline characteristics and stratification variables were well balanced between the two groups.
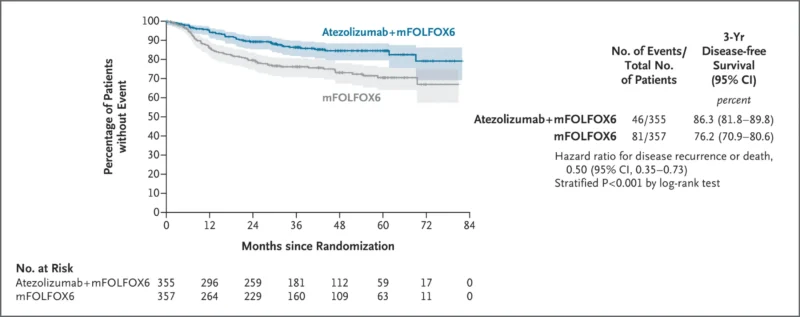
At a median follow-up of 40.9 months, 127 DFS events had occurred: 46 in the atezolizumab-plus-mFOLFOX6 arm and 81 in the chemotherapy-alone arm. The addition of atezolizumab significantly improved disease-free survival.
Key efficacy findings included:
- 3-year DFS: 86.3% with atezolizumab plus mFOLFOX6 vs 76.2% with mFOLFOX6 alone
- Hazard ratio for recurrence or death: 0.50 (95% CI, 0.35–0.73)
- P value: P<0.001
- The absolute improvement in 3-year DFS was approximately 10 percentage points
- Findings were consistent in centrally confirmed dMMR and per-protocol analyses
A prespecified subgroup analysis suggested that the benefit of atezolizumab may have depended on adequate chemotherapy exposure. Among patients who received more than six cycles of mFOLFOX6, the hazard ratio for recurrence or death was 0.41 (95% CI, 0.27–0.64). In contrast, among those who received six cycles or fewer, no clear advantage was seen, with a hazard ratio of 0.97 (95% CI, 0.44–2.11). These findings are exploratory and suggest, but do not prove, that the benefit of adjuvant atezolizumab may depend on adequate chemotherapy exposure.
At the time of this analysis, overall survival data were immature. With a median follow-up for overall survival of 45.8 months, there had been 31 deaths in the atezolizumab arm and 33 deaths in the control arm. The 5-year overall survival was 89.7% with atezolizumab plus mFOLFOX6 and 87.9% with mFOLFOX6 alone, with no significant difference between the groups.
Safety
Safety was evaluated in all treated patients. The incidence of adverse events of any grade was similar in both groups, but grade 3 or 4 adverse events were more frequent with the addition of atezolizumab.
- Grade 3–4 adverse events: 84.1% with atezolizumab plus mFOLFOX6 vs 71.9% with mFOLFOX6 alone
- Nonhematologic grade 3–4 toxicities were more common in the atezolizumab arm (69.4% vs 54.5%).
- Fatigue was notably increased: 10.1% vs 3.3%
- Grade 3–4 neutrophil count decrease: 43.6% vs 35.9%
Immune-related adverse events such as hypothyroidism, hyperglycemia, colitis, and maculopapular rash were seen more often with atezolizumab, although the incidence of suspected immune-related grade 3 or 4 adverse events was similar between the two groups. Grade 5 adverse events occurred in six patients in the atezolizumab arm and two patients in the mFOLFOX6-alone arm; two deaths in the atezolizumab arm were considered treatment-related by investigators.
Conclusion
In this phase 3 trial, the addition of atezolizumab to mFOLFOX6 improved disease-free survival compared with mFOLFOX6 alone in patients with resected stage III dMMR colon cancer. The 3-year disease-free survival was higher in the atezolizumab group, with a hazard ratio for recurrence or death of 0.50. Overall survival data remain immature, and grade 3 or 4 adverse events were more frequent with the addition of atezolizumab.
These findings also support universal MMR testing in all patients with newly diagnosed colon cancer, both to identify Lynch syndrome and to determine eligibility for immunotherapy.
The full article is available in NEJM.





