The second week of April highlights recent discussions across GI Oncology, covering colorectal, pancreatic, gastric, hepatocellular, and esophageal cancers. These selected posts reflect ongoing work in epidemiology, tumor biology, translational research, and early clinical investigation.
Key topics include nationwide data on early-onset colorectal cancer, analyses of cellular senescence in pancreatic cancer, and preclinical models exploring tumor heterogeneity using patient-derived organoids. Mechanistic studies further examine the role of β-cell signaling in obesity-associated pancreatic cancer and stromal factors in colorectal cancer.
Clinical research updates include early-phase data on immunotherapy strategies in esophageal cancer and hepatocellular carcinoma, as well as emerging therapeutic approaches such as cMET-targeting antibody–drug conjugates in gastric cancer. In parallel, discussions continue around immunotherapy approaches in dMMR/MSI-H colorectal cancer.
Patient-related outcomes are also addressed, including long-term effects after radiotherapy for anal cancer.
Together, these posts provide a concise overview of current research directions and clinical considerations in GI oncology.
Jeremy Meyer – Digestive, Colorectal and Robotic Surgeon; Coordinator of the Colorectal Cancer Center; Associate Professor, Geneva University Hospitals (HUG), Switzerland
“Proud to share our latest publication in the European Journal of Cancer (IF 7.2), where I had the privilege to lead the first nationwide study in Switzerland on early-onset colorectal cancer, in collaboration with the Geneva Cancer Registry and the National Agency for Cancer Registration teams.
Almost 100’000 patients over 40 years
Key findings:
- 6.1% of all colorectal cancers occur before age 50
- Incidence in young adults is increasing steadily (+0.46–0.49% per year), while it is declining in older populations
- 1 in 4 young patients (27.7%) is diagnosed at stage IV
- The rise is largely driven by rectal cancer (31.8% of cases)
Red flags should never be ignored—regardless of age:
rectal bleeding, iron deficiency anemia, change in bowel habits, weight loss should prompt medical consultation.”
Nelson Dusetti – Research Director, INSERM; Pancreatic Cancer and Translational Oncology; Co-founder of Predicting Med, developing transcriptomic tools for precision oncology, Marseille, France
“Happy to share a new publication from our group on pancreatic cancer!!
In this review, we discuss the complex role of cellular senescence in pancreatic ductal adenocarcinoma. While senescence is traditionally viewed as a protective mechanism limiting oncogenic transformation, growing evidence shows that in established tumors it can also promote tumor progression through stromal remodeling, immune suppression and therapy resistance.
Understanding this dual role may open new therapeutic opportunities by targeting senescent cells or their secretory programs.This work is also the result of a long-standing Franco-Argentine scientific collaboration between researchers from the CRCM – Centre de Recherche en Cancérologie de Marseille and Institut Paoli-Calmettes in Marseille, together with colleagues from CONICET and the University of Buenos Aires.
As part of the Franco-Argentine Joint Laboratory for Pancreatic Cancer Research, Daniel Grasso recently spent time in our laboratory in Marseille, which provided the opportunity to work together on this review.
Science advances through collaboration!!”
Arndt Vogel – Head of the Center for Personalized Medicine, MHH at Medical University of Hanover, Germany
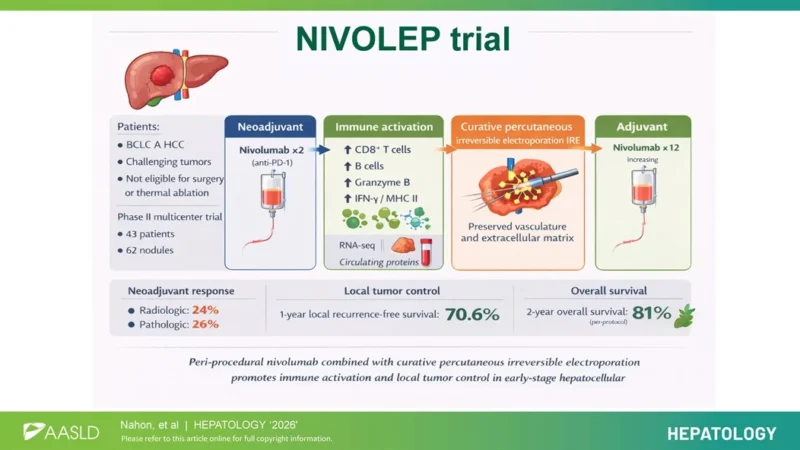
“Neoadjuvant and adjuvant nivolumab associated with irreversible electroporation in BCLC A HCC and high risk of recurrence
- 43 pts, NIVOLEP trial
- ORR 25% after Nivo
- 2yrs OS rate 75%
Not sure whether there is a future for IRI in HCC, but interesting data”
Mandar Muzumdar – Associate Professor of Genetics, Yale University School of Medicine, USA
“New paper alert. We found that β cells are bona fide drivers of obesity-associated Pancreatic Cancer progression → targeting the endocrine pancreas might prevent exocrine cancer.
Background: Obesity increases PancreaticCancer (PDAC) risk, but how? Previously, we found that obesity accelerates pancreatic tumorigenesis in mice and induces the expression of the hormone CCK in β cells, whereas weight loss reverses these effects.
New insight: Now, we show that β cell-derived CCK is necessary and sufficient for obesity-driven PDAC development, resulting in increased tumor formation near islets. What’s more? The effect is independent of insulin, challenging conventional wisdom on obesity and cancer.
Key Findings:
β cell CCK promotes tumorigenesis in lean mice
Pancreatic CCK knockout ablates obesity-driven PDAC progression
Pancreatic CCK expression, but not insulin, is positively associated with disease
Obesity increases islet-proximal tumor formationMechanism:
Obesity rewires β cell identity towards an immature, stress-adapted state in mice and humans. Obesity → increased ER stress → increased JNK/cJun activation → increased CCK expression → transcriptional alterations in islet-proximal exocrine acinar cells → increased islet-proximal tumorigenesisTechnical Innovations:
Integration of PDAC/obesity models with genetic perturbation of CCK and lineage tracing
scRNA-seq coupled with cutting-edge computational tools
In silico hypothesis generation with experimental validation in vivoTakeaways:
β cells are essential PDAC drivers
β cell stress biology links to tumorigenesis
Obesity-driven PDAC is not just about insulin. It’s CCK-dependent.
Endocrine–exocrine CCK signaling is a PDAC driver and potential target for prevention”
Jonathan Cohen – Director of Clinical Research, Sharett Institute of Oncology, Hadassah–Hebrew University Medical Center, Israel
“New research out in Clinical Cancer Research!
Our multi-center study reports on the safety and efficacy of Telisotuzumab Adizutecan (ABBV-400) in advanced gastric cancer. It’s been rewarding to play a part in the early development of this cMET targeting ADC through the AbbVie P1PER network.”
Ilyas Sahin – GI Medical Oncologist; Early-Phase Clinical Trials, Massachusetts General Hospital, Harvard Medical School, USA
“Locally advanced esophageal cancer (ESCC) remains challenging.
Phase II: adding tislelizumab (anti–PD-1) to chemo + radiation without maintenance improved outcomes, with better survival.”

David Tougeron – Professor of Oncology, Poitiers University Hospital, France
“A very important paper concerning the sensitivity of mono versus bi IO in dMMR/MSI-H mCRC.”
Haylee Duval – PhD/MBA Student in Biomedical Sciences, MaineHealth Institute for Research, USA
“Excited to share my abstract published by the American Association for Cancer Research, which I’ll be presenting in San Diego next week! This work represents a collaborative effort at the MaineHealth Institute for Research, combining expertise across the Reagan, Ryzhov, and Lindner labs, and has also been accepted for publication in Oncotarget pending minor revisions.
Using three independent in vivo studies, this work highlights the pro-tumorigenic role of host-derived CTHRC1 secreted by colon cancer stromal cells. Its absence in the tumor microenvironment increases survival, decreases tumor burden across multiple measures, and modifies both intra-tumoral and systemic immune cell landscapes. Ultimately, this work identifies CTHRC1 as a potential target for future colorectal cancer therapies and highlights the need to further investigate the mechanisms driving the protective phenotype observed in Cthrc1 KO mice.”
Johanne Hollands Steffensen – MD, PhD Student, Department of Experimental Clinical Oncology, Aarhus University Hospital, Denmark
“What should we tell our patients about sexual and vaginal side effects after radiotherapy for anal cancer?
Our systematic review in British Journal of Cancer summarises the current evidence on prevalence, risk factors, and interventions:Clinical takeaways:
• Symptoms are common and often long-term
• Higher vaginal dose may increase risk
• Evidence for prevention and rehabilitation remains limited”
Markus Tschurtschenthaler – Junior Group Leader, Technical University of Munich; Founder & CSO, Fluvion, Germany
“I am happy to share that our latest work is now published in Journal of Experimental & Clinical Cancer Research:
‘A translational colorectal cancer organoid biobank mirrors patients’ tumor histology, molecular profiles, and treatment responses’.
Colorectal cancer (CRC) is a highly heterogeneous disease – not only between patients, but also within individual tumors. Capturing this complexity in preclinical models remains a major challenge.
In this study, we established a comprehensive biobank of patient-derived organoids (PDOs) spanning the major histological CRC subtypes. Our key findings:
- PDOs retain distinct subtype-specific morphologies and molecular features
- Orthotopic transplantation generates PDOXs that recapitulate patient tumor architecture and signaling
- The PDO-PDOX cycle maintains both molecular fidelity and therapy response (e.g. FOLFOX)
Together, this establishes a robust and predictive platform that captures the full spectrum of CRC heterogeneity and enables functional precision oncology.
Many thanks and congratulations to all co-authors and collaborators, especially Moritz Jesinghaus, Miguel Silva, and Dieter Saur.”
Find out 10 Must-Read Posts in GI Oncology from the first week of April on OncoDaily.



