ZSAB-neoGOLP Trial evaluated the role of neoadjuvant GOLP—gemcitabine, oxaliplatin, lenvatinib, and the PD-1 inhibitor toripalimab—in patients with resectable intrahepatic cholangiocarcinoma with high-risk features for recurrence. Published in The New England Journal of Medicine, the phase 2–3 randomized study compared neoadjuvant GOLP followed by surgery and adjuvant capecitabine with upfront surgery followed by adjuvant capecitabine. The trial demonstrated that neoadjuvant therapy significantly prolonged event-free survival and produced meaningful tumor regression while maintaining a manageable safety profile.
Title: Neoadjuvant GOLP in Resectable High-Risk Intrahepatic Cholangiocarcinoma
Authors: Guo-Ming Shi, M.D., Xiao-Yong Huang, M.D., Fei Liang, M.D., Xiao Liang, M.D., Rui Dong, M.D., Qing-Hai Ye, M.D., Qiang Gao, M.D., Yuan Ji, M.D., Zheng-Ping Yu, M.D., Wen-Long Zhai, M.D., Jia-Cheng Lu, M.D, Xiao-Wu Li, M.D., Fu-Bao Liu, M.D., Kui Wang, M.D., Wei Yang, M.D., Jia-Lin Zhang, M.D., Zhi-Qiang Hu, M.D., Shuang-Jian Qiu, M.D., Xiao-Ying Wang, M.D., Xin-Rong Yang, M.D., Hui-Chuan Sun, M.D., Ying-Hong Shi, M.D., Zhen-Bin Ding, M.D., Wei-Ren Liu, M.D., Xiao-Wu Huang, M.D., Cheng Huang, M.D., Ying-Hao Shen, M.D., Jian Sun, M.D., Yi-Feng He, M.D., Yuan-Fei Peng, M.D., Yang Xu, M.D., Jian-Jun Zou, M.D., Jian Zhou, M.D., and Jia Fan, M.D., for the ZSAB Study Group
Background
Intrahepatic cholangiocarcinoma (ICC) is the second most common primary liver cancer and is associated with poor long-term outcomes. Even after potentially curative liver resection, recurrence rates exceed 50%, and the overall 5-year survival remains approximately 30%. Patients with high-risk features—such as large tumor size (>5 cm), multifocal disease, vascular invasion, lymph-node involvement, or elevated serum CA19-9—have particularly unfavorable outcomes due to early recurrence and rapid disease progression.
Despite these risks, no neoadjuvant therapy has been established as standard treatment for patients with resectable high-risk ICC. Current management typically involves upfront surgery followed by adjuvant capecitabine, based largely on evidence from the BILCAP trial. However, the benefit of surgery alone with adjuvant therapy remains modest in high-risk disease, and strategies aimed at reducing early recurrence are urgently needed.
Advances in systemic therapy for biliary tract cancers have suggested that combination regimens integrating chemotherapy, targeted therapy, and immunotherapy may improve outcomes. Chemotherapy can enhance tumor antigen release and immune activation, while anti-angiogenic agents such as lenvatinib may improve immune-cell infiltration and normalize tumor vasculature, thereby augmenting the effects of immune checkpoint inhibitors.
The GOLP regimen—consisting of gemcitabine, oxaliplatin, lenvatinib, and a PD-1 inhibitor—has previously demonstrated encouraging activity in advanced biliary tract cancers. Building on this biological rationale and early clinical signals, the ZSAB-neoGOLP trial evaluated whether this combination could improve outcomes when used as neoadjuvant therapy before surgery in patients with resectable high-risk ICC.
Methods
The ZSAB-neoGOLP trial was an investigator-initiated, multicenter, open-label phase 2–3 randomized controlled trial conducted across 11 hospitals in China. The study was designed to evaluate the efficacy and safety of neoadjuvant GOLP therapy followed by surgery compared with immediate surgery alone in patients with resectable high-risk intrahepatic cholangiocarcinoma.
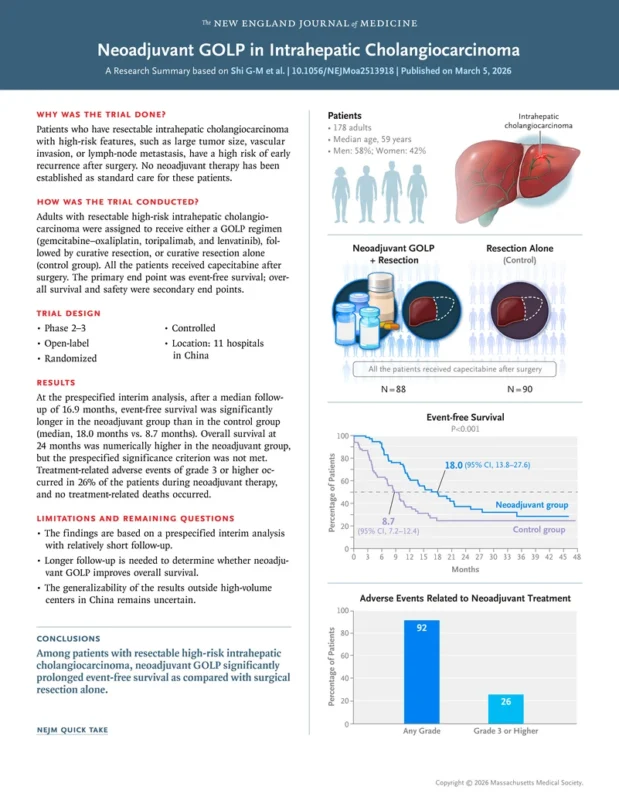
Eligible patients were aged 18–70 years with an Eastern Cooperative Oncology Group (ECOG) performance status of 0 and preserved liver function (Child–Pugh class A). All patients had resectable ICC with at least one high-risk factor for recurrence, including tumor diameter greater than 5 cm, vascular invasion, multiple intrahepatic tumors, hepatic portal lymph-node metastasis, or elevated serum CA19-9 levels.
Patients were randomly assigned in a 1:1 ratio to receive neoadjuvant therapy followed by surgery or surgery alone. The primary endpoint was event-free survival (EFS), defined as the time from randomization to progression preventing surgery, postoperative recurrence, second primary cancer, or death. Secondary endpoints included overall survival (OS), objective response rate (ORR), R0 resection rate, pathological response, recurrence-free survival, and safety.
Study Design
Patients in the neoadjuvant group received the GOLP regimen before surgery:
- Toripalimab 240 mg intravenously every 3 weeks for 3 cycles
- Lenvatinib 8 mg orally once daily for 9 weeks
- Gemcitabine 1 g/m² intravenously on days 1 and 8
- Oxaliplatin 85 mg/m² intravenously on day 1
Chemotherapy cycles were repeated every 3 weeks for a total of three cycles. After completion of neoadjuvant therapy, tumor resectability was reassessed by a multidisciplinary team within 2–4 weeks. Curative resection with regional lymphadenectomy was then performed by experienced hepatobiliary surgeons.
Following surgery, patients in both treatment arms received adjuvant capecitabine (2500 mg/m² per day in two divided doses on days 1–14 of a 3-week cycle) for eight cycles.
Patients assigned to the control group underwent immediate curative resection followed by the same adjuvant capecitabine regimen.
Radiologic responses were evaluated according to RECIST version 1.1. Pathologic responses were assessed centrally by blinded independent review. Safety was evaluated according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0.
The study planned to enroll 178 patients, providing 80% power to detect a hazard ratio of approximately 0.57 for event-free survival.
Results
Between January 2021 and February 2025, 178 patients with resectable high-risk intrahepatic cholangiocarcinoma were enrolled and randomized:
- Neoadjuvant GOLP group: 88 patients
- Control group: 90 patients
Baseline characteristics were well balanced between the groups. The median age was 59 years. High-risk features included tumors larger than 5 cm in 60%, vascular invasion in 72%, multiple tumors in 17%, portal lymph-node metastasis in 15%, and elevated CA19-9 levels in 51% of patients.
In the neoadjuvant group, 99% of patients received at least one dose of therapy, and 94% completed all planned cycles. Surgery was performed in 97% of patients in the neoadjuvant group and 99% in the control group.
Event-Free Survival
At a median follow-up of 16.9 months, 95 events had occurred. Neoadjuvant therapy significantly improved event-free survival.
Median EFS:
- Neoadjuvant group: 18.0 months (95% CI 13.8–27.6)
- Control group: 8.7 months (95% CI 7.2–12.4)
The improvement was statistically significant (P < 0.001) and crossed the prespecified efficacy boundary.
Because the proportional hazards assumption was violated, restricted mean survival time analysis was performed. The restricted mean survival time was 23.1 months in the neoadjuvant group versus 16.6 months in the control group, corresponding to a 6.6-month difference.
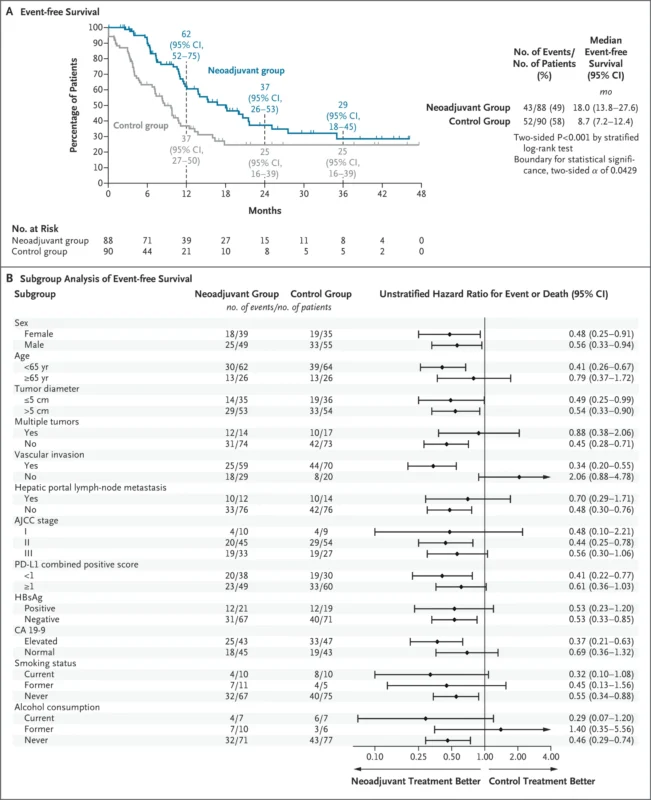
At 24 months, event-free survival rates were:
- 37% in the neoadjuvant group
- 25% in the control group
Overall Survival
At the interim analysis, 49 deaths had occurred:
- 18 deaths (20%) in the neoadjuvant group
- 31 deaths (34%) in the control group
Two-year overall survival rates were:
- 79% in the neoadjuvant group
- 61% in the control group
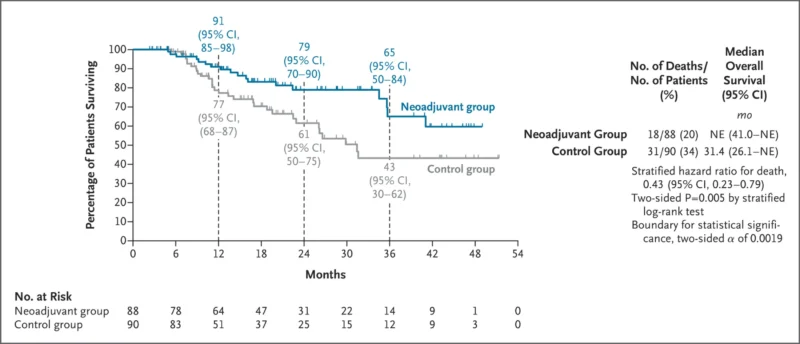
The hazard ratio for death was 0.43 (95% CI 0.23–0.79; P = 0.005). Although this suggested a survival benefit, the result did not reach the prespecified statistical significance threshold.
Additional Efficacy Outcomes
Neoadjuvant therapy also demonstrated meaningful tumor responses:
- Objective response rate: 55% (all partial responses)
- Major pathological response: 19%
- Pathologic complete response: 5%
The R0 resection rate was high in both groups:
- 95% in the neoadjuvant group
- 93% in the control group
Median recurrence-free survival was 15.4 months in the neoadjuvant group versus 9.7 months in the control group.
Key Findings
The ZSAB-neoGOLP trial demonstrated that neoadjuvant combination therapy integrating chemotherapy, targeted therapy, and immunotherapy can significantly improve outcomes in patients with high-risk resectable intrahepatic cholangiocarcinoma.
Neoadjuvant GOLP nearly doubled median event-free survival, increasing it from 8.7 months to 18.0 months. The regimen produced meaningful tumor shrinkage, with more than half of patients achieving radiologic responses and nearly one-fifth demonstrating major pathological response.
Importantly, neoadjuvant treatment did not compromise surgical feasibility. Resection rates remained high, and postoperative outcomes were similar between groups.
Although the interim overall survival analysis suggested a clinically meaningful reduction in mortality risk, longer follow-up will be required to determine whether the improvement translates into a statistically significant survival benefit.
Conclusion
The ZSAB-neoGOLP phase 2–3 randomized trial provides the first robust evidence supporting a neoadjuvant combination regimen for resectable high-risk intrahepatic cholangiocarcinoma. Neoadjuvant GOLP therapy significantly prolonged event-free survival compared with surgery alone and was associated with meaningful tumor regression and manageable toxicity.
Although overall survival results remain immature, the substantial improvement in event-free survival and favorable safety profile suggest that neoadjuvant systemic therapy may become an important strategy for managing high-risk ICC.
Further validation in international cohorts and longer follow-up will be essential to confirm the durability of the observed benefits and to determine whether this approach should be incorporated into future treatment guidelines.


