Cancer treatments are among the most groundbreaking and urgently needed medical advancements today. However, behind every breakthrough lies a complex, costly, and uncertain journey. The average cost of developing and bringing an oncology drug to market has soared in recent years.
In many cases, taking a new cancer medication from lab discovery to patient access now exceeds billions of dollars. Even more daunting is the fact that nearly 90% of drugs entering clinical trials ultimately fail to secure approval.
In this article , we look at each phase of the drug development and approval process – from preclinical research through post-marketing – detailing the timeline, costs, and challenges faced by companies while developing the drug. In this article, you will also find insights into the phases of clinical trials and their associated costs, as well as an overview of the EMA and FDA approval procedures that oncology drugs must navigate.
What is drug development and how does it work?
Drug development follows drug discovery, an exploratory phase in which researchers investigate various potential approaches to treating a medical condition. Once a promising compound is identified, scientists study its interactions with the human body, refine its formulation, and address any limitations to enhance its safety and effectiveness—this phase is called development. These stages lay the foundation for subsequent phases, including regulatory approval and the post-marketing phase for a specific drug.
Key Terms Explained
- Cancer drug – A cancer drug is a chemical-based medication used to prevent, treat, or manage cancer by targeting cancer cells or their environment. It works through various mechanisms, such as inhibiting the growth of abnormal cells, killing cancer cells, or boosting the immune system’s ability to fight cancer.
- Drug approval -This is the process by which regulatory agencies approve the use of a drug and confirm its effectiveness for public use.
- Preclinical Research – The stage of drug development before human testing, where scientists test a new drug in laboratory and animal studies.
- New Drug Application (NDA) / Biologics License Application (BLA) – Formal requests submitted to the FDA to approve a new drug (NDA for chemical drugs, BLA for biological treatments).
Phase 1 Clinical Trials
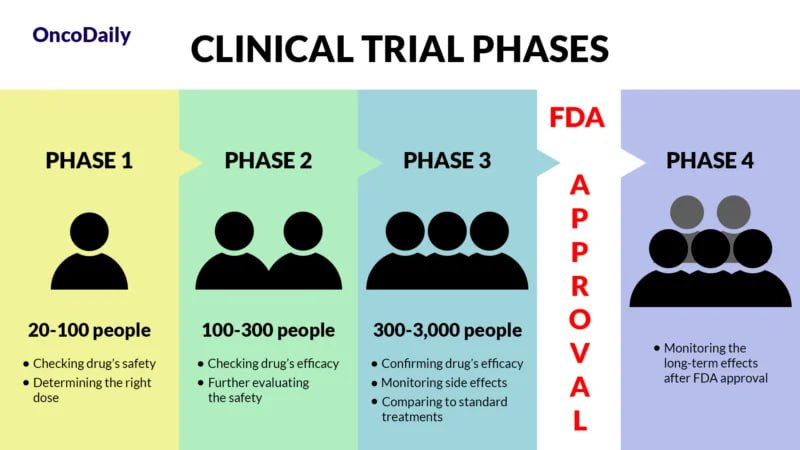
Phase 1 marks the first stage of testing a new cancer drug in humans, focusing primarily on dosage and potential side effects. Unlike other medical fields, where healthy volunteers often participate, oncology Phase 1 trials typically involve patients with advanced cancers who have exhausted standard treatment options.
In this phase, researchers prioritize assessing tumor response and addressing critical questions such as: What side effects does the drug cause? How should they be managed? How is the drug metabolized and eliminated? Many compounds are discontinued at this stage if human toxicity is found to be unacceptable. Nonetheless, relative to later phases, Phase 1 is smaller in scale. If the drug encounters serious safety issues here, the company might abandon the project early, cutting losses.
- Phase 1 oncology trials typically last from several months to 1–2 years.
- Average Phase 1 trial duration across all diseases is about 2.3 years.
- Oncology trials may progress faster with promising agents or expedited designations
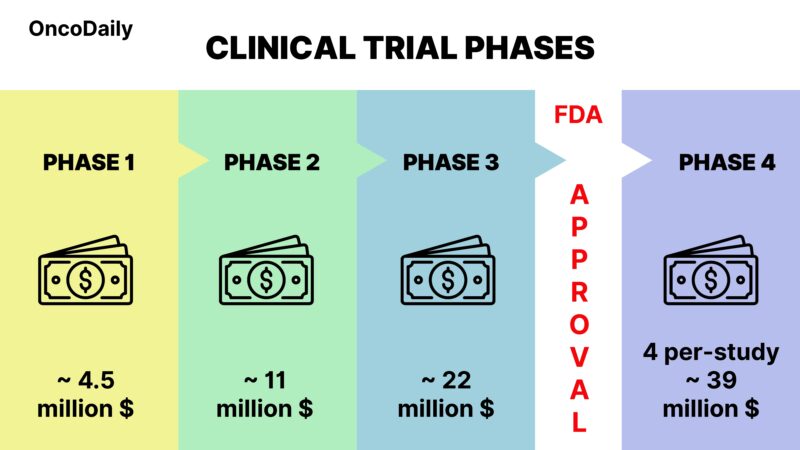
According to the U.S. Department of Health and Human Services (aspe.hhs.gov) Phase 1 trials are the least expensive clinical phase, but still a significant outlay. They often cost on the order of a few million dollars. For oncology, one study estimated the average per-trial cost of Phase 1 at about $4.5 million.
Nonetheless, if Phase 1 shows an acceptable safety profile (and perhaps a hint of efficacy), it’s a green light to proceed to the next stage. According to BIO’s ‘Clinical Development Success Rates and Contributing Factors 2011–2020’ study.
“Success for the 2016 for Phase 1 to Phase 2 was 62.8% ,meanwhile now it is 40.9%”

Phase 2 Clinical Trials
Phase 2 is where the experimental drug is put to an initial test of effectiveness. These trials enroll more patients and often focus on a specific cancer type or subset of patients. The objective of Phase 2 is to provide proof that the drug works in its intended population. Phase 2 helps researchers determine whether the drug’s benefits justify proceeding to the much larger (and more expensive) Phase 3 trials. It’s essentially a “gatekeeper” phase where many drugs fail if they don’t demonstrate enough efficacy.
For instance, if a new chemotherapy shows a tumor response in only 5% of patients, while 30% was expected, it is unlikely to progress further. However, a strong outcome in Phase 2 is often sufficient to warrant continued examination and development of the drug.
According to the College of Medicine at the University of Cincinnati, Phase II studies determine the effectiveness of an experimental drug on a particular disease or condition in approximately 100 to 300 volunteers. This phase may last from several months to two years.
The timeline depends on how difficult it is to recruit patients (for rare cancers, enrollment can be slow), and how long patients are treated and followed. Because Phase 2 involves a larger patient group, more extensive data collection, and often multiple participating hospitals or cancer centers, it is more expensive than Phase 1.
Derep Maxime 2022 study ‘What’s the average time to bring a drug to market?’
- A typical Phase 2 trial runs about 1.5 to 3 years.
- Industry data put the average Phase II duration at roughly 3.6 years
According to the U.S. Department of Health and Human Services (aspe.hhs.gov), these trials typically cost in the low tens of millions of dollars, with the average cost for an oncology Phase 2 trial around $11 million. According to BIO’s ‘Clinical Development Success Rates and Contributing Factors 2011–2020’ study.
“Success for the 2016 for Phase 2 to Phase 3 was the same 24.6%”
Phase 3 Clinical Trials
The trials in Phase 3 can enroll anywhere from a few hundred to several thousand patients, often across multiple countries. These trials usually compare the new drug against the current standard of care or a placebo (if no standard treatment exists) .For oncology drugs, Phase 3 trials typically look at endpoints like overall survival, disease-free survival, or sustained tumor response, providing the robust evidence needed for regulators to evaluate the durg’s risk-benefit profile. According to BIO’s ‘Clinical Development Success Rates and Contributing Factors 2011–2020’ study.
“Success for the 2016 for Phase 3 to NDA and BLA was 40.1%,meanwhile now it is 47.7%”
Derep Maxime 11/05/2022 study ‘What’s the average time to bring a drug to market?’
- An analysis of drug development timelines found Phase III averages about 3.3 years
In rare cases where interim results are overwhelmingly positive (or negative), trials can sometimes be stopped early. For example, an independent monitoring board might halt a trial if the new drug clearly improves survival, to allow all patients to access it sooner. Conversely, if interim data show the drug isn’t working or has safety issues, the trial will be terminated early to avoid harming patients.
The interesting part is that even after thorough examination of the drug its approval is still not assured. Many drugs fail in Phase 3 because they do not confirm the hoped-for benefits seen in Phase 2. In oncology, the success rate of drugs getting through Phase 3 to approval is relatively low compared to other fields. According to BIO’s ‘Clinical Development Success Rates and Contributing Factors 2011–2020’ study.
“About 95% of oncology drug projects never achieve FDA approval, underscoring how high the bar is for safety and efficacy”.
Phase 3 trials can cost anywhere from tens to hundreds of millions of dollars each. According to the U.S. Department of Health and Human Services (aspe.hhs.gov) for oncology, a study on clinical trials costs around $22 million. However, this number can vary significantly. For instance, two pivotal trials for different cancer indications can double or even triple the expenses. Common cancers, such as lung or breast cancer, can easily cost over $50–100 million when factoring in thousands of patients, lengthy follow-ups, expensive imaging, and, in some cases, companion diagnostic tests.
Regulatory Approval – FDA VS EMA
The logical continuation of Phase 3 trials is obtaining regulatory approval to market the drug. The specific application process varies by country. In the United States, companies must submit a New Drug Application (NDA) for traditional drugs or a Biologics License Application (BLA) for biologic products to the Food and Drug Administration (FDA). Similarly, in the European Union, a Marketing Authorization Application (MAA) is submitted to the European Medicines Agency (EMA). Additionally, companies must comply with the regulatory requirements of other agencies worldwide.
FDA (United States) Process
In USA Food and Drug Administration Center for Drug Evaluation and Research (CDER) will review a New Drug Application (NDA) for traditional drugs or a Biologics License Application (BLA) for biologic products. It is also important to mention that FDA has target timelines for review
- Typically 10 months for a standard review or 6 months for a priority review.
- From NDA submission to FDA decision, the timeline is usually around 6 to 12 months (faster for breakthrough therapies or those with expedited review), according to Lythgoe et al study published in JAMA Network Open in 2022.
For example, drugs on the FDA’s priority review have shorter clocks: one study noted FDA’s accelerated approval pathway reduced median review time by about 31 days compared to standard
EMA (European Union) Process
For all the EU member states approval is sought through the EMA’s centralized procedure. The company submits a Marketing Authorization Application, and it is evaluated by the Committee for Medicinal Products for Human Use (CHMP). The EMA review timeline is roughly 12 to 15 months for a standard review.
- The formal clock is 210 days of active evaluation, but “clock stops” for the company to answer questions often extend the total time.
FDA approval allows marketing in the U.S. only, EMA approval covers all EU countries, and other nations have their own licenses – so a global launch requires navigating all these processes. That said, FDA and EMA decisions are highly influential; a positive FDA approval often paves the way for faster acceptance in other countries. Once approvals are secured, the work isn’t over – it enters a new phase of post-marketing surveillance and expansion. There have even been instances where the FDA approved an oncology drug that the EMA initially rejected or restricted, reflecting different risk-benefit judgments
Costs of Regulatory Submission
Seeking regulatory approval also entails significant costs. Simply preparing the massive submission (thousands of pages of technical documents) can cost a company millions in labor and consulting.
- In the EU, the EMA charges fees for the MAA review; historically it has been on the order of €138,700 (e.g. informed consent [Art 10c Dir 2001/83/EC]) to €357,600 (new active substance [Art 8 (3) Dir 2001/83/EC]) for a new medicine application
- In the U.S., the FDA’s user fee for filing a new drug application requiring clinical data is about $4.0 million in FY 2024
During the regulatory phase, companies often begin ramping up marketing and commercialization plans in anticipation of approval. However, they must be careful – until approval is granted, a drug cannot be promoted. It’s a tense period: the company has no guarantee of approval, yet has invested perhaps a decade and a billion dollars by this point.
Phase 4 Clinical Trials
This phase is so-called post-marketing studies, which refers to research conducted after a drug has been approved and is on the market. At this stage, the drug is available to the broader patient population, but both regulators and the company continue to monitor its performance and safety. The primary goal in Phase 4 is to ensure long-term safety and effectiveness of the drug in the general population. With a much larger and more diverse group of patients using the medication, rare or long-term side effects that weren’t apparent in the smaller trials may emerge.
Regulators like FDA often require post-marketing surveillance programs or registries for certain drugs. A case in point: some immunotherapy drugs got early approval for metastatic cancer based on tumor response rates, but then had to prove in Phase 4 that they actually extended survival. Using the example of durvalumab, which was approved through an accelerated program, we can see that if a drug fails to demonstrate its expected benefits, its approval may be revoked by the phase 4.
- Conducting additional trials can be expensive – sometimes tens of millions of dollars for large outcome studies. An analysis of post-approval (Phase 4) trial costs showed that oncology drugs had an average Phase 4 per-study cost of about $39 million
Phase 4 is effectively open-ended – it continues as long as the drug is on the market. Companies will keep researching their product to stay ahead of competition (for instance, developing improved formulations like an oral pill instead of IV infusion, or finding biomarkers that predict response to personalized treatment). They also must stay vigilant for any late-arising safety issues. In the best case, Phase 4 studies continue to show that the drug is benefiting patients, perhaps in new ways not initially envisioned, and any risks remain manageable. This ongoing phase ensures that the drug’s life cycle is managed responsibly while extending its reach to help as many patients as possible.
The drug development and approval process for cancer treatments is a lengthy, costly, and highly complex journey. As patients and advocates, it’s important to remain informed about the stages of drug development and the challenges faced by pharmaceutical companies.
While the approval process is rigorous to ensure safety and efficacy, patients should also stay updated on clinical trial opportunities and advancements in treatments. Engaging with healthcare providers about new therapies and available trials can be essential for gaining access to the most promising innovations. Ultimately, while the road to approval is difficult, the progress made continues to offer hope for better cancer treatments in the future.
Read more about the most promising cancer drugs of 2024 in OncoDaily’s insight – “10 Most Promising Cancer Drugs in 2024. What Patients Need to Know”
Written by Zhanna Chakhalyan
FAQ
How long does it typically take to develop a new drug?
Developing a new drug typically takes an average of 10 to 15 years, depending on various factors such as the type of drug and regulatory processes.
What is the average cost of developing a new drug?
The average cost of developing a new drug is estimated to be around $1 billion to $2.6 billion, with significant variability across different therapeutic areas.
What are the main phases of drug development?
The main phases include discovery and development, preclinical research, clinical trials (Phase I, II, III), FDA review, and post-marketing surveillance.
What is the purpose of preclinical trials?
Preclinical trials aim to assess the safety and efficacy of drug candidates through laboratory and animal testing before human trials begin.
What percentage of drugs entering clinical trials are eventually approved?
Only about 12% of drugs entering clinical trials are eventually approved by regulatory bodies like the FDA.
Why do many drug candidates fail during clinical trials?
Many drug candidates fail due to issues with efficacy, safety, or lack of therapeutic effect in humans, despite promising results in preclinical stages.
How do advancements in technology impact drug development?
Technologies like AI and machine learning can significantly reduce the time and cost of drug development by enhancing data analysis and candidate identification.
What are the different types of drugs in terms of innovation and cost?
Drugs can be categorized as new concept drugs, precedent drugs, generics, and biosimilars, each with varying levels of innovation and cost.
What role does the FDA play in drug development?
The FDA is responsible for reviewing and approving drugs for safety and efficacy before they can be marketed in the U.S., and it also oversees post-marketing surveillance.

