Minhua Chu, Managing Partner at Transition Value Partner, shared a post on X:
“ESMO Breast Cancer 2026.
Duality Bio presented a Fast Oral, reporting first‑line data for TROP2 ADC Sacituzumab, Drozuntecan (DB‑1305/BNT325) + PD‑L1×VEGF‑A bispecific pumitamig (BNT327/BMS986545) in a/mTNBC.
Study design:
- Phase I/IIa (NCT05438329), dose escalation + expansion
- Expansion cohort: untreated unresectable/advanced/metastatic TNBC
- Regimen: pumitamig 30 mg/kg Q3W + DB‑1305/BNT325 3.5 mg/kg Q3W
- Primary endpoints: ORR, safety
- Median age 55.5; 83.3% Chinese patients
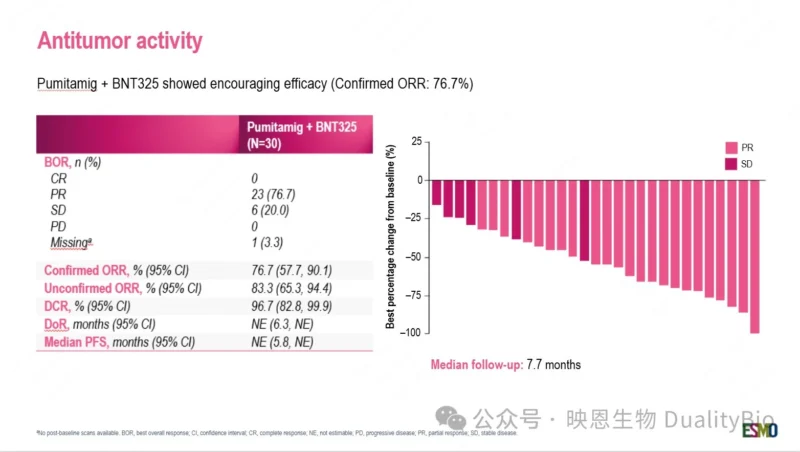
Key results (n=30):
- uORR 83.3%, cORR 76.7%
- DCR 96.7%
- Tumor shrinkage in all evaluable patients
- 95.2% maintained response at 6 months
Safety: Manageable profile.
- Grade 3–4 TRAEs: 43.3%
- Discontinuation due to TRAEs: 3.3%
- Common TRAEs (>40%): mucositis, weight loss, ALT elevated, alopecia (mostly Grade 1–2).”
Other articles featuring Minhua Chu on OncoDaily.


