Loay Kassem, Assistant Professor of Clinical Oncology at Kasr Al-Ainy School of Medicine, Cairo University, shared a post on LinkedIn:
“This is one of the biggest misleading interpretation of a clinical trial in breast cancer history.
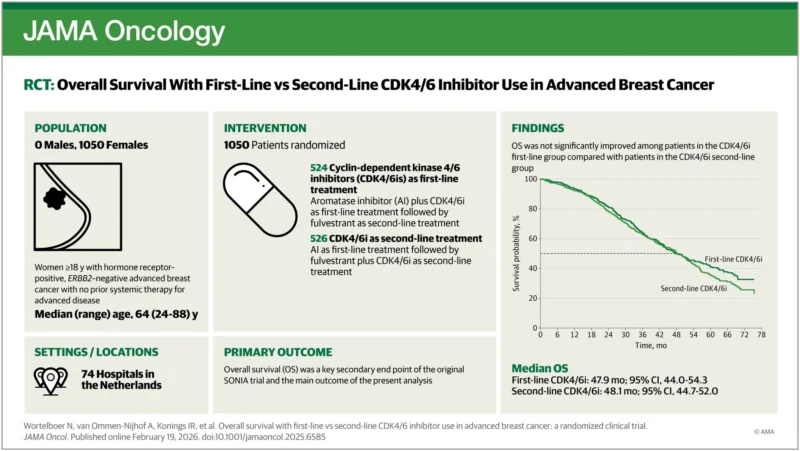
The SONIA study, that randomized patients to first line CDK4/6 inhibitor versus deferring its use to second line setting, showed that there is no difference between the 2 strategies regarding PFS2 and overall-survival.
This is the main conclusion of the final analysis published in JAMA Oncology few days ago. And consequently, many patients with metastatic breast cancer will be offered to delay CDK4/6 inhibitor therapy beyond first line.
I totally disagree with this interpretation for many reasons:
- In the same study, the premenopausal subgroup showed 50% reduction in the risk of death if started first line therapy. Such observation was statistically significant. To me, offering the delay strategy to young patients is a risky under-treatment.
- The SONIA study mostly used palbociclib which didn’t show OS benefit when used in the earlier lines (unlike ribociclib and abemaciclib). In the same line, both adjuvant abemaciclib and ribociclib (in the early setting) were positive while palbociclib was negative.
- Those patients were kept under a very close follow-up of the clinical trial setting. Unfortunately, real-world patients are not followed that close and might not be fit to switch to second line CDK4/6 inhibitors upon progression.
I would like to hear your opinion on this. Feel free to share in the comments.
N.B. I put the premenopausal versus postmenopausal survival curves in the comments to see the difference.”
Other articles about CDK4/6 on OncoDaily.


