BOT/BAL Advancements, BATTMAN Trial, FDA Approval Challenges and Key Highlights from Agenus Stakeholder Briefing
On August 27, 2025, Agenus Inc. hosted a pivotal virtual Stakeholder Briefing to unveil critical developments surrounding its immuno-oncology pipeline, particularly focusing on botensilimab (BOT) and balstilimab (BAL), the BATTMAN trial. The session featured expert presentations from senior management, including Garo H. Armen, PhD, and Jennifer Buell, PhD, alongside thought leaders in oncology such as Dr. Richard M. Goldberg and Dr. Nicholas C. DeVito. These experts discussed the transformative potential of BOT and BAL, groundbreaking clinical trial results, and the need for regulatory evolution to address the accelerating pace of scientific advancements.
The briefing outlined the robust progress of the BATTMAN Phase 3 trial, exploring the unfulfilled needs in colorectal cancer (CRC), and emphasized the importance of accelerating patient access to promising new therapies. This article offers a comprehensive look at the key takeaways, including the latest data from BOT and BAL trials, the urgency for expanded patient access, and reflections on FDA regulations and the pathway to accelerated drug approvals.
What Drugs are Botensilimab and Balstilimab?
Botensilimab is a human Fc-enhanced CTLA-4 blocking antibody designed to unleash powerful anti-tumor immune responses by targeting cold tumors—those that typically do not respond to traditional therapies. By modulating both innate and adaptive immunity, BOT aims to transform the treatment landscape for cancers that are resistant to current standard-of-care treatments.
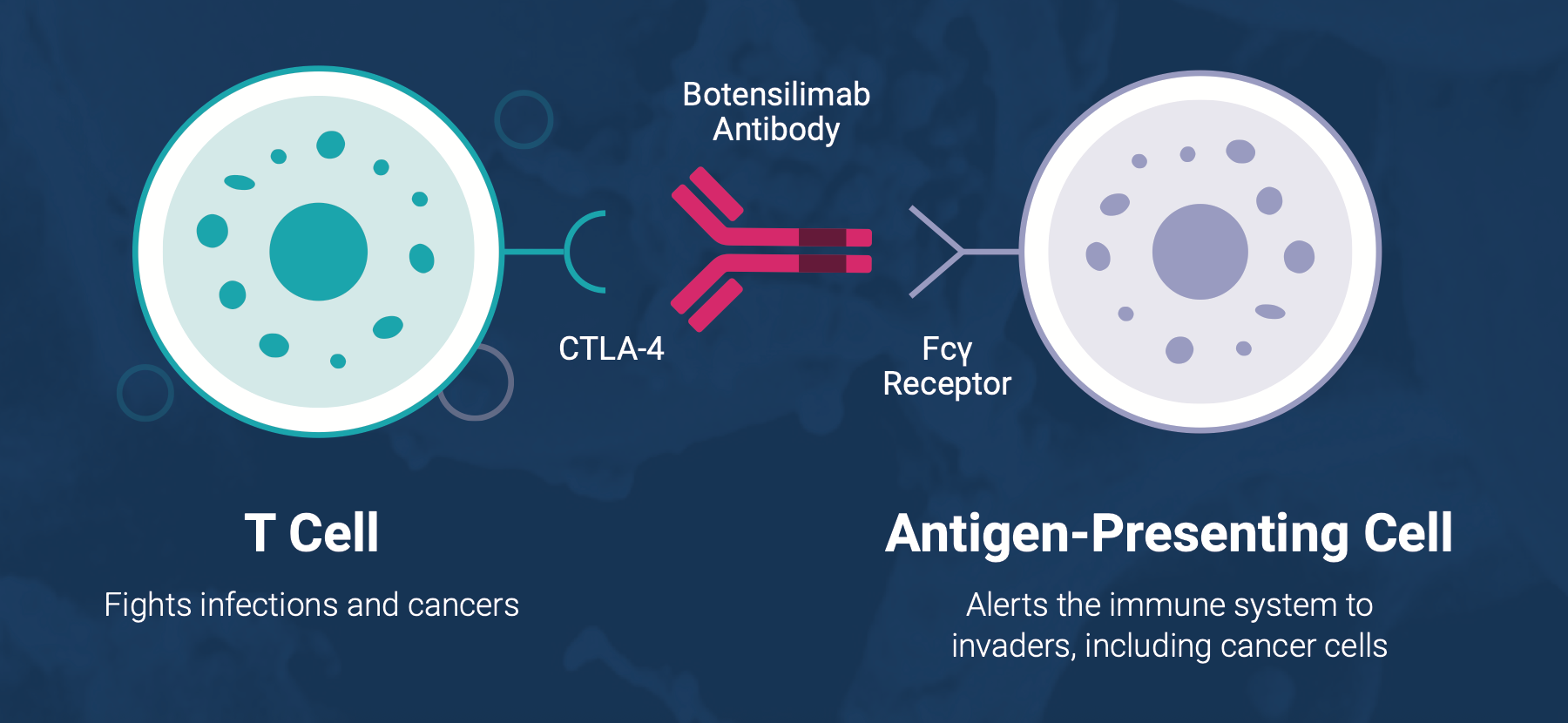
As of the latest clinical trials, approximately 1,200 patients have participated in BOT studies across multiple tumor types, with a focus on late-stage metastatic cancers. BOT has shown promise in conjunction with Agenus’ PD-1 antibody, balstilimab (BAL), in driving meaningful immune responses. Clinical data have demonstrated strong efficacy in nine metastatic cancers, including colorectal, melanoma, and non-small cell lung cancer.
Balstilimab is a fully human monoclonal IgG4 antibody that blocks PD-1 (programmed cell death protein 1), a checkpoint protein that tumors exploit to avoid immune system attack. By inhibiting PD-1, BAL helps restore the immune system’s ability to target and destroy cancer cells. With over 900 patients treated across several cancer types, BAL has shown a favorable safety profile and clinical efficacy, providing significant therapeutic benefits for patients with high unmet needs.
Latest Data and Trial Results
The briefing highlighted recent data from both Phase 1 and Phase 2 trials of BOT and BAL, particularly in colorectal cancer (CRC), where these therapies have shown groundbreaking results. In a Phase 1 study involving late-stage microsatellite stable (MSS) colorectal cancer patients—who typically respond poorly to immunotherapies—BOT demonstrated a 42% two-year survival rate. These results are unprecedented when compared to the 10 to 14 months survival seen with traditional chemotherapy regimens.
“These patients had either exhausted all approved treatment options or were left with no therapeutic options. Despite this, we saw a remarkable median overall survival of 21 months, far outstripping the current best standard of care.”
Dr. Richard Goldberg, CDO of Agenus.
Moreover, in early-stage trials, preliminary data from studies like NEST, Unicorn, and Neoasis suggest even more dramatic responses when BOT and BAL are administered before chemotherapy, a strategy known as neoadjuvant therapy. These results underline the potential for combining BOT/BAL to not only improve survival but also reduce the need for harsh chemotherapy regimens.
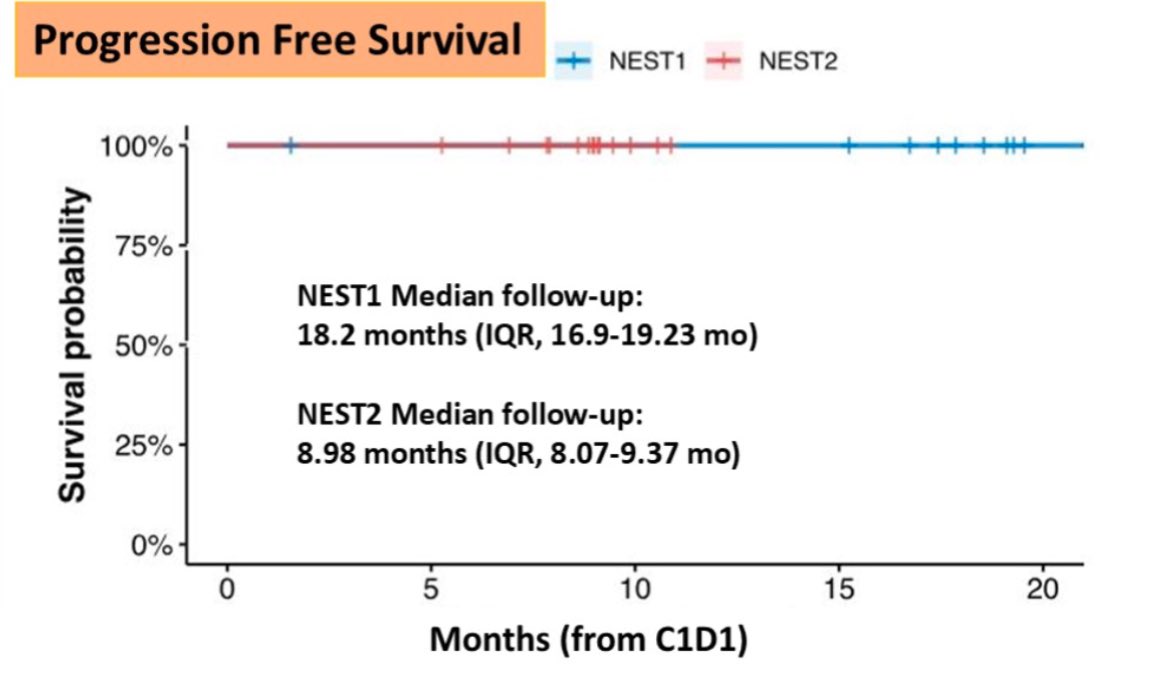
In the NEOASIS trial, BOT/BAL showed a 70% pathological complete response (pCR) in MSI-H tumors and 20% in MSS tumors, demonstrating efficacy in traditionally hard-to-treat cancers. The NEST-1 and NEST-2 trials, evaluating neoadjuvant therapy in colorectal cancer, reported 100% ctDNA-negative status and 47% major pathological response in MSS tumors, suggesting a shift towards curative strategies.
The UNICORN trial showed 93% pCR in dMMR tumors and 29% in pMMR tumors with BOT/BAL treatment, underlining the combination’s efficacy in early-stage CRC. In Hepatocellular Carcinoma (HCC), BOT/BAL achieved a 17% overall response rate and 72% disease control rate, with a 12.3-month median overall survival, demonstrating promise for liver cancer patients previously unresponsive to first-line therapies.
Regulatory Landscape and the Need for Accelerated Approval
One of the most pressing topics addressed during the briefing was the evolving regulatory environment and the need to expedite approval processes for therapies like BOT and BAL. Despite the promising data, regulatory bodies such as the FDA continue to mandate large, randomized Phase 3 trials before approval, often delaying patient access to potentially life-saving treatments.
Dr. Goldberg emphasized the urgent need for reform:
“Patients are waiting, and every day that passes, lives are lost. The science is moving faster than the regulatory framework allows. It’s time for the FDA and other regulatory agencies to adapt and reflect the rapid advancements in immuno-oncology.”
The conversation highlighted the challenges of navigating bureaucratic hurdles while patients with aggressive cancers desperately await access to innovative therapies. Stakeholders from Agenus and its academic collaborators called for a more flexible approach to approvals, advocating for conditional approvals based on early-stage clinical signals and real-world data to accelerate access for patients in urgent need.
Dr. Chris O’Callaghan, a senior investigator at the Canadian Cancer Trials Group, reflected on the urgency of expanding access to these therapies.
“We know that if we can introduce these immunotherapies earlier in the treatment regimen, the potential benefits are enormous. Our goal is to ensure that patients who need it most can access these drugs as quickly as possible.”
The BATTMAN Trial: A Phase III Study of Botensilimab and Balstilimab in Metastatic MSS CRC
One of the key developments discussed during the briefing was the launch of the BATTMAN Phase 3 study focusing on the efficacy of BOT and BAL in metastatic CRC, particularly for the MSS subgroup, which has historically been resistant to immunotherapy. The trial design, approved by the FDA, eliminates the need for a separate BOT monotherapy control arm, streamlining the path to approval. The trial is designed as a randomized study to compare the combination of BOT/BAL against BOC in a heavily pretreated population with advanced MSS CRC who have exhausted all other treatment options. Agenus aims to expand enrollment through strategic collaborations and is focused on accelerating patient access to this promising combination therapy, with the trial set to launch in Q4 2025.
Dr. Nicholas DeVito, an assistant professor of medical oncology at Duke University, spoke about the challenges facing CRC patients:
“The rise in colorectal cancer in younger patients is alarming, and the need for new therapies is clear. BOT and BAL offer hope where few options exist.”
The enthusiasm for these trials is palpable. Dr. DeVito shared an inspiring case of a 30-year-old patient who, after enrolling in a first-line BOT/BAL trial, showed significant improvement with minimal side effects, demonstrating the potential of these therapies to change the treatment paradigm for CRC.
MiNK Therapeutics and INKT Cells
In addition to BOT and BAL, MiNK Therapeutics, a subsidiary of Agenus Inc, has been focusing on the development of Invariant Natural Killer T (INKT) cells for immuno-oncology. INKT cells are a subset of immune cells that bridge both innate and adaptive immunity, and they are being investigated for their ability to treat cancers and potentially infectious diseases.
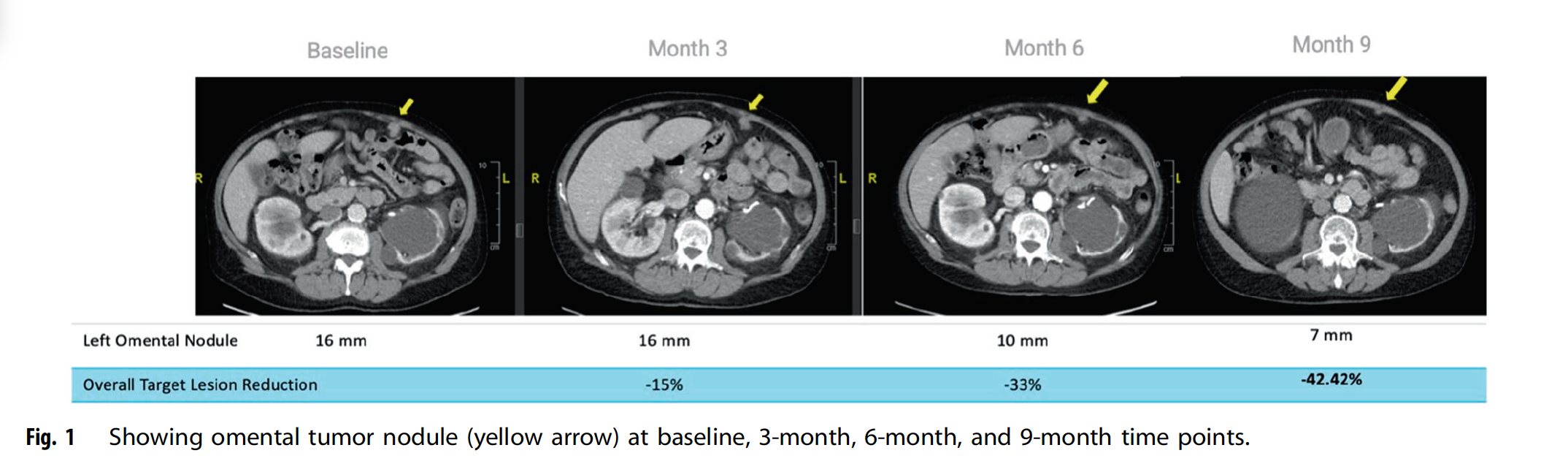
Figure of metastatic germ cell tumor patient treated with iNKT, Report Published on Nature’s Oncogene, 2025: Read Summary Here
Dr. Jennifer Buell, CEO of MiNK Therapeutics, discussed the potential of INKT cells, saying,
“INKT cells are unique in their ability to respond rapidly and modulate the immune system’s adaptive response. Their potential goes beyond cancer, as they may also offer therapeutic benefits in treating infectious diseases.”
MiNK Therapeutics is working on advancing the clinical use of INKT cells, both independently and in combination with other immunotherapies, to address unmet needs in cancer treatment and beyond.
Moving Towards a New Era in Cancer Treatment
Agenus’ Stakeholder Briefing on August 27, 2025, highlighted the immense potential of botensilimab and balstilimab to revolutionize cancer treatment. With promising clinical results, particularly in colorectal cancer, these therapies offer hope for patients who have exhausted traditional options. However, the road to broader patient access remains challenging, with regulatory bottlenecks standing in the way of expedited approvals.
“Patients want more than just survival; they want quality of life. Chemotherapy often comes with debilitating side effects like neuropathy and cytopenias. BOT and BAL offer a path forward, with manageable side effects and the promise of longer, better lives.”
As the immuno-oncology field continues to advance at an unprecedented pace, it is crucial that regulatory agencies adapt to keep up with the science. The need for accelerated drug approvals has never been more urgent, and it is clear that therapies like BOT and BAL have the potential to significantly improve outcomes for patients in desperate need of new treatment options.
As concluded by Garo Armen, PhD, Founder, Chairman, and CEO of Agenus.
“We believe that the regulators need to reflect upon and update the approval process. Science is moving fast, and regulatory processes need to move at the same pace as the science. We are committed to advancing immuno-oncology and accelerating access to therapies that can save lives today.”

Read More Immune-Oncology Updates on OncoDaily IO
-
Challenging the Status Quo in Colorectal Cancer 2024
December 6-8, 2024
-
ESMO 2024 Congress
September 13-17, 2024
-
ASCO Annual Meeting
May 30 - June 4, 2024
-
Yvonne Award 2024
May 31, 2024
-
OncoThon 2024, Online
Feb. 15, 2024
-
Global Summit on War & Cancer 2023, Online
Dec. 14-16, 2023