
Zanidatamab (Ziihera): Patient Guide for 2025
Zanidatamab, also known by its brand name Ziihera, is a new type of cancer medicine designed to treat cancers that have a specific protein called HER2. It is a targeted therapy, meaning it works by focusing on cancer cells that have this HER2 protein, helping to stop their growth. Zanidatamab has been approved by the FDA in November 2024 to treat a rare and serious cancer called biliary tract cancer (cancer of the bile ducts) when it is advanced or cannot be removed by surgery. Doctors are also studying its use in other types of HER2-positive cancers, like breast, stomach, and lung cancers.
This article will help you understand what Zanidatamab is, how it works, what cancers it treats, what to expect during treatment, possible side effects, and ongoing research. The goal is to empower you with the information you need to discuss treatment options with your healthcare team.
What Is Zanidatamab and How Does It Work?
Zanidatamab is a type of medicine called a bispecific antibody. To explain that simply: antibodies are proteins that your immune system makes to fight infections. Cancer medicines like Zanidatamab are designed in the lab to work like these antibodies, but to specifically find and attack cancer cells.
What makes Zanidatamab special is that it can bind to two different parts of the HER2 protein at the same time. Think of it like a double-sided Velcro that sticks more strongly to cancer cells with HER2. This helps the medicine block signals that cancer cells use to grow and survive.
By attaching to HER2 on the cancer cells, Zanidatamab helps the immune system recognize and destroy these cancer cells. It also helps pull the HER2 proteins inside the cancer cells, stopping them from sending growth signals. This dual action makes Zanidatamab effective even in cancers that might have stopped responding to older HER2 treatments.
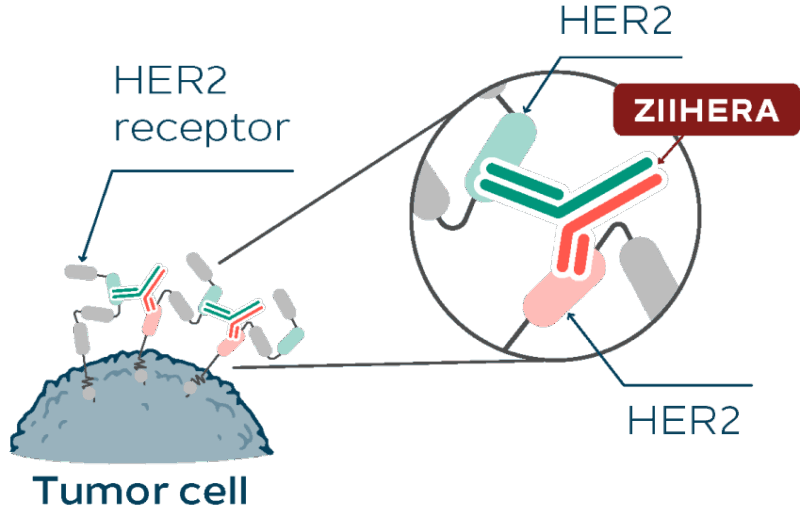
Source: Ziihera® Official Website
What Cancers Does Zanidatamab Treat?
Zanidatamab is currently approved for adults with HER2-positive biliary tract cancer that is advanced or cannot be removed by surgery and has already been treated with other medicines. This type of cancer starts in the bile ducts, which are tubes that carry bile from the liver to the intestines. The FDA approval is based on how well Zanidatamab works in patients whose tumors have high levels of HER2 protein, usually tested by a lab technique called immunohistochemistry (IHC 3+).
Doctors are also testing Zanidatamab in other cancers that have HER2 protein, including:
- Breast cancer
- Stomach (gastroesophageal) cancer
- Lung cancer
- Pancreatic cancer
- Colorectal cancer
- Ovarian cancer
This is because many cancers can have HER2 protein involved in their growth, and Zanidatamab may help control these tumors.
What Is a Clinical Trial and Why Does It Matter?
A clinical trial is a research study designed to test new drugs and treatments in patients to determine their safety and effectiveness. Before Zanidatamab was approved, it went through multiple phases of clinical trials to assess how well it worked, what side effects it caused, and whether it was better than existing treatments. Clinical trials are essential because they provide scientific evidence that a drug can help patients while ensuring it is safe for widespread use.
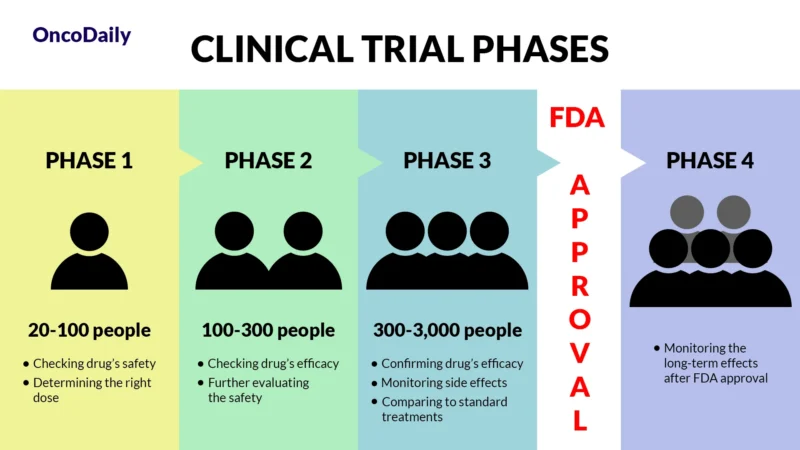
What Does FDA Approval Mean?
When a drug receives FDA approval, it means that after rigorous testing in clinical trials, it has been shown to be both safe and effective for treating a specific condition. This approval makes the drug widely available for doctors to prescribe and helps patients access new, cutting-edge treatments sooner.
Efficacy and Results from Clinical Trials
The approval of Zanidatamab for biliary tract cancer came from a study called HERIZON-BTC-01, which was published in The Lancet Oncology in 2023. In this study, 87 patients with HER2-positive biliary tract cancer who had already tried other treatments received Zanidatamab every two weeks through an IV infusion.
The results were encouraging. About 41% of patients experienced tumor shrinkage, which is called an objective response. For many, this response lasted for more than a year (about 13 months). Additionally, almost 69% of patients saw their disease controlled, meaning their cancer stopped growing or shrank. On average, patients lived about 12 months after starting Zanidatamab.
Zanidatamab was also studied in combination with chemotherapy for patients with advanced stomach or gastroesophageal cancers. A four-year follow-up study published in the Journal of Clinical Oncology in 2025 showed that patients lived a median of 36.5 months, which means many lived for more than three years after treatment started. The combination treatment also had a manageable safety profile. These trials show that Zanidatamab can offer meaningful benefits, especially in cancers where HER2 plays an important role.
What Can You Expect During Treatment?
Zanidatamab is given through an intravenous (IV) infusion, which means the medicine is slowly delivered into a vein, usually in your arm. Treatments are typically scheduled every two weeks. Each infusion can last from about 1 to 2 hours depending on how you tolerate the medicine.
Before each infusion, you will receive medications to reduce the chance of side effects. These may include acetaminophen (Tylenol), an antihistamine like diphenhydramine, and sometimes a corticosteroid. These help prevent infusion reactions, such as fever or chills. Your healthcare team will monitor you during and after the infusion to watch for any side effects or reactions. You may need blood tests and heart function checks throughout treatment.

Learn more about Cholangiocarcinoma: Latest Risk Factors, Symptoms, Diagnosis, and 2025 Advanced Treatment Options for Bile Duct Cancer on OncoDaily.
Zanidatamab Side Effects and Management
Zanidatamab may cause side effects, like all cancer treatments. This happens because while the medicine is designed to attack cancer cells, it can sometimes affect healthy cells too. Knowing what to expect and how side effects are managed can help patients feel more prepared and supported during treatment.
Common side effects
The most common side effects include changes in blood counts, mild liver irritation, stomach or digestive issues, and infusion-related reactions. Some patients may develop anemia, which can cause fatigue or weakness. Others may notice higher liver enzyme levels during blood tests, which usually means the liver is under stress but not necessarily damaged. Digestive problems such as diarrhea, nausea, vomiting, loss of appetite, or stomach pain are fairly common and may vary in intensity. Reactions during or shortly after the infusion can also happen, leading to symptoms like fever, chills, flushing, or shortness of breath. Fatigue and skin rash are also reported by many patients.
Less common side effects
Less commonly, zanidatamab can affect the heart by lowering its pumping ability, which is why regular heart monitoring is recommended throughout treatment. In rare cases, inflammation of the lungs (called pneumonitis) may occur and requires stopping the drug. Some patients may also develop electrolyte imbalances, such as low sodium or potassium levels, which are checked regularly with blood tests.
How Side Effects Are Managed?
To help prevent or reduce infusion-related reactions, patients are usually given medications such as acetaminophen, antihistamines, and corticosteroids before each dose. If a reaction happens, the infusion may be slowed down, paused, or sometimes stopped altogether, depending on how severe it is. Digestive side effects like diarrhea are usually managed with supportive care, but if the diarrhea is severe or long-lasting, treatment may be temporarily stopped or adjusted.
Because zanidatamab can sometimes affect the heart, patients undergo heart function tests before and during treatment. If the heart shows signs of weakness, the drug may be paused until recovery or permanently stopped if the problem is more serious.
Overall, most side effects are manageable with close monitoring and supportive care. The care team will regularly check blood counts, liver function, electrolytes, and heart function to make sure treatment remains as safe and effective as possible. Patients should always report new or worsening symptoms right away so adjustments can be made promptly.

What to Expect Long-Term?
Zanidatamab is not considered a cure, but it can help control cancer growth and improve survival for many patients. Clinical trials have shown that some patients respond to Zanidatamab for many months or even years, helping to manage their disease and maintain quality of life. If your cancer progresses during treatment, your doctor will discuss next steps, which might include switching therapies or supportive care options. Ongoing research aims to improve how long Zanidatamab works and who benefits most.
What Should You Avoid During Treatment?
While on Zanidatamab, it is important to avoid starting any new medications, including over-the-counter drugs and herbal supplements, without checking with your doctor. Some medications can interfere with treatment or increase side effects. Alcohol should be limited or avoided because it can worsen side effects like nausea, fatigue, and liver irritation.
Vaccinations, especially live vaccines (such as certain flu vaccines), should generally be avoided unless your healthcare provider says otherwise, as Zanidatamab can affect your immune system. If you feel dizzy or unwell during or after infusions, avoid driving or operating heavy machinery until you feel better. It is important not to miss doses or delay treatment without discussing it with your care team, as this can reduce the effectiveness of Zanidatamab.
Real-Life Effectiveness
Real-world use of Zanidatamab is still being studied, but early data from clinical trials and patient experiences show promising results in controlling HER2-positive cancers, especially biliary tract and gastroesophageal cancers. Patients often report improvements in symptoms and quality of life alongside tumor control. Healthcare providers continue to gather information to better understand how Zanidatamab works outside of clinical trials, helping to refine treatment guidelines and improve patient care.
Looking Ahead – The Future of Treatment
Research on Zanidatamab continues to expand. Scientists are exploring how it might be combined with other cancer treatments to boost effectiveness. New studies are testing Zanidatamab in different types of HER2-positive cancers and in earlier stages of disease. Advances in understanding how cancer cells grow and resist treatment will help doctors tailor therapies like Zanidatamab to the right patients, potentially improving outcomes even more. As more data becomes available, Zanidatamab could become part of standard care for a wider range of patients with HER2-positive cancers.
What Other Trials Are Ongoing?
Research continues to explore Zanidatamab in many other cancers and settings. One major ongoing study is called DiscovHER PAN-206, which looks at Zanidatamab in patients with a variety of HER2-positive solid tumors who have had previous treatments. This includes breast, lung, ovarian, pancreatic, colorectal, and gastric cancers. Researchers are also studying Zanidatamab in combination with other therapies like immunotherapy, which helps the immune system fight cancer more effectively. These trials aim to find new ways to improve outcomes and expand the types of cancer patients who can benefit.
If you’re a healthcare provider, access the professional version here.
-
Challenging the Status Quo in Colorectal Cancer 2024
December 6-8, 2024
-
ESMO 2024 Congress
September 13-17, 2024
-
ASCO Annual Meeting
May 30 - June 4, 2024
-
Yvonne Award 2024
May 31, 2024
-
OncoThon 2024, Online
Feb. 15, 2024
-
Global Summit on War & Cancer 2023, Online
Dec. 14-16, 2023
