Obrixtamig plus ezabenlimab represents a novel immunotherapeutic strategy being explored in small cell lung cancer (SCLC), a disease that remains one of the most challenging malignancies to treat, particularly in the relapsed setting. Despite initial sensitivity to chemotherapy, most patients experience rapid disease progression, and therapeutic options after first-line treatment remain limited. This has driven growing interest in approaches targeting tumor-specific antigens such as DLL3.
DLL3 is highly expressed on the surface of neuroendocrine tumors, including SCLC, making it an attractive therapeutic target. At the European Lung Cancer Congress 2026, early-phase data from a dose-escalation trial further evaluated this combination strategy in patients with advanced DLL3-positive
 disease.
disease.
Study Design and Patient Population
This phase I study (NCT05879978), sponsored by Boehringer Ingelheim, investigated the safety, tolerability, and preliminary efficacy of obrixtamig plus ezabenlimab in patients with advanced or relapsed DLL3-positive neuroendocrine carcinomas, including SCLC.
Eligible patients had received at least one prior line of platinum-based chemotherapy or were considered ineligible for standard treatment. Nearly half of the enrolled population had previously been treated with PD-1 or PD-L1 inhibitors, reflecting a heavily pretreated cohort with limited options.
Obrixtamig was administered weekly in three-week cycles using a step-up dosing strategy, followed by escalation to target doses ranging from 30 to 1080 μg/kg. Ezabenlimab was administered at a fixed dose of 240 mg every three weeks.
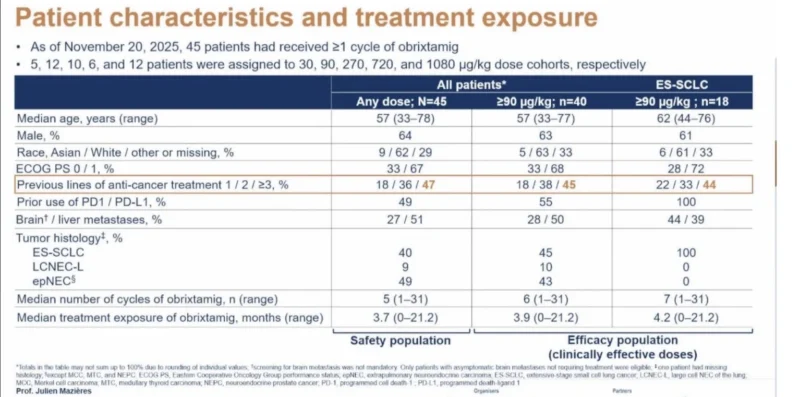
A total of 45 patients received at least one cycle of therapy, with a median of five treatment cycles and a median age of 57 years.

Safety and Tolerability
The combination demonstrated a manageable safety profile, consistent with expectations for T-cell engager therapies. Dose-limiting toxicities were observed during the step-up phase, but importantly, no dose-limiting toxicities occurred at target dose levels, and the maximum tolerated dose was not reached.
Treatment-related adverse events were common, occurring in 98% of patients, with grade 3 or higher events reported in 51%. The most frequently observed toxicity was cytokine release syndrome, reported in 78% of patients; however, all cases were low grade, and no grade 3 or higher CRS events were observed.
Neurological toxicities, a known risk with T-cell engager therapies, were relatively uncommon but clinically relevant. Grade 3 or higher neurological adverse events occurred in 7% of patients and included immune effector cell–associated neurotoxicity syndrome (ICANS) and encephalopathy.
Approximately 13% of patients discontinued treatment due to adverse events, indicating that while manageable, toxicity requires careful monitoring and supportive care.
Key Efficacy Results
The study demonstrated encouraging antitumor activity, particularly at higher dose levels of obrixtamig.
Among patients treated at clinically active doses of at least 90 μg/kg, the objective response rate was 30%, with a disease control rate of 58%. Median progression-free survival was 4.4 months, and median duration of response reached 8.8 months, suggesting meaningful clinical benefit in a heavily pretreated population.
Notably, outcomes improved at the highest dose level of 1080 μg/kg. In this subgroup, the objective response rate increased to 58%, and the disease control rate reached 83%, with a median progression-free survival of 10 months.
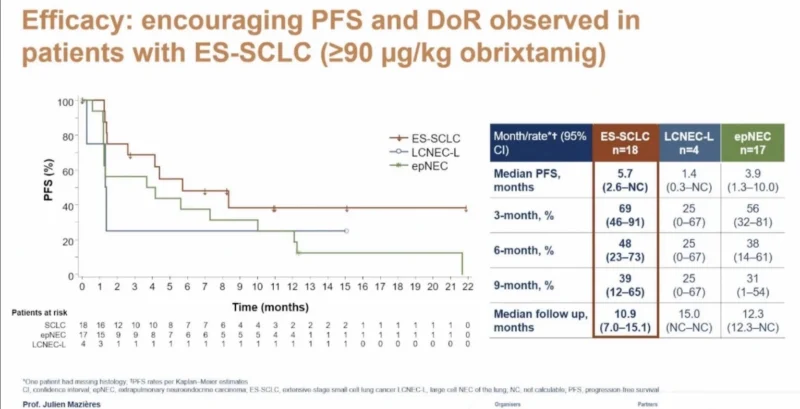
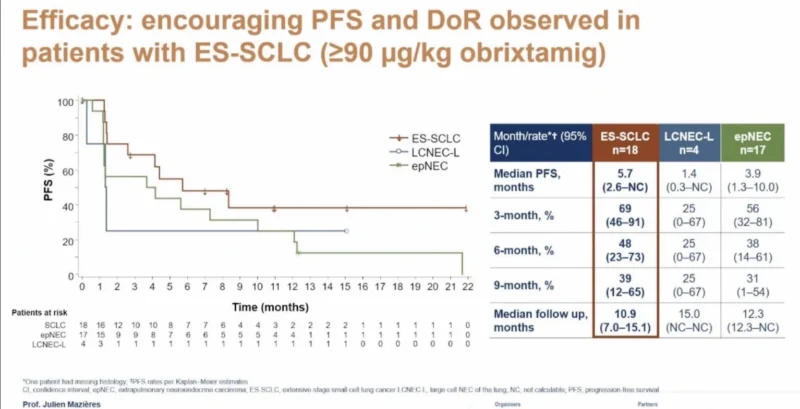
In patients with SCLC specifically, response rates were also encouraging. Among 18 patients treated at active dose levels, the objective response rate was 33%, with a disease control rate of 67% and a median progression-free survival of 5.7 months.
These findings highlight a potential dose-response relationship and support further exploration of higher-dose regimens in future studies.
Mechanistic Rationale
Obrixtamig is an IgG-like bispecific T-cell engager that simultaneously binds DLL3 on tumor cells and CD3 on T cells, promoting targeted immune-mediated tumor cell killing. Preclinical data suggest that this approach can also upregulate PD-1 and PD-L1 expression, potentially creating an adaptive resistance mechanism.
The addition of ezabenlimab, a PD-1 inhibitor, is designed to counteract this effect, enhancing antitumor immune activity. This combination strategy reflects a rational approach to overcoming immune escape in SCLC and other neuroendocrine carcinomas.
Clinical Implications
The results of this phase I trial suggest that combining DLL3-targeted T-cell engagers with immune checkpoint inhibition may represent a promising new therapeutic strategy in relapsed SCLC.
The observed efficacy, particularly at higher doses, is notable given the limited treatment options and poor prognosis in this setting. However, these findings are preliminary and require validation in larger studies to define the optimal dose, safety profile, and long-term benefit.