Health Canada approved selumetinib (Koselugo), the oral MEK1/2 inhibitor developed by Alexion, AstraZeneca Rare Disease, for the treatment of adult patients with neurofibromatosis type 1 (NF1) who have symptomatic, inoperable plexiform neurofibromas (PN) on March 9, 2026. The decision, announced from Mississauga, Ontario, was based on data from the KOMET Phase 3 trial, the largest and only placebo-controlled global study ever conducted in adult NF1 patients with PN.
The approval resolves a long-standing clinical inequity: selumetinib has been available for pediatric patients with NF1-PN in Canada, the U.S., EU, Japan, and China, but adults with the same disease mechanism have had no approved targeted therapy. For Canadian patients specifically, this decision extends access to a treatment that until now required pediatric trial enrollment or off-label use, neither a reliable nor scalable solution for adults living with a progressive, disfiguring, and often painful disease.
Understanding NF1 and Plexiform Neurofibromas: A Disease of Constant Growth
Neurofibromatosis type 1 is a rare, progressive genetic condition caused by loss-of-function mutations in the NF1 gene, which encodes neurofibromin, a negative regulator of RAS signaling. When neurofibromin is absent or non-functional, RAS remains constitutively active, driving downstream MEK/ERK pathway hyperactivation and uncontrolled cell proliferation. The result is a systemic neurocutaneous disorder affecting multiple organ systems, with a global prevalence of approximately 1 in 3,000 individuals.
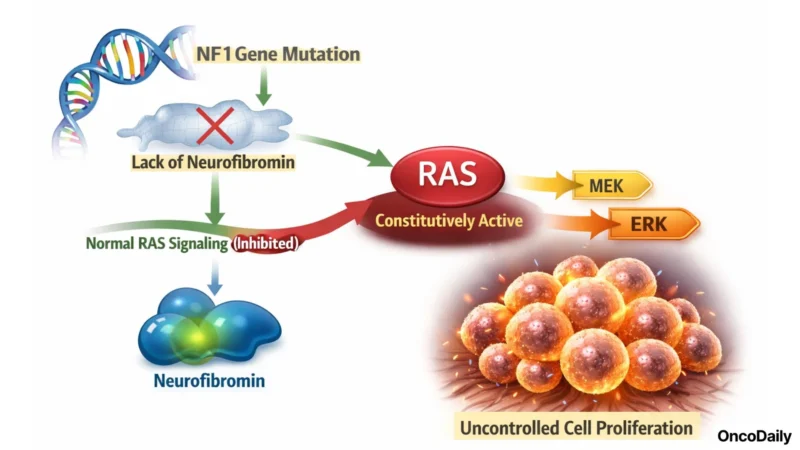
Plexiform neurofibromas (PN) are complex, diffuse peripheral nerve sheath tumors that develop along multiple nerve fascicles in up to 50% of NF1 patients. Unlike cutaneous neurofibromas, which are superficial, PNs grow deep along nerve trunks and can invade surrounding tissues, encasing blood vessels, infiltrating the spine, orbits, airway, and bladder. They grow continuously and unpredictably, and their anatomical complexity makes complete surgical resection impossible in the majority of cases without risking permanent neurological injury, airway compromise, or major vessel damage.
The clinical burden is substantial and non-abstract: disfigurement of the face or limbs, motor dysfunction, chronic pain that is often severe and refractory, visual impairment when orbital nerves are involved, and respiratory or bowel/bladder dysfunction when thoracic or pelvic nerves are infiltrated. Prior to the approval of selumetinib in pediatric patients, no drug had demonstrated a clinically meaningful tumor response in NF1-PN in a randomized controlled trial.
How Selumetinib Works: Interrupting the MEK-Driven Growth Signal
Selumetinib is an oral, selective MEK1 and MEK2 inhibitor. By blocking these kinases, the immediate downstream effectors of constitutively active RAS in NF1 tumor cells, selumetinib interrupts the proliferative signaling cascade that drives PN growth. The mechanism is not cytotoxic; rather than killing cells outright, selumetinib slows and in some cases reverses the aberrant cellular overgrowth that generates tumor volume.
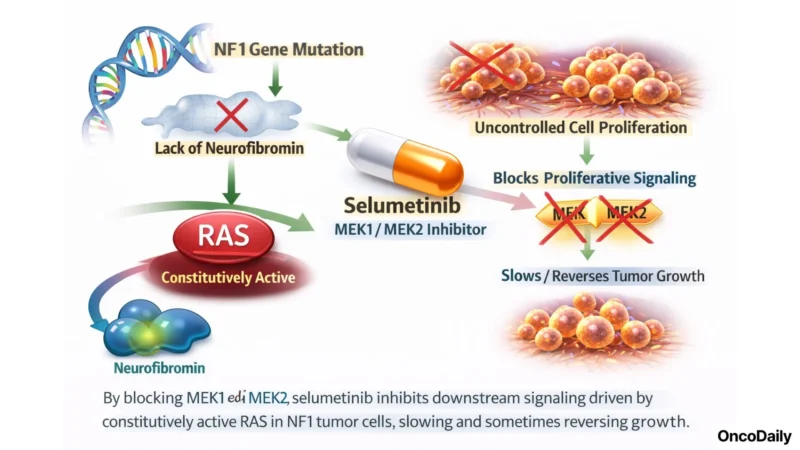
In NF1, where MEK overactivation is the direct consequence of neurofibromin loss, the mechanistic match between drug and biology is precise. The drug is dosed twice daily orally at 25 mg/m² (based on BSA), a dosing approach established in pediatric trials and carried through into the adult KOMET study.
KOMET Phase 3: Trial Design and Efficacy Results
KOMET (NCT04924608) is a global, randomized, double-blind, placebo-controlled, multicenter Phase 3 trial, the first of its kind in adult NF1 patients. Key design elements:
- Population: 145 adults (≥18 years) with NF1 and at least one symptomatic, inoperable PN measurable by volumetric MRI, randomized 1:1 to selumetinib (n=71) or placebo (n=74) for 12 cycles
- Inoperability criteria: PN defined as inoperable due to encasement of or proximity to vital structures, invasiveness, or high vascularity
- Primary endpoint: Confirmed ORR by cycle 16, assessed by independent central review using Response Evaluation in Neurofibromatosis and Schwannomatosis (REiNS) criteria, where response is defined as ≥20% reduction in PN tumor volume
Results from KOMET’s primary analysis, presented at ASCO 2025 and published in The Lancet, demonstrated:
- ORR 20% (14/71, 95% CI: 11.2–30.9) in the selumetinib arm vs. 5% (4/74, 95% CI: 1.5–13.3) with placebo (p=0.011)
- 86% of responders in the selumetinib arm had a duration of response of at least 6 months
- Chronic pain score improvement was observed in the selumetinib arm, though the difference did not reach statistical significance (p=0.070) in patients with baseline pain scores ≥3
- The safety profile was consistent with selumetinib’s established profile from pediatric use, including manageable toxicities such as gastrointestinal effects, skin toxicity, and elevated creatine phosphokinase
An ORR of 20% in a placebo-controlled PN trial, against a 5% placebo response rate, with durable responses in the majority of responders, constitutes the most robust efficacy evidence ever generated in adult NF1-PN, a population in which the prior standard of care was surgical debulking, pain management, and watchful waiting.
Karen Heim, Vice President and General Manager, Alexion Canada, spoke to the equity dimension of the approval:
Alexion is committed to bringing innovative medicines to patients with unmet need. With this approval, both children and adults living with NF1 PN now have a treatment option available to them in Canada. Koselugo was recently listed on the common drug list under the National Strategy for Drugs for Rare Disease, which is a testament to its impact on patients.

Regulatory Context: Canada Following a Wave of Global Approvals
Canada’s Health Canada decision tracks a global regulatory alignment around the KOMET dataset. The FDA approved selumetinib for adults with NF1-PN on November 19, 2025, based on the same KOMET data, following earlier approvals in the EU, Japan, and other jurisdictions. Health Canada’s March 9 decision closes the gap for Canadian patients who had watched the drug receive regulatory endorsement in peer countries while awaiting local authorization.
Notably, Koselugo’s listing on Canada’s common drug list under the National Strategy for Drugs for Rare Disease, mentioned by Alexion Canada, is a critical parallel development. Regulatory approval alone does not guarantee affordability; reimbursement listing is what converts access into real-world availability for patients who cannot pay out-of-pocket for a specialty rare disease drug. The combination of Health Canada approval and CDL listing represents a meaningful step toward genuine population-level access, not merely regulatory permission to use the drug.
EU Parallel: Tovorafenib CHMP Opinion in Pediatric Glioma
The Health Canada selumetinib decision arrived in the same week that the EMA’s CHMP issued a positive opinion for tovorafenib (Ojemda) in pediatric low-grade glioma, a regulatory event that provided simultaneous deal-adjacent validation for Servier’s $2.5 billion acquisition of Day One Biopharmaceuticals. The CHMP opinion and the Health Canada approval, landing in the same week, together illustrate an unusually dense regulatory period in rare oncology on both sides of the Atlantic, a moment when the patient communities served by these drugs are watching pipeline-to-access timelines compress.

Read more on Servier’s $2.5 billion acquisition of Day One Biopharmaceuticals on OncoDaily.
Competitive Landscape and Forward-Looking Analysis
In NF1-PN, selumetinib has no approved competitors in any jurisdiction. The condition’s rarity, the complexity of trial design in a molecularly defined rare disease, and the decade-long development timeline that preceded KOMET’s pivotal readout create meaningful barriers to competitive entry. Mirdametinib (a next-generation MEK inhibitor being studied by SpringWorks Therapeutics) and cabozantinib have shown activity in NF1 studies, but neither has reached or completed Phase 3 enrollment in adult PN specifically.
The practical implication is that selumetinib is likely to remain the only approved targeted option for adult NF1-PN for several years, a position that gives Alexion/AstraZeneca time to build real-world evidence, establish treatment guidelines, and optimize dosing protocols across the pediatric-to-adult continuum.
The remaining gap is in the population that does not respond to selumetinib, 80% of patients by the KOMET ORR. Understanding the biology of non-response, identifying biomarkers that predict tumor volume reduction, and developing combination strategies that augment the MEK inhibitor signal are the next-generation research questions that the NF1 field will need to address. The global KOMET dataset, across 13 countries and 145 carefully characterized patients, represents the richest evidence base the field has ever had for this disease. That infrastructure will matter for the next wave of trials.
Read more biotech insights on OncoDaily Biotech.
Written by: Semiramida Nina Markosyan, Editor, OncoDaily Canada


