The National Comprehensive Cancer Network (NCCN) has updated its 2026 Clinical Practice Guidelines in Oncology for bladder cancer to include Anktiva (nogapendekin alfa inbakicept-pmln) plus Bacillus Calmette-Guérin (BCG) as a Category 2A recommendation for patients with BCG-unresponsive papillary-only non-muscle invasive bladder cancer (NMIBC). This landmark guideline inclusion, announced on March 17, 2026, expands the use of ImmunityBio’s IL-15 superagonist immunotherapy beyond its current focus on carcinoma in situ (CIS), marking a significant milestone for patients who have exhausted standard BCG therapy.

You can read more about regulatory approval for ANKTIVA on OncoDaily.
Guideline Update: Expanding Treatment Beyond CIS to Papillary-Only Disease
Previously, NCCN guidelines recommended Anktiva + BCG only for BCG-unresponsive NMIBC with CIS ± papillary tumors, aligning with Anktiva’s initial FDA approval in April 2024 for CIS (with or without papillary disease). The new update (Version 1.2026, effective March 16, 2026) adds papillary-only high-grade NMIBC to the eligible patient population, meaning patients whose bladder tumors are papillary lesions without concurrent CIS can now be considered for the Anktiva + BCG regimen. The recommendation is classified as Category 2A, indicating uniform consensus by NCCN experts despite reliance on phase 2/3 trial data rather than phase 3 alone.
This guideline expansion addresses an important unmet need. Patients with papillary-only BCG-unresponsive disease, who previously had limited treatment options short of radical cystectomy, now have an immunotherapy-based, bladder-sparing alternative to consider. The NCCN guidelines are widely used by oncologists, urologists, and payers to inform treatment decisions and reimbursement, so this inclusion is expected to boost adoption and insurance coverage for Anktiva in this setting.
Patrick Soon-Shiong, M.D., Founder, Executive Chairman, and Global Chief Scientific and Medical Officer of ImmunityBio, underscored the importance of the guideline update:
These updated NCCN guideline recommendations in bladder cancer represent an important milestone for patients with BCG-unresponsive NMIBC papillary-only disease who have exhausted standard BCG therapy. The addition of Anktiva plus BCG for papillary-only disease in the NCCN guidelines reflects the growing body of clinical data evaluating Anktiva in this patient population and reinforces our commitment to developing comprehensive treatment solutions that address the full spectrum of patients living with BCG-unresponsive NMIBC.
He also shared his enthusiasm on X, highlighting the significance of recent trial publications and the need to improve access to BCG for all eligible patients:
Pleased with the NCCN guidelines for papillary only disease in non-muscle invasive bladder cancer (NMIBC). Validates the publication in Journal of Urology. Now next step is to provide BCG for all patients who could benefit in bladder cancer.

Patrick Soon-Shiong
Mechanism of Action: A Novel IL-15 Superagonist that Amplifies BCG’s Punch
Anktiva represents a first-in-class immunotherapy that synergizes with BCG by leveraging the power of the immune system’s natural killer (NK) cells and memory T cells. The drug is a novel IL-15 receptor agonist IgG1 fusion protein:
- It consists of a mutated IL-15 cytokine fused to the IL-15 receptor alpha domain, forming a complex that avidly engages IL-15 receptors on NK and CD8+ T cells.
- BCG is known to activate immune responses in the bladder; however, by adding Anktiva, NK cells and CD8+ “killer” T cells receive a potent activation signal, overcoming tumor immune evasion and generating prolonged anti-tumor memory.
- This mechanism amplifies the depth and duration of response beyond what BCG alone can achieve, effectively “supercharging” the local immune attack on bladder cancer cells.
The durable complete responses observed in trials are consistent with the mechanism: Anktiva helps restore immune surveillance in a tumor environment that previously escaped immune control. It is administered directly into the bladder (intravesical) alongside BCG, ensuring high local concentrations at the tumor site without systemic exposure. Given its unique design, Anktiva has established itself as a key component of ImmunityBio’s broader Cancer BioShield platform, which aims to enhance both innate and adaptive immunity against cancer
Trial Data Driving the Change: QUILT-3.032 Shows Durable Outcomes
The guideline update is underpinned by compelling clinical evidence from ImmunityBio’s bladder cancer development program, particularly cohort B of the phase 2/3 QUILT-3.032 trial. In this study, 80 patients with high-grade papillary-only BCG-unresponsive NMIBC received Anktiva 400 μg plus BCG 50 mg intravesically once weekly for 6 weeks:
- Primary Endpoint (12-month Disease-Free Survival): 2% of patients remained disease-free at 12 months, meeting the trial’s primary efficacy goal.
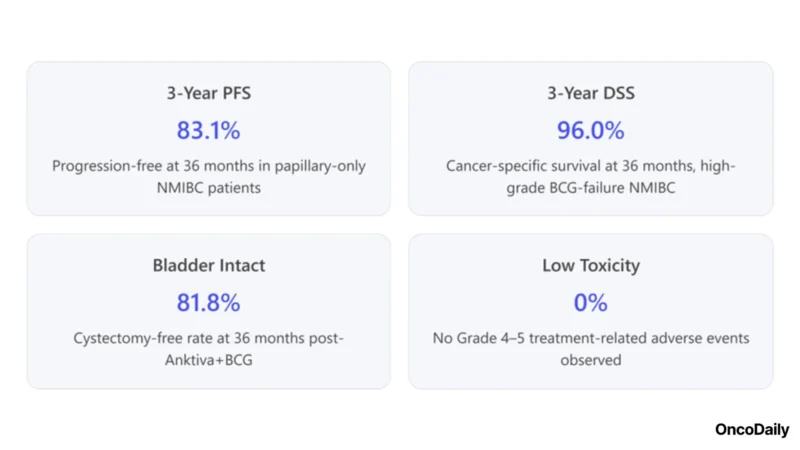
- Durable Responses: Extended follow-up showed 24-month DFS of 52.1% and 36-month DFS of 38.2%, indicating that a substantial subset of patients maintained remission up to three years.
- Progression-Free & Cancer-Specific Survival: At 36 months, progression-free survival (PFS) was 1%, and disease-specific survival (DSS) was an impressive 96.0%. Notably, median PFS and DSS were not reached at ~30 months median follow-up, signifying sustained protection against disease progression and mortality in this high-risk population.
- Bladder Preservation: Importantly, the cystectomy-free survival rate at 36 months was 8%, and the median time to cystectomy was not reached. In other words, over 80% of patients avoided bladder removal for at least three years, reflecting a major quality-of-life benefit.
- Safety Profile: The regimen’s side effects were predominantly low-grade. Across combined cohorts A and B (n = 180), 61% of treatment-related adverse events were Grade 1–2, only 3% were Grade 3, and no Grade 4 or 5 events were observed. There were no immune-related Grade ≥3 adverse events and no treatment-related deaths, indicating that Anktiva + BCG is generally well tolerated.
These practice-changing results were published in the Journal of Urology in January 2026, providing peer-reviewed validation of Anktiva’s efficacy. The data not only show that many patients can achieve prolonged remission but also that bladder cancer progression and mortality can be dramatically reduced in a setting where the historical standard of care was radical cystectomy or limited chemotherapy options.
Regulatory Status and Next Steps: FDA Review Underway
The NCCN’s endorsement of Anktiva for papillary-only NMIBC comes at a pivotal time in the therapy’s regulatory journey. ImmunityBio has an active supplemental Biologics License Application (sBLA) under FDA review, seeking to formally expand Anktiva’s label to include BCG-unresponsive papillary tumors in addition to CIS. The company resubmitted this sBLA in March 2026 after incorporating additional long-term data from QUILT-3.032.
While Anktiva is already FDA-approved (April 2024) for BCG-unresponsive NMIBC CIS, the papillary-only disease subset remains an off-label use until the sBLA is approved. The updated NCCN guidelines serve to bridge the gap by informing clinicians and payers that this regimen is an accepted option based on evidence and expert consensus. ImmunityBio’s leadership has expressed optimism that the FDA will consider the robust data favorably, aiming to align regulatory labeling with guideline recommendations in the near future.
Richard Adcock, President and CEO of ImmunityBio, noted the broader significance:
This update is an important step in the continued evolution of clinical guidance for patients with BCG-unresponsive NMIBC and validates the growing role of immune-based therapies in the treatment landscape.
If approved for papillary-only disease, Anktiva would reinforce an emerging bladder-sparing treatment paradigm alongside existing options like systemic pembrolizumab (approved for BCG-unresponsive CIS in patients ineligible for cystectomy). The convergence of guideline support and anticipated FDA expansion could accelerate uptake in clinical practice, offering patients a chance to avoid or delay cystectomy.
Competitive Landscape and Strategic Implications
The NCCN’s recognition of Anktiva + BCG for papillary-only NMIBC highlights an innovative approach in a space with few alternatives. Historically, patients whose papillary bladder tumors failed BCG often had to face:
- Radical cystectomy (bladder removal), an invasive procedure with significant morbidity.
- Off-label intravesical chemotherapy (e.g., gemcitabine, valrubicin) or systemic immunotherapy trials, which provided mixed results in this setting.
The only other immunotherapy approved after BCG failure is pembrolizumab (Keytruda), but its indication is limited to high-risk CIS (a different form of tumor) in patients who cannot undergo cystectomy. Papillary-only patients, especially those who are candidates for surgery, have not had a drug approved as an alternative to cystectomy.
With Anktiva’s emergence:
- Bladder Preservation Strategy: The high cystectomy-free survival rates observed with Anktiva + BCG demonstrate a viable bladder-sparing strategy. This is in line with a broader movement in uro-oncology to preserve organ function whenever possible.
- Guideline Inclusion Driving Adoption: NCCN’s endorsement will likely encourage more urologists and oncologists to integrate Anktiva into treatment algorithms for BCG-relapsing patients, potentially standardizing care for this subgroup. It may also prompt insurance coverage for off-label use, as payers often consider NCCN guidelines in coverage determinations.
- Clinical Pipeline Expansion: ImmunityBio’s bladder cancer program continues to advance, including a pivotal trial in BCG-naïve NMIBC (evaluating Anktiva + BCG in patients before any BCG exposure). Success in that setting could further expand the drug’s reach, possibly challenging BCG monotherapy as the first-line standard in the future.
- Market Position: Anktiva’s progress represents a commercial opportunity to capture a significant share of the NMIBC treatment market, especially as it addresses a patient segment previously underserved by novel therapies. With over 100 million insured lives already covered for the existing indication , the expansion into papillary-only disease could bolster ImmunityBio’s growth and justify its heavy investment in IL-15-based technology.
Lastly, the broader trend in oncology is clear: immune-based therapies are increasingly taking center stage even in earlier-stage cancers, reducing reliance on radical surgeries. The NCCN’s guideline update for bladder cancer is a prime example of this shift, reinforcing the importance of harnessing the immune system to achieve cures or sustained remissions while maintaining quality of life.
Key Takeaways
- NCCN Update: 2026 NCCN Guidelines for Bladder Cancer now include Anktiva (IL-15 agonist) + BCG for BCG-unresponsive papillary-only NMIBC, expanding beyond the prior CIS-focused recommendation.
- Milestone for Patients: Offers a new bladder-sparing immunotherapy option for patients who would otherwise face radical cystectomy or limited alternatives after BCG failure.
- Clinical Efficacy: Phase 2/3 trial data (QUILT-3.032) showed 2% 12-month DFS, with durable 36-month outcomes: 83.1% PFS, 96.0% DSS, and 81.8% cystectomy-free survival, demonstrating long-term disease control.
- Safety: Manageable toxicity profile; majority Grade 1–2 side effects and no severe treatment-related adverse events, enabling patients to complete therapy without significant interruptions.
Read more biotech insights on OncoDaily Biotech.
Written by: Semiramida Nina Markosyan, Editor, OncoDaily Canada


