Andrea Necchi, Associate Editor at Journal of Clinical Oncology, shared on X about a recent paper he and colleagues co-authored:
“Interim results of Sunrise4 are released The Lancet Oncology
Neoadjuvant TAR200 + cetrelimab vs cetrelimab monotherapy in patients with MIBC
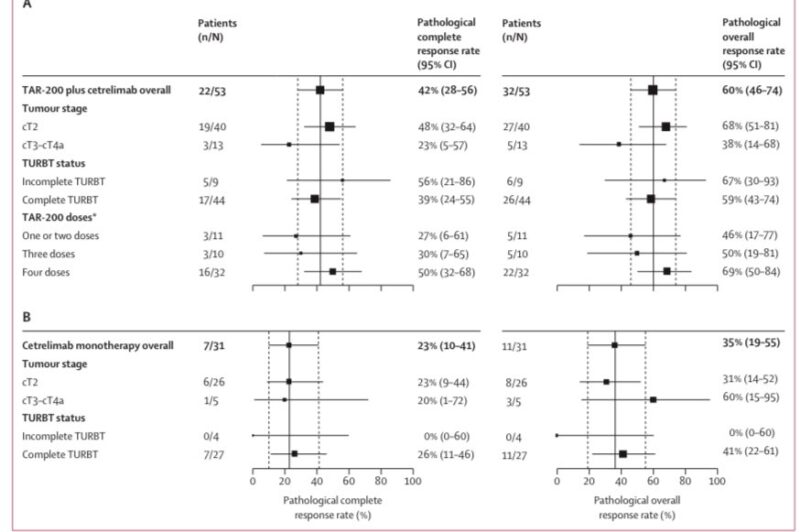
TAR200+CET: pCR 42%, Major path Response 60%
in cT2 pts baseline: pCR
48% TAR200 exposure-response association: pCR 50% if TAR200x4 full cycles
Can we cure selected patients with MIBC with intravesical Rx + single agent IO?
Future: is there a way to avoid radical cystectomy by consolidating Response with TAR200 in selected patients?
Primary results with full biomarker results are awaited.”
Daniel Castellano, Medical Oncologist at University Hospital October 12, shared this post, adding:
“yes, that is the way for MIBC! Intravesical therapy + systemic therapy to avoid the radical Surgery!!”
Title: TAR-200 plus cetrelimab versus cetrelimab monotherapy as neoadjuvant therapy in patients with muscle-invasive bladder cancer who are ineligible for or decline neoadjuvant cisplatin-based chemotherapy (SunRISe-4): interim analysis of a randomised, open-label phase 2 trial
Authors: Andrea Necchi, Félix Guerrero-Ramos, Paul L Crispen, Bernardo Herrera-Imbroda, Rohan Garje, Thomas Powles, Charles C Peyton, Benjamin Pradere, Ja Hyeon Ku, Neal Shore, Martin Bögemann, Mark A Preston, Evanguelos Xylinas, Cristina Sanchez de Llano, Mohamad Hasan, Hind Stitou, Sumeet Bhanvadia, Hussein Sweiti, Sarah P Psutka

More posts featuring Andrea Necchi on OncoDaily.


