In prostate cancer radiotherapy, spacing refers to the intentional separation of the prostate and anterior rectal wall along the rectoprostatic fascia, creating a physical displacement that reduces rectal dose exposure during external beam radiation therapy (EBRT). Spacers are implantable devices or substances utilized for this purpose, which remain in place during a regimen of radiation and are resorbed by the patient’s body after a course of treatment concludes.
In localized prostate cancer cases, radiation-based therapies offer a potentially curative alternative to radical prostatectomy, while providing similar oncologic control.1,2 As advancements in imaging, planning and fractionation have driven continuous innovation in EBRT,3 spacer technologies have also evolved.
This article will discuss the role of spacing in prostate cancer radiation treatment planning, recent developments in spacer technology, and how the ongoing shift toward shorter-course radiation therapies has led to further innovations in spacing.
The Role of Spacing: Creating a Safety Margin
The prostate and rectal wall are separated by only a few millimeters. Consequently, even the most advanced forms of radiation treatment carry a risk of unintentional irradiation of the rectum. Along with fractionation optimization, imaging, and delivery techniques, rectal spacing is a key clinical strategy for reducing irradiation-associated risks.
Multiple randomized and prospective studies have demonstrated the benefits of perirectal spacing.4,5,6 Increasing the amount of separation between the prostate and rectum lowers rectal dose exposure, reducing toxicity risk, and improving the therapeutic ratio.
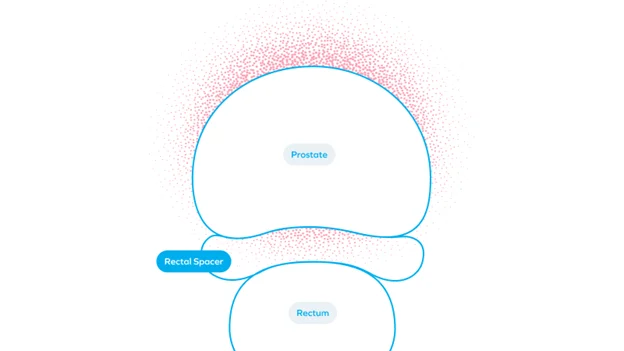
Spacers are also an important component for lowering radiation exposure for advanced treatment. Technological advancements are enabling safe dose escalation,7,8 and higher-dose regimens are increasingly selected by clinicians and patients for convenience and tumor-control potential.9 Such regimens increase dose per fraction and reduce the margin for error which heightens the need for anatomic precision. Rectal spacing adds a safety margin, lowering rectal dose and toxicity while helping preserve quality of life.4,5,10
The Shift Toward Shorter-Course Regimens
Advancements in rectal spacing have been driven by a clear movement toward shorter-course radiotherapy.9 Modern approaches such as moderate hypofractionation and ultra-hypofractionation/Stereotactic Body Radiation Therapy (SBRT) are now commonplace in clinical practice.9
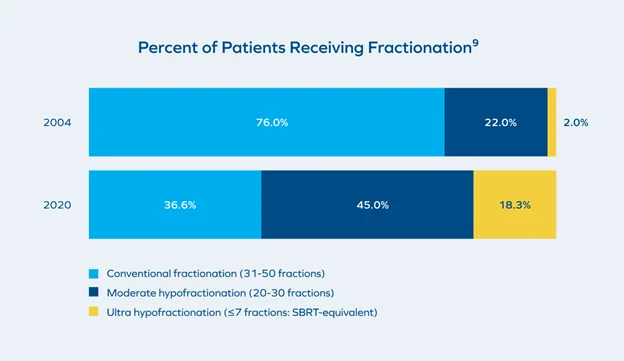
Technological shifts, clinical validation, and the potential convenience of fewer visits are factors that may be driving the movement toward shorter treatment courses.9 Advancements in imaging, planning, and delivery have enabled precise targeting at higher dose per fraction,11 and randomized trials and guidelines have established these approaches as safe and effective standards for localized prostate cancer.8,11,14 Guidelines from ASTRO/ASCO/AUA and NCCN endorse moderate hypofractionation as a standard option for localized prostate cancer, with SBRT recognized as an accepted treatment option for appropriately selected low- and favorable intermediate-risk patients.11,12
Dosimetric Consequences and Clinical Implications of Shorter Courses
For a clinical team planning for radiotherapy to treat prostate cancer, the shift toward shorter-course regimens has reshaped questions around dosimetry and risk. As dose per fraction increases, the margin for error narrows, and anatomical or intrafraction motion can influence dose to the target and surrounding tissue.13
In localized prostate cancer cases, the rectum is the primary dose-limiting structure, and with fewer fractions, each exposure carries greater biological effect on surrounding tissues.11 Protecting normal tissue is central to modern prostate radiation therapy, and rectal spacing serves as a component of risk mitigation as practice evolves.4,5,10
The Value of Apical Spacing: Coverage Where It Matters Most
The apex of the prostate is the closest region to the anterior rectal wall, with just a few millimeters creating a narrow separation between the gland and bowel.15 Because separation is smallest at the prostate apex, even modest variations in spacing can increase rectal dose.16 Consistent base-to-apex spacing significantly reduces high-dose rectal exposure and rectal toxicity in randomized controlled data.4
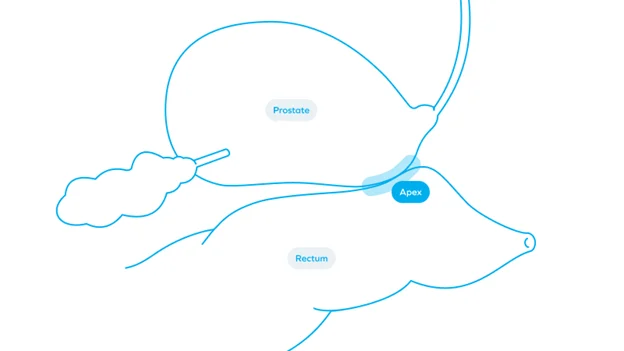
As treatment planning standards have advanced, spacing in the apical region has become a growing concern. Higher-dose, fewer-fraction regimens have become widely adopted,9 and advanced prostate radiotherapy protocols rely on strict rectal dose-volume constraints.17 With continued adoption of SBRT and adaptive image guidance, the field is entering a new phase,9 in which spacing is evolving along with modern treatment demands.
Shifting Spacing Strategies
Prostate-rectal spacing has historically been achieved using injectable gel spacers placed between the prostate and rectum. Polyethylene glycol (PEG)–based hydrogels polymerize in situ after injection and are intended to create separation between the prostate and the anterior rectal wall. An alternative gel spacer utilizes non-animal stabilized hyaluronic acid (NASHA), a viscoelastic gel described as non-polymerizing and intended for the same general purpose.
A recent entrant to the field is a non-gel balloon spacer for prostate cancer radiation therapies, which has been brought to market as the BioProtect Balloon Spacer. A pre-formed balloon with predictable volume and geometry, the BioProtect Balloon Spacer is a biodegradable Poly(L-lactide-co-ε-caprolactone) (PLCL) furled balloon that is placed utilizing a blunt dilator, and then inflated in situ with standard 0.9% saline solution to create 10-18 mm of space.19 In addition to creating symmetric space,19 BioProtect offers consistent apex-to-base separation and stable space with apical coverage.19,22
The Apex of Spacing: Clinical Evidence of Balloon Spacer Performance
Balloon spacer utilization is supported by a robust and growing body of evidence. The BioProtect Balloon Spacer has appeared in 22 studies and trials. The spacer has also been evaluated in a pivotal prospective multicenter randomized controlled subject-blind phase III trial of 222 subjects across 16 sites, published in the International Journal of Radiation Oncology, Biology, Physics.4
Co-primary trial endpoints were efficacy (≥25% reduction in rV70 in >75% subjects) and safety (non-inferiority), with prostate-rectal wall separation, additional dosimetry, and patient-reported quality of life (EPIC) as secondary endpoints.4
The balloon spacer met the efficacy endpoint, with >25% rectal V70 reduction in 97.9% of BioProtect subjects (p < 0.001), as well as exhibiting dose reduction performance across dosimetric parameters.4
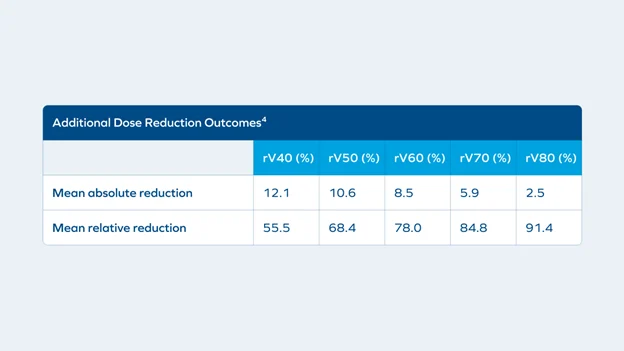
The safety endpoint was also met, with 18% of BioProtect subjects experiencing rectal and implantation procedure-related adverse events vs. 23.1% for control (p < 0.001), resulting in demonstrated non-inferiority.4

Measured pivotal trial secondary endpoints support findings of post-implantation space creation with the balloon spacer, as well as stability across the treatment course and after-treatment resorption.4

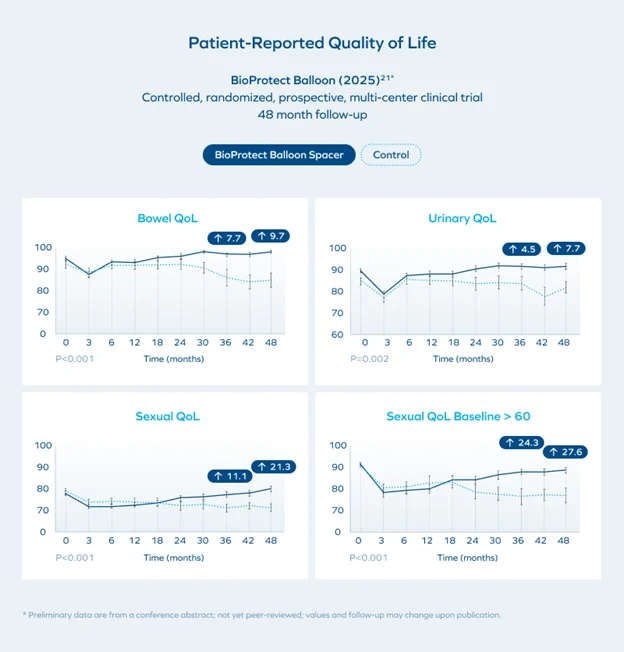
Prioritizing Life After Treatment: Quality of Life Preservation
Adequate spacing at the apex of the prostate has been associated with lower off-target dose and preservation of quality of life (QoL).16 Apical separation ≥14 mm has been shown to reduce radiation dose to the rectum, penile bulb, and bladder compared with separation <14 mm.22 In a hydrogel spacer trial, apical separation ≥10 mm correlated with less QoL decline.16 Comparative data demonstrate that balloon spacers reliably create measurable apex spacing, while gel spacers may not consistently do so.21*
Extended abstract follow-up of BioProtect Balloon Spacer pivotal trial subjects over 48 months demonstrated preserved patient-reported bowel, urinary, and sexual quality of life, along with lower rates of rectal and urinary toxicities.21*This is the first study to show a statistically significant improvement in sexual quality of life over control.21*
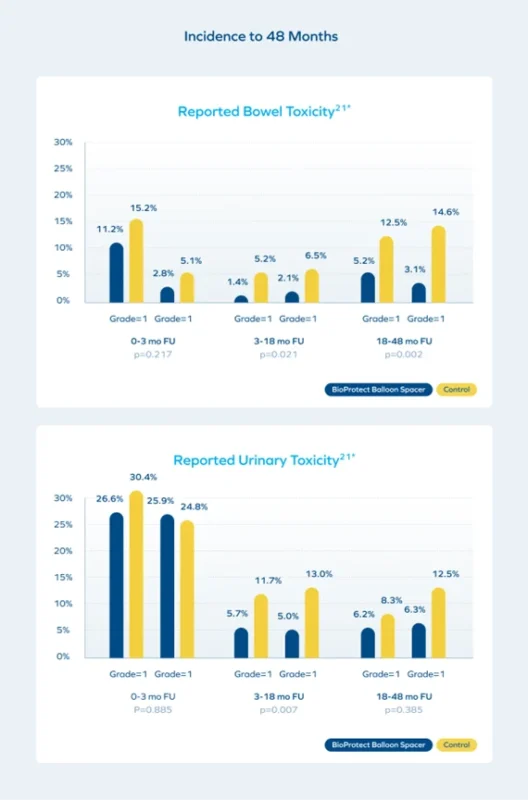
Short-Course Protection: Balloon Spacer Dosimetry
Relative dosimetric performance of the balloon spacer has been studied in a multicenter, double-arm, central core lab retrospective comparison of rectal dosimetry of BioProtect Balloon Spacer and a leading gel spacer in 59 subjects.20 The balloon spacer achieved significantly greater mean rectal V70 reduction than the gel spacer in conventional fractionation.20 Rectal dose reductions were also observed across all dose levels in both conventional and ultra hypofractionated regimens.20

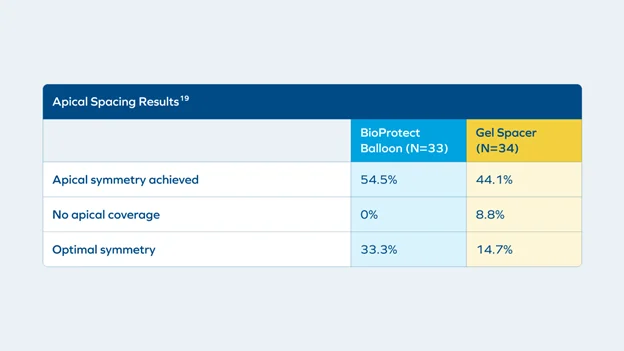
Apical Performance: Greater Symmetry, More Uniform Separation
Consistent separation of base, mid-gland, and apex with the BioProtect Balloon Spacer has been observed in a retrospective dual-arm comparative study of geometry, symmetry, rectal dosimetry, and GI/GU toxicity of BioProtect Balloon Spacer and a PEG gel spacer utilizing 67 subjects.19 In the study, the balloon achieved anterior-posterior spacing that was significantly greater at apex, mid-gland, and base (p <0.001), as well as more uniform separation, including apical symmetry. Apical separation was achieved in 100% of subjects.19
Key Takeaways
Prostate cancer radiotherapy is shifting toward shorter courses, dose escalation, and advanced fractionation. Consistency, predictability, and apical coverage matter more than ever, and additional clinical strategies for managing irradiation-associated risk are gaining increased relevance.
For clinicians seeking risk mitigation strategies that aid treatment planning, potential benefits of utilizing a balloon spacer include:
● The creation of consistent base-to-apex separation with reliable apical coverage.4,19,20
● Dosimetry well-suited for modern hypofractionation and SBRT planning approaches.4,20
● Preservation of patient quality of life post-treatment, including bowel, urinary, and sexual benefits.21*
Additional information about the BioProtect Balloon Spacer can be obtained at BioProtect.com.
References
1.Sanda, Martin et al. “Clinically localized prostate cancer: AUA/ASTRO guideline (2022).” Journal of urology. 2022;208(4):793-800. doi:10.1097/JU.0000000000002757
2.National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Prostate Cancer. Version 1.2025. Published 2025. Accessed February 10, 2026.
3.Morgan, Scott et al. “Hypofractionated radiation therapy for localized prostate cancer: executive summary of an ASTRO, ASCO, and AUA evidence-based guideline.” Practical radiation oncology. 2018;8(6):354-360. doi:10.1016/j.prro.2018.08.002
4.Song, Daniel et al. “Prospective, Randomized Controlled Pivotal Trial of Biodegradable Balloon Rectal Spacer for Prostate Radiation Therapy.” International journal of radiation oncology, biology, physics vol. 120,5 (2024): 1410-1420. doi:10.1016/j.ijrobp.2024.07.2145
5.Mariados, Neil et al. “Hydrogel Spacer Prospective Multicenter Randomized Controlled Pivotal Trial: Dosimetric and Clinical Effects of Perirectal Spacer Application in Men Undergoing Prostate Image Guided Intensity Modulated Radiation Therapy.” International journal of radiation oncology, biology, physics vol. 92,5 (2015): 971-977. doi:10.1016/j.ijrobp.2015.04.030
6.Mariados, Neil et al. “Hyaluronic Acid Spacer for Hypofractionated Prostate Radiation Therapy: A Randomized Clinical Trial.” JAMA oncolology. 2023;9(4):511–518. doi:10.1001/jamaoncol.2022.7592
7.Zelefsky, Michael et al. “Improved clinical outcomes with high-dose image guided radiotherapy compared with non-IGRT for the treatment of clinically localized prostate cancer.” International journal of radiation oncology, biology, physics. 2012;84(1):125-129. doi:10.1016/j.ijrobp.2011.11.047
8.Widmark, Anders, et al. “Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial.” Lancet. 2019;394(10196):385-395. doi:10.1016/S0140-6736(19)31131-6
9.Yu, James et al. “Increasing use of shorter-course radiotherapy for prostate cancer.” JAMA oncology. 2023;9(12):1696-1701. doi:10.1001/jamaoncol.2023.4267
10.Hamstra, Daniel et al. “Continued benefit to rectal separation for prostate radiation therapy: final results of a phase III trial.” International journal of radiation oncology, biology, physics. 2017;97(5):976-985. doi:10.1016/j.ijrobp.2016.12.024
11.Morgan, Scott et al. “Hypofractionated Radiation Therapy for Localized Prostate Cancer: An ASTRO, ASCO, and AUA Evidence-Based Guideline.” Journal of clinical oncology. 2018;36(34):JCO1801097. doi:10.1200/JCO.18.01097
12.Spratt, Daniel et al. “Prostate cancer, version 3.2026, NCCN Clinical Practice Guidelines in Oncology.” Journal of the national comprehensive cancer network. 2025;23(11):469-493. doi:10.6004/jnccn.2025.0052
13.Faccenda, Valeria et al. “Dosimetric Impact of Intrafraction Prostate Motion and Interfraction Anatomical Changes in Dose-Escalated Linac-Based SBRT.”Cancers (Basel). 2023;15(4):1153. doi:10.3390/cancers15041153
14.Jackson, William et al. “Stereotactic body radiation therapy for localized prostate cancer: a systematic review and meta-analysis of over 6,000 patients treated on prospective studies.” International journal of radiation oncology, biology, physics. 2019;104(4):778-789. doi:10.1016/j.ijrobp.2019.03.051
15.Song, Danny et al. “A multi-institutional clinical trial of rectal dose reduction via injected polyethylene-glycol hydrogel during intensity modulated radiation therapy for prostate cancer: analysis of dosimetric outcomes.” International journal of radiation oncology, biology, physics. 2013;87(1):81-87. doi:10.1016/j.ijrobp.2012.12.019
16.King, Martin et al. “Evaluating the Quality-of-Life Effect of Apical Spacing with Hyaluronic Acid Prior to Hypofractionated Prostate Radiation Therapy: A Secondary Analysis.” Practical Radiation Oncology. 2024; May-Jun;14(3):e214-e219
17.Wang, Kyle et al. “Prostate SBRT: An Overview of Genitourinary / Rectal Toxicity and Dose Response.” International journal of radiation oncology, biology, physics. 2021 May 1;110(1):237-248. doi: 10.1016/j.ijrobp.2020.09.054
18.BioProtect Balloon ImplantTM System 510(k) K222972, August 2023.
19.Ben-Dor, Yossi et al. “Comparative Evaluation of Symmetry, Dosimetry, and Toxicity in Prostate Cancer EBRT with Spacing Techniques.” Frontiers in oncology. 2025;15:1668726. doi: 10.3389/fonc.2025.1668726
20.Kos, Michael et al. “Multicenter, dual fractionation scheme, single core lab comparison of rectal volume dose reduction following injection of two biodegradable perirectal spacers.” Journal of applied clinical medical physics vol. 24,10 (2023): e14086. doi:10.1002/acm2.14086
21.Song, Daniel et al. “Long-term toxicity and patient-reported quality of life after prostate IMRT with or without biodegradable balloon rectal spacer: analysis of a pivotal randomized trial.” Abstract presented at: American Society for Radiation Oncology (ASTRO) 67th Annual Meeting; 2025; San Francisco, CA.
22.Ng, Victor C et al. “Prostate–Rectum Spacing from Apex to Base and Its Impact on Organs-At-Risk Dosimetry in Prostate Cancer SBRT.” Radiation 2026;6:8. doi:10.3390/radiation6010008
* Preliminary data are from a conference abstract; not yet peer-reviewed; values and follow-up may change upon publication.


