Endometrial cancer is one of the most common gynecologic malignancies, with rising incidence and mortality in advanced and recurrent disease. Molecular classification—originating from The Cancer Genome Atlas and refined through systems such as ProMisE—has defined biologically distinct subgroups, including mismatch repair–deficient and mismatch repair–proficient tumors, each with different therapeutic sensitivities.
Immune checkpoint inhibitors have demonstrated clear benefit in dMMR/MSI-H disease, whereas the much larger pMMR population—representing roughly 70–80% of cases—has shown limited responsiveness to immunotherapy alone. This has driven the development of combination strategies integrating chemotherapy, immunotherapy, anti-angiogenic therapy, or PARP inhibition to overcome intrinsic immune resistance.
The phase III DUO-E trial evaluated the addition of durvalumab to carboplatin–paclitaxel followed by maintenance durvalumab with or without olaparib in advanced or recurrent endometrial cancer. Beyond demonstrating progression-free survival benefit, particularly with olaparib maintenance, the study provided an opportunity to explore the biological heterogeneity of pMMR disease and its implications for biomarker-guided treatment selection.
What treatment looked like before DUO-E
For newly diagnosed advanced or recurrent endometrial cancer, standard systemic therapy has historically centered on:
- Carboplatin + paclitaxel (CP) as the foundational regimen
- Immunotherapy with the clearest single-agent activity mainly in dMMR/MSI-H, while pMMR generally required combination strategies to generate consistent benefit
What DUO-E tested
DUO-E randomized patients 1:1:1 to:
- CP (control strategy)
- CP + durvalumab → durvalumab maintenance
- CP + durvalumab → durvalumab + olaparib maintenance
The exploratory paper focuses on the pMMR subgroup, asking: How do PD-L1, TP53, HRR/BRCA alterations, and histology overlap — and do these features change the direction of benefit?

The Key Efficacy Signal to Remember
Even within mismatch repair–proficient (pMMR) endometrial cancer, the DUO-E primary analysis revealed a clear stepwise improvement in outcomes.
Adding durvalumab to carboplatin–paclitaxel chemotherapy significantly prolonged progression-free survival compared with chemotherapy alone (HR 0.77).
Importantly, extending this strategy with olaparib maintenance after durvalumab-based therapy produced an even greater benefit, further reducing the risk of progression (HR 0.57).
Clinical interpretation:
In pMMR disease, the meaningful therapeutic advance is not simply the introduction of immunotherapy, but the integration of immune checkpoint blockade with chemotherapy followed by maintenance intensification. Within this framework, the combination of durvalumab plus olaparib delivers the strongest progression-free survival signal and represents the most biologically and clinically compelling strategy emerging from DUO-E.
Biological Insights from the Exploratory Biomarker Analyses
The exploratory analyses of DUO-E provide an important biological framework for understanding treatment activity in mismatch repair–proficient (pMMR) endometrial cancer, extending beyond simple efficacy comparisons.
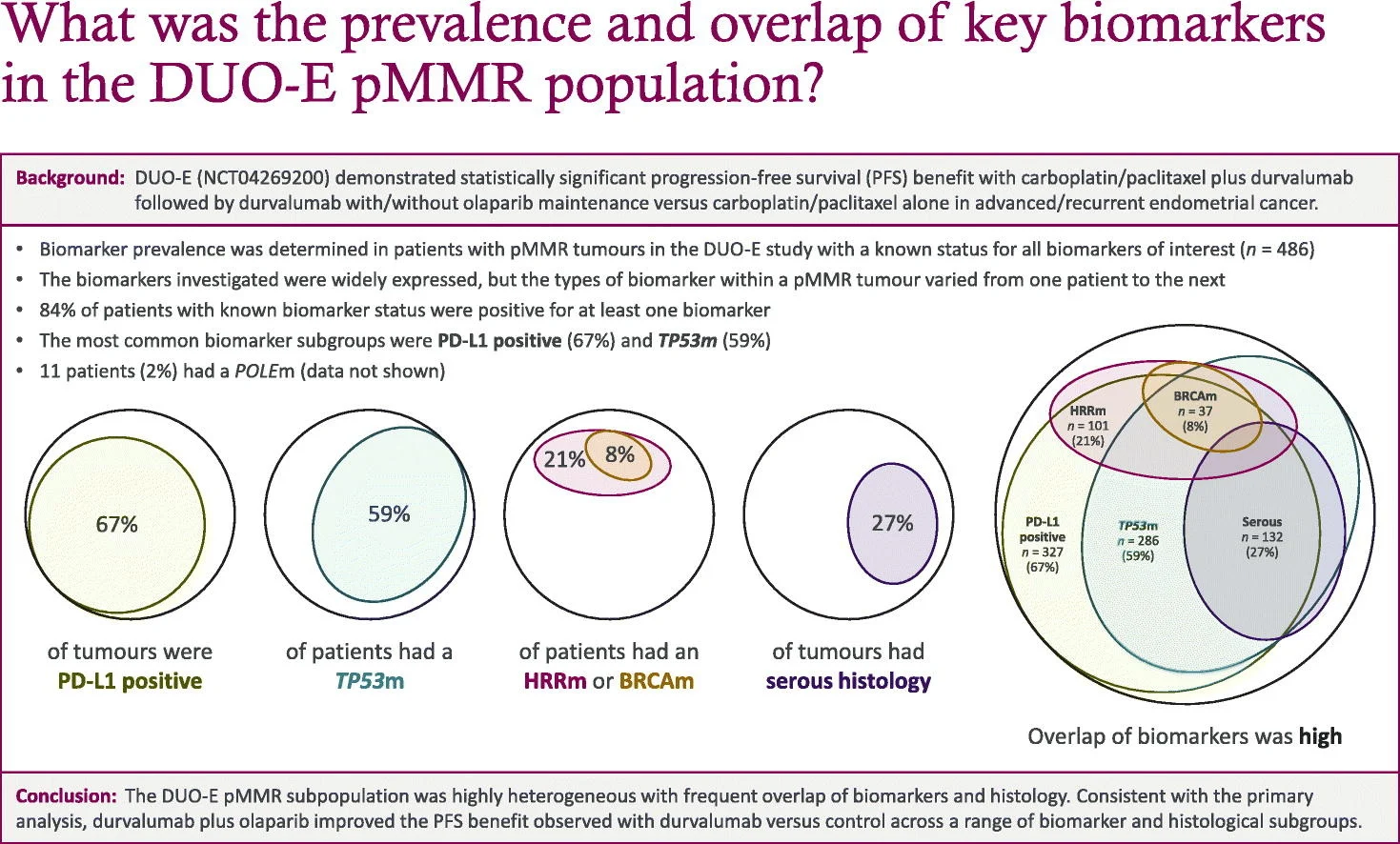
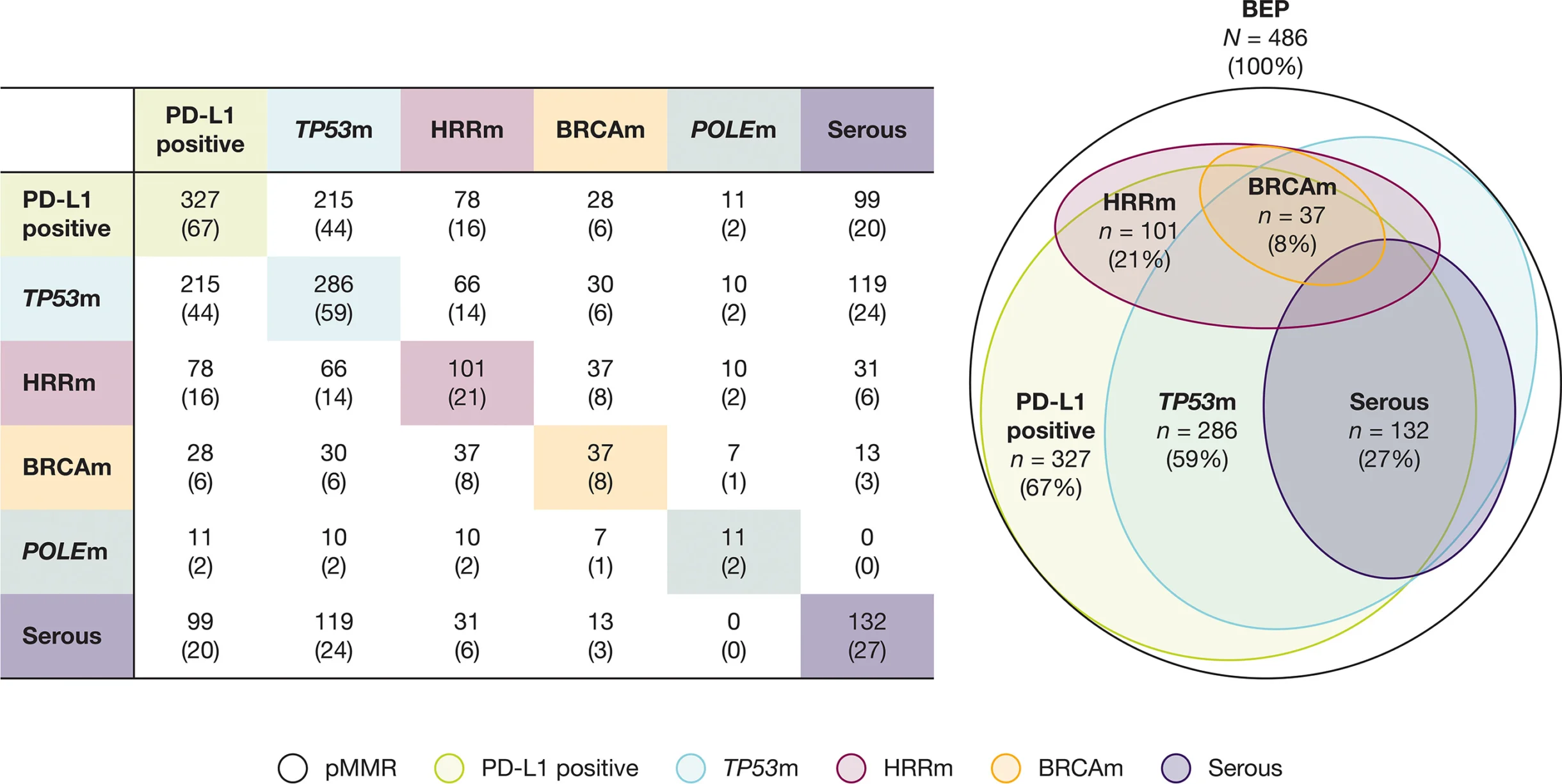
pMMR disease is molecularly dense and highly overlapping
Most pMMR tumors harbored at least one actionable or biologically relevant alteration, and co-occurrence of multiple biomarkers was common. Tumors could simultaneously demonstrate PD-L1 expression, TP53 mutation, or homologous recombination repair (HRR) alterations, while histologic subtype—such as serous morphology—showed association with certain genomic features (particularly TP53 mutation) but did not fully define tumor biology.
Collectively, these findings reinforce that pMMR endometrial cancer represents a heterogeneous molecular ecosystem rather than a single clinical entity.
Clinical benefit is not restricted to a single biomarker-defined subgroup
Across a wide range of biomarker and histologic strata, progression-free survival hazard ratios consistently favored durvalumab-containing regimens over chemotherapy alone, with the durvalumab plus olaparib maintenance strategy demonstrating the most consistent signal across subgroups.
This pattern suggests that treatment efficacy in pMMR disease is platform-driven rather than biomarker-exclusive, reflecting the combined biological impact of cytotoxic priming, PD-L1 blockade, and PARP-mediated stress signalingoperating across diverse tumor contexts.
Biological rationale for PARP inhibition in pMMR beyond BRCA-mutated disease
The observed activity of olaparib maintenance in pMMR tumors lacking classical BRCA mutations supports an expanded mechanistic model. PARP inhibition can amplify DNA damage accumulation, genomic instability, and downstream inflammatory signaling, thereby increasing neoantigen exposure and immune recognition.
This immune-modulatory effect provides a plausible basis for synergy with PD-L1 blockade, particularly in tumors that are not intrinsically immune-inflamed.
Overall biological interpretation
Taken together, the DUO-E exploratory analyses shift the conceptual framework of pMMR endometrial cancer from single-biomarker selection toward a multidimensional therapeutic platform, where integrated targeting of DNA damage response, immune checkpoints, and chemotherapy-induced tumor priming enables clinically meaningful benefit across biologically heterogeneous disease.
Key Takeaway Messages
- DUO-E redefines therapeutic expectations in pMMR endometrial cancer.
The trial demonstrates that chemo-immunotherapy provides clinically meaningful benefit in pMMR disease, with durvalumab plus olaparib maintenance further enhancing progression-free survival, thereby shifting the historical perception of pMMR tumors as uniformly immunotherapy-insensitive. - pMMR tumors represent a biologically heterogeneous and overlapping molecular landscape.
High rates of concurrent biomarker positivity indicate that single-parameter stratification—such as PD-L1 expression or histology alone—cannot adequately explain treatment response, underscoring the need for multidimensional biological interpretation. - Treatment effect appears platform-driven rather than biomarker-restricted.
Consistent hazard ratio trends across multiple biomarker and histologic subgroups suggest that benefit arises from the combined therapeutic architecture (chemotherapy priming, PD-L1 blockade, and PARP-mediated genomic stress) rather than dependence on a single predictive marker. - Clinical implications favor integrated, biology-aware stratification.
These findings support a transition toward composite clinical decision-making incorporating MMR status, molecular alterations, and histologic context, while emphasizing caution against over-interpretation of exploratory subgroup signals in the absence of prospective validation.
You Can Read All Article Here


