Allogeneic stem cells are emerging as a powerful foundation for next-generation immunotherapies, offering the potential for scalable, off-the-shelf treatments that overcome the manufacturing and timing limitations of autologous platforms. Building on this concept, researchers at UCLA have developed an innovative approach for one of the deadliest cancers: pancreatic cancer. Despite advances in chemotherapy, pancreatic tumors remain exceptionally difficult to treat due to their dense stroma, profound immunosuppression, antigen heterogeneity, and high metastatic potential. Traditional CAR-T cell therapies have repeatedly fallen short in this setting, hindered by poor tumor penetration, limited persistence, and rapid immune escape.
To address these barriers, the UCLA team engineered IL-15–enhanced, mesothelin-targeted CAR-NKT cells derived from human cord blood hematopoietic stem and progenitor cells (HSPCs). These allogeneic CAR-NKT cells—referred to as Allo15MCAR-NKT—leverage the antitumor specificity of CAR technology while preserving the innate cytotoxicity, rapid trafficking, and low immunogenicity inherent to invariant natural killer T (iNKT) cells. Because they originate from engineered stem cells, the therapy can be manufactured at scale, stored, and administered without the need for patient-specific cell collection. This combination of durability, universal availability, and biological versatility represents a major step toward a clinically viable immunotherapy for pancreatic cancer.
CAR T-Cell Therapy vs iNKT Cell Therapy: The Future of Engineered Immune Cells in Cancer Treatment
A Scalable and Clinically Adaptable Manufacturing Platform
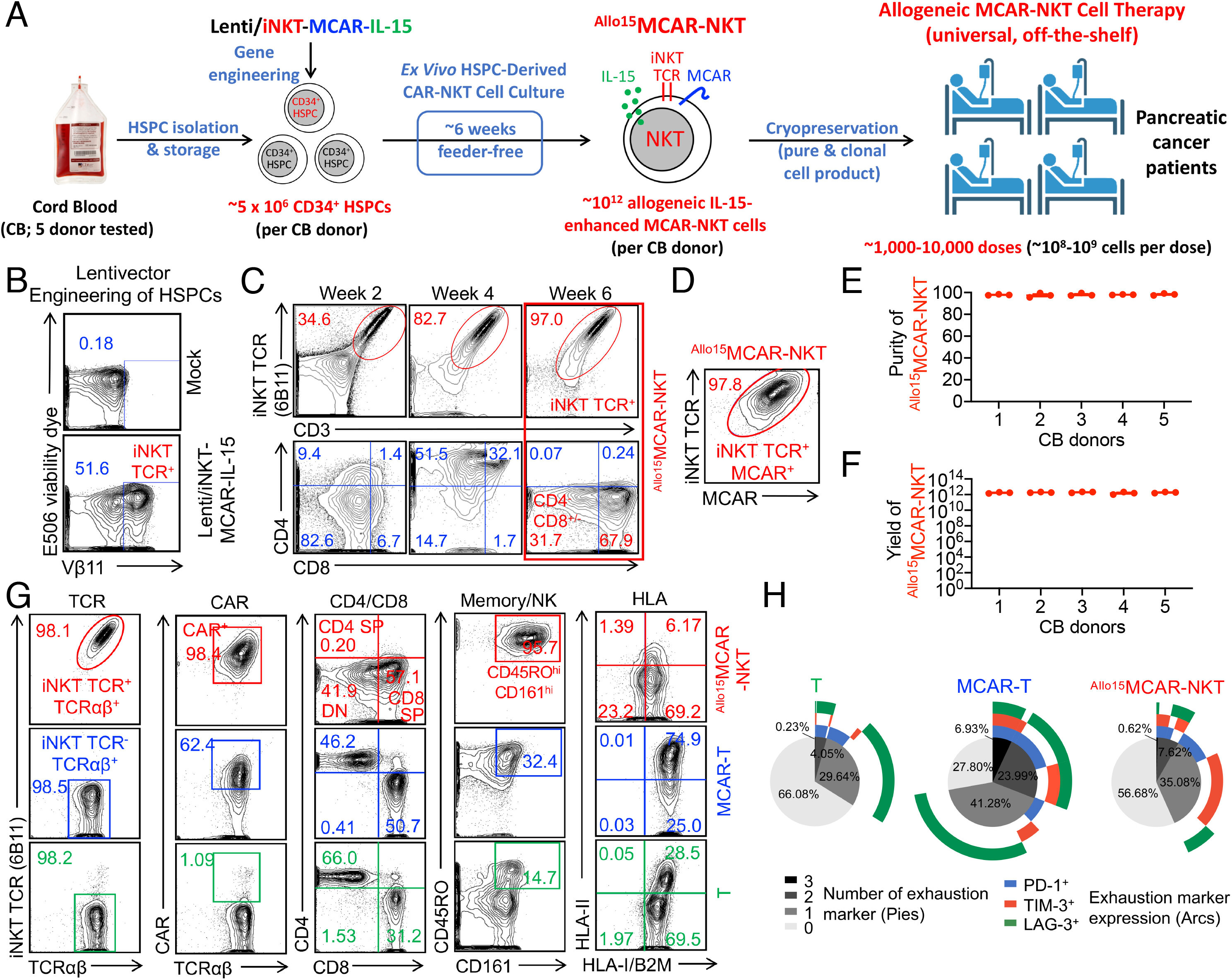
Researchers engineered human cord blood CD34+ HSPCs with a single lentiviral vector encoding:
- The invariant NKT TCR
- A mesothelin-targeting 3rd-generation CAR (SS1 scFv with CD28 + 4-1BB costimulation)
- Human soluble IL-15 to enhance persistence and effector function
Using a 6-week feeder-free differentiation protocol, the team generated highly pure (>97%) CAR+/iNKT+ cell populations with consistent quality across donors. Remarkably, a single cord blood donor yielded over 10¹² therapeutic cells, enough for thousands of patient doses—something impossible with autologous CAR-T manufacturing.
These cells displayed a hybrid T/NK phenotype, high expression of activating NK receptors (NKG2D, DNAM-1, NKp30, NKp44), strong effector programs, and low levels of exhaustion markers such as PD-1, TIM-3, and LAG-3.
Dual CAR + NK Receptor Cytotoxicity Overcomes Antigen Escape
In vitro assays showed that Allo15MCAR-NKT cells:
- Killed both MSLN-positive and MSLN-negative pancreatic cancer cells
- Eliminated tumor heterogeneity in mixed cultures
- Maintained cytotoxicity through serial tumor killing assays, reflecting resistance to exhaustion
- Exhibited reduced function only when NKG2D or DNAM-1 were blocked, confirming NK receptor–mediated tumor recognition
Conventional CAR-T cells failed against MSLN-negative or heterogeneous tumors, highlighting a major advantage of CAR-NKT therapy in overcoming antigen escape—a key barrier in solid tumor CAR therapy.
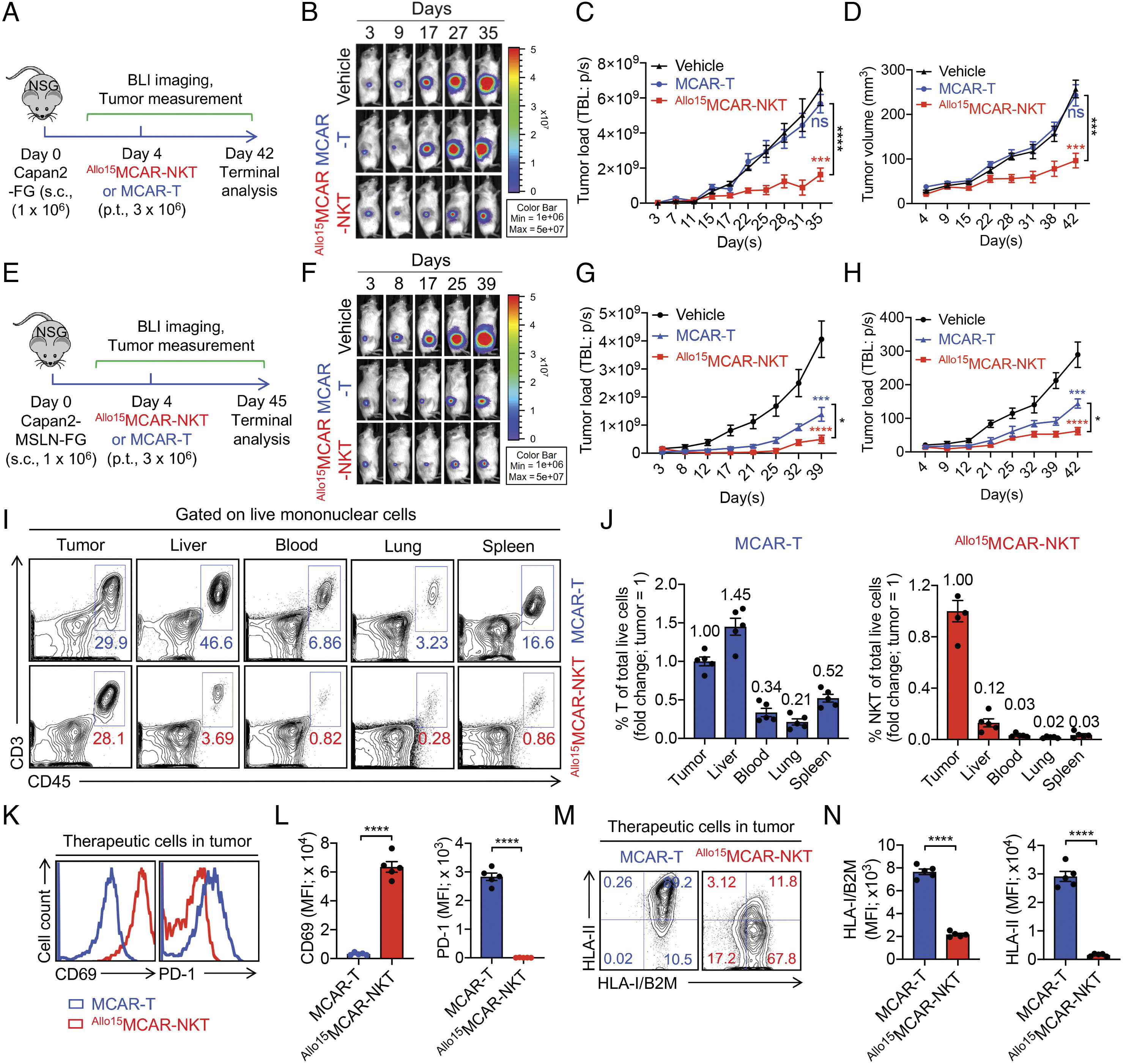
Superior Tumor Penetration and Antitumor Activity
In preclinical pancreatic cancer models, Allo15MCAR-NKT cells showed markedly better performance than traditional CAR-T cells. In an orthotopic model, these engineered NKT cells not only controlled tumors more effectively but also demonstrated a unique ability to migrate deep into the tumor core—where CAR-T cells largely remained confined to the periphery. Inside the tumor, they maintained strong activation (high CD69, IFN-γ, perforin, granzyme B) with lower exhaustion, and preserved low HLA-I/II expression, reducing the risk of immune rejection.
The advantage persisted in metastatic disease. In lung-dominant metastatic models, Allo15MCAR-NKT cells trafficked efficiently to distant sites, achieved greater tumor reduction, and significantly extended survival compared with CAR-T therapy. Even in an inflammatory environment, they avoided exhaustion and continued to function effectively.
Together, these results indicate that Allo15MCAR-NKT cells overcome key barriers that limit CAR-T therapy in solid tumors, offering one of the strongest preclinical signals to date for targeting both primary and metastatic pancreatic cancer.
Antigen-Independent Tumor Killing and Safety Advantages
Allo15MCAR-NKT cells demonstrated meaningful activity even in antigen-negative tumors—a setting where CAR-T cells had no effect. In MSLN-negative subcutaneous models, these NKT cells were able to control tumor growth, remain localized at the tumor site, and sustain strong activation with minimal exhaustion. This highlights a key advantage of the platform: potent, CAR-independent cytotoxicity that can overcome antigen heterogeneity, one of the major challenges in solid tumors.
Beyond efficacy, Allo15MCAR-NKT cells showed a favorable safety and immunogenicity profile. They maintained low HLA expression even after inflammatory stimulation and induced minimal activation of allogeneic immune cells, supporting their potential as a universal, off-the-shelf therapy. Unlike allogeneic CAR-T cells, they did not trigger graft-versus-host disease in mice, caused no weight loss or clinical toxicity, and showed no signs of alloreactivity in vitro—consistent with their CD1d-restricted biology.
In addition, inflammatory toxicity was markedly reduced. In a peritoneal cytokine-release syndrome model, Allo15MCAR-NKT cells produced substantially lower IL-6 and SAA-3 levels, while maintaining normal body weight and preserving efficient antitumor cytokine production. This translated into a significantly lower risk of severe CRS without compromising antitumor activity.
You Can Read All Article Here


