PRINCE trial addresses a critical challenge in metastatic castration-resistant prostate cancer, where immune checkpoint inhibitors have shown limited benefit despite success in other malignancies. Radioligand therapy with [177Lu]Lu-PSMA-617 has demonstrated survival benefit, yet resistance and disease progression remain common. Combining targeted radiation with immunotherapy represents a biologically rational strategy to enhance anti-tumor activity. This study evaluates the safety and clinical activity of [177Lu]Lu-PSMA-617 in combination with pembrolizumab in patients with advanced disease.
Title: [177Lu]Lu-PSMA-617 in combination with pembrolizumab for treatment of metastatic castration resistant prostate cancer (PRINCE): a single-arm, phase 1b/2 study
Authors: Prof Shahneen Sandhu, MBBS, Prof Anthony M Joshua, PhD, Prof Louise Emmett, MD, Mathias Bressel, MS, Angelyn Anton, MBBS, Lavinia Spain, MBBS, Prof Lisa G Horvath, PhD, Anupama Pasam, MS, Sofie H Tolmeijer, PhD, Timothy J Akhurst, MBBS, Ramin Alipour, MD, Patricia Banks, MBBS, James P Buteau, MD, Erin Cassidy, PGCerta, Megan Crumbaker, PhD,
Nattakorn Dhiantravan, MBBS, Wen Xu, MBBS, Joanna Chan, PhD, Nadia Hitchen, MD, Mark Scalzo, BAppS, Aravind S Ravi Kumar, MBBS, Grace Kong, MBBS, Roslyn Wallace, MBBS, Narelle Williams, MPH, Prof Scott Williams, MD, Prof Nicole M Haynes, PhD, Prof Paul Neeson, PhD, Alexander W Wyatt, DPhil, Prof Rodney J Hicks, MBBS, Prof Michael S Hofman, MBBS
Background
Metastatic castration-resistant prostate cancer (mCRPC) remains a challenging disease with limited responsiveness to immune checkpoint inhibitors (ICIs), largely due to its immunologically “cold” tumor microenvironment characterized by low tumor mutational burden and weak T-cell infiltration. Radioligand therapy with [177Lu]Lu-PSMA-617 has demonstrated improvements in progression-free survival and overall survival in this population, establishing itself as a standard therapeutic option.
However, resistance and disease progression remain common. Preclinical evidence suggests that radiotherapy may enhance anti-tumor immunity by inducing immunogenic cell death and promoting antigen release, thereby providing a rationale for combining radioligand therapy with ICIs.
The PRINCE trial was designed to evaluate whether combining [177Lu]Lu-PSMA-617 with pembrolizumab could improve clinical outcomes while maintaining an acceptable safety profile.
Methods
PRINCE was a multicenter, single-arm, phase 1b/2 clinical trial conducted across four Australian centers. Eligible participants were men aged 18 years or older with histologically confirmed mCRPC, ECOG performance status 0–1, and prior treatment with at least one androgen receptor pathway inhibitor, with prior docetaxel permitted. High PSMA expression on imaging was required for enrollment.
Patients received up to six cycles of [177Lu]Lu-PSMA-617 administered intravenously every 6 weeks, starting at 8.5 GBq and decreasing incrementally with each cycle. Pembrolizumab was administered at a fixed dose of 200 mg intravenously every 3 weeks for up to 24 months. Treatment continued until disease progression, unacceptable toxicity, or completion of planned therapy.
Co-primary endpoints included safety and prostate-specific antigen (PSA) response rate defined as a ≥50% decline from baseline. Secondary endpoints included radiographic progression-free survival (rPFS), overall survival (OS), objective response rate (ORR), and patient-reported outcomes.
Study Design
This was a non-randomized, single-arm study with a planned sample size of 37 treated participants, designed to detect common toxicities and provide preliminary estimates of clinical activity. All patients who received at least one dose of therapy were included in the analysis.
Participants were followed with imaging every 12 weeks, including CT scans and bone imaging, and underwent serial biomarker assessments including PSMA PET/CT, circulating tumor DNA, and genomic profiling. Safety was assessed using CTCAE version 5.0, and survival outcomes were analyzed using Kaplan–Meier methods.
Mechanism of Action: How the Combination Works
The therapeutic rationale of the PRINCE trial is based on the complementary mechanisms of radioligand therapy and immune checkpoint inhibition.
[177Lu]Lu-PSMA-617 is a PSMA-targeted radioligand that delivers β-particle radiation directly to tumor cells, causing localized DNA damage and cell death with a short penetration range of 1–3 mm.
In addition to its cytotoxic effect, radioligand therapy can induce immunogenic cell death, leading to tumor antigen release and activation of immune pathways such as STING, which may enhance T-cell priming.
Pembrolizumab, an anti–PD-1 antibody, restores T-cell–mediated immunity by blocking PD-1 signaling.
Together, this combination aims to couple direct tumor killing with immune activation, potentially improving anti-tumor responses
Results
Between August 2019 and December 2020, 37 patients were treated, with a median age of 72 years. Most patients had advanced disease, with 95% having bone metastases and 73% previously treated with docetaxel.
Patients received a median of six cycles of [177Lu]Lu-PSMA-617 and 12 cycles of pembrolizumab, with a median follow-up of 30 months.
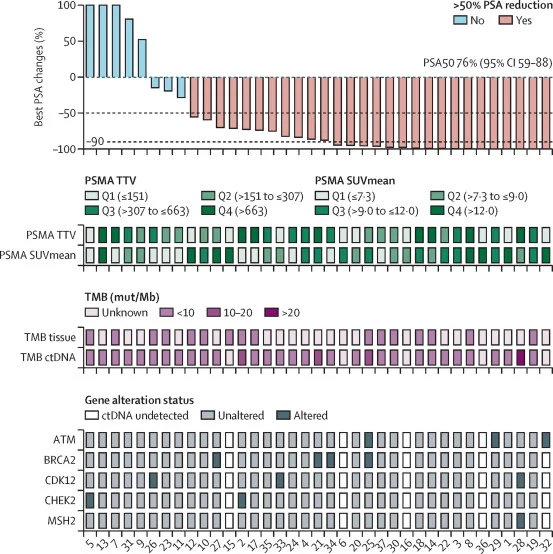
The study demonstrated strong anti-tumor activity. A PSA decline of at least 50% (PSA50 response) was observed in 76% of patients, while 46% achieved a PSA decline of at least 90%. Among patients with measurable disease, the objective response rate was 70%, with the majority achieving partial responses.
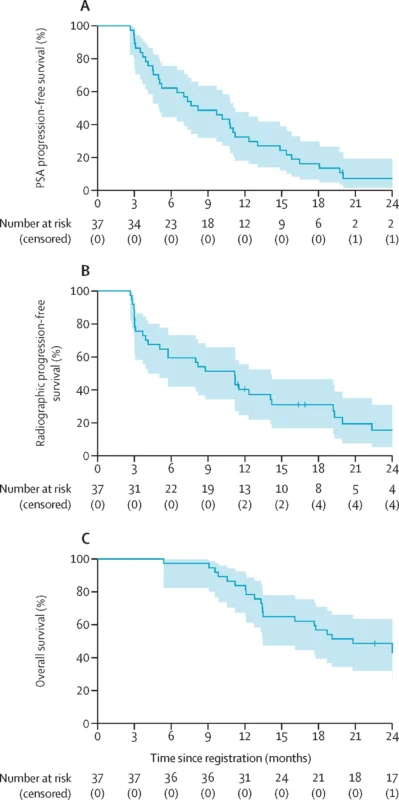
Median PSA progression-free survival was 8.2 months, while median radiographic progression-free survival reached 11.2 months. Median overall survival was 20.8 months. At 12 months, overall survival was 84%, decreasing to 49% at 24 months. Radiographic progression-free survival rates were 40% at 12 months and 16% at 24 months.
Key Findings
The combination of [177Lu]Lu-PSMA-617 and pembrolizumab resulted in high PSA response rates and meaningful clinical activity in a heavily pretreated mCRPC population. Deep responses were observed across subgroups, including patients with low tumor mutational burden, suggesting that the benefit was not solely dependent on traditional immunotherapy biomarkers.
Treatment was generally well tolerated. Most adverse events were grade 1–2, with common toxicities including xerostomia (78%), fatigue (46%), and nausea (27%). Grade 3 immune-related adverse events occurred in 30% of patients, but there were no grade 4 toxicities or treatment-related deaths.
Importantly, quality-of-life measures and pain scores remained stable over time, supporting the tolerability of this combination approach.
Conclusion
The PRINCE trial demonstrates that multicycle [177Lu]Lu-PSMA-617 combined with pembrolizumab is associated with high response rates, manageable toxicity, and promising survival outcomes in patients with mCRPC. While the single-arm design limits definitive conclusions regarding the contribution of pembrolizumab, the results support continued exploration of radioligand–immunotherapy combinations.
Future studies should focus on randomized comparisons and identification of predictive biomarkers to define which patients derive the greatest benefit from this approach. As radioligand therapy continues to expand across oncology, combination strategies such as this may play an important role in overcoming resistance and improving outcomes in advanced cancers.


