Bacillus Calmette-Guérin (BCG) has long been the standard treatment for high-risk non–muscle-invasive bladder cancer (NMIBC), providing durable disease control when delivered with appropriate induction and maintenance. The article “BCG and Beyond,” published in the Journal of Clinical Oncology in March 2026, reviews evidence from the phase III CREST, POTOMAC, and ALBAN trials, which evaluated the addition of systemic PD-1/PD-L1 inhibitors to intravesical BCG in BCG-naïve NMIBC. The analysis highlights modest improvements in recurrence-based outcomes while emphasizing the need to balance potential benefits with increased toxicity and treatment burden.
Title: Bacillus Calmette-Guérin (BCG) and Beyond: Is Systemic Immunotherapy for BCG-Naïve Non–Muscle-Invasive Bladder Cancer Progress or Overreach?
Authors: Ashish M. Kamat, MD, MBBS , Patrick J. Hensley, MD, Brigida A. Maiorano, MD, PhD, MSc , Roger Li, MD, Sarah P. Psutka, MD, MS, Kent W. Mouw, MD, PhD , Amir Horowitz, PhD, Shilpa Gupta, MDX, Andrea Necchi, MD
Published in JCO Oncology, March 2026
Background
Intravesical Bacillus Calmette-Guérin (BCG) has been the cornerstone of treatment for high-risk non–muscle-invasive bladder cancer (NMIBC) for decades. When delivered appropriately following complete transurethral resection and maintained according to established protocols such as SWOG 8507, BCG produces durable disease control in a large proportion of patients. Contemporary clinical trials report 2-year event-free survival (EFS) rates of approximately 80%–83% with optimized BCG therapy.
The success of immune checkpoint inhibitors (ICIs) in metastatic and perioperative urothelial carcinoma has encouraged investigation of systemic immunotherapy in earlier disease stages, including BCG-naïve NMIBC. Three recent phase III trials—CREST (sasanlimab + BCG), POTOMAC (durvalumab + BCG), and ALBAN (atezolizumab + BCG)—tested whether combining systemic checkpoint inhibition with intravesical BCG could further reduce recurrence and disease progression.
While some studies demonstrated statistically significant improvements in event-free survival or disease-free survival, the magnitude of benefit remains modest, and concerns have emerged regarding toxicity, cost, and the clinical relevance of surrogate endpoints in this setting.
Methods
The authors conducted a comparative analysis of outcomes from the CREST, POTOMAC, and ALBAN phase III trials, which evaluated systemic immunotherapy combined with intravesical BCG in BCG-naïve patients with high-risk NMIBC. Because individual patient data were not publicly available, the investigators reconstructed pseudo individual patient data (IPD) from published Kaplan–Meier survival curves using a validated method described by Guyot and colleagues.
Kaplan–Meier curves were digitized using WebPlotDigitizer (version 4.0) to extract time-to-event survival coordinates. These were combined with reported numbers at risk and total event counts to estimate individual event and censoring times. The reconstructed datasets were then analyzed using Cox proportional hazards models to compare outcomes between trial arms.
This approach allowed the investigators to examine patterns of event-free survival (EFS) and disease-free survival (DFS) across trials, providing a framework to interpret statistically significant results within a broader clinical context.
Study Design
The analysis focused on three randomized phase III trials evaluating systemic PD-1 or PD-L1 inhibition combined with intravesical BCG in patients with BCG-naïve high-risk NMIBC:
- CREST trial: sasanlimab + BCG versus BCG alone
- POTOMAC trial: durvalumab + BCG versus BCG alone
- ALBAN trial: atezolizumab + BCG versus BCG alone
These studies evaluated composite endpoints such as event-free survival or disease-free survival, typically defined as time from randomization to:
- recurrence of high-risk NMIBC
- persistence of carcinoma in situ (CIS)
- progression to muscle-invasive disease or metastatic disease
- death without recurrence.
Across these trials, EFS improvements were modest, with hazard ratios around 0.68 in trials demonstrating statistical significance. However, follow-up durations, endpoint definitions, and BCG maintenance schedules differed between studies, complicating cross-trial comparisons.
Results
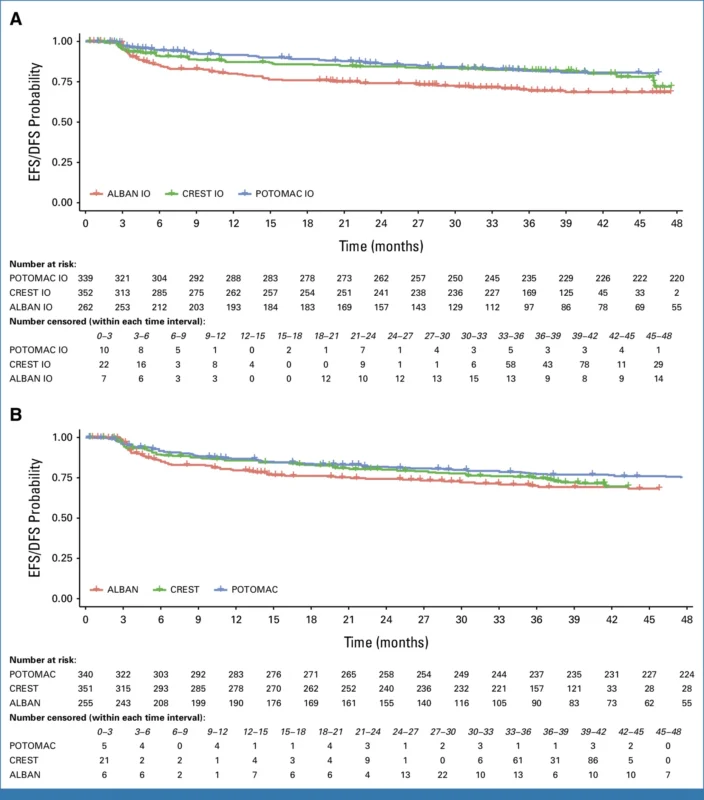
Reconstructed IPD analyses revealed that survival curves across the experimental arms of the trials were broadly similar, though differences between studies were evident.
Pairwise Cox analyses demonstrated:
- ALBAN vs POTOMAC: HR 1.84 (95% CI 1.32–2.58; P < .001)
- ALBAN vs CREST: HR 1.61 (95% CI 1.15–2.27; P = .006)
- POTOMAC vs CREST: no statistically significant difference (P = .45)
These results indicate that outcomes with atezolizumab in the ALBAN trial appeared inferior to those observed with durvalumab or sasanlimab in the other trials.
Comparisons of BCG-only control arms also demonstrated heterogeneity between studies.
For example:
- POTOMAC vs ALBAN: HR 0.62 (95% CI 0.42–0.93; P = .022)
- No statistically significant differences were seen between CREST and ALBAN or POTOMAC and CREST.
These findings suggest that differences in BCG delivery, maintenance schedules, or treatment protocols may partially explain variations in outcomes across trials.
Absolute clinical benefit
Although CREST and POTOMAC showed statistically significant improvements in EFS or DFS, the absolute benefit was limited, generally in the range of 5%–7% improvement in EFS.
Importantly, these benefits were largely driven by recurrence events, rather than meaningful changes in progression or survival.
Rates of progression to muscle-invasive bladder cancer (MIBC) or metastatic disease remained low in both treatment arms across trials.
For example:
- In CREST, only 24 patients (3.4%) experienced disease progression.
- In ALBAN, progression occurred in three patients total (two in the BCG arm and one in the atezolizumab + BCG arm).
No improvements were observed in:
- overall survival
- progression events
- cystectomy-free survival
These endpoints are clinically important but are difficult to power statistically in NMIBC due to the relatively long natural history of the disease.
Toxicity
The addition of systemic immunotherapy was associated with substantially higher toxicity rates.
Serious adverse events (SAEs) were consistently higher in the ICI + BCG arms compared with BCG alone.Examples include:
POTOMAC trial
- SAEs: 32% with durvalumab + BCG vs 19% with BCG alone
- Treatment-related SAEs: 13% vs 4%
ALBAN trial
- SAEs: 38% vs 21%
- Treatment-related SAEs: 24% vs 8%
CREST trial
- Treatment-related SAEs: 18% with sasanlimab + BCG vs 1% with BCG alone
Many immune-related toxicities, including endocrinopathies, may persist long-term and require lifelong treatment.
Key Findings
- This analysis highlights that systemic immunotherapy added to BCG provides only modest improvements in EFS or DFS in BCG-naïve high-risk NMIBC.
- When outcomes from multiple trials are examined collectively, the magnitude of benefit appears limited, especially considering that optimized BCG therapy already produces strong disease control.
- Most treatment effects were driven by recurrence events, which are often manageable with additional intravesical therapies.
- Critically, progression to muscle-invasive disease, metastasis, or death was rare in both treatment groups.
- The analysis also underscores the heterogeneity in outcomes across BCG control arms, suggesting that differences in BCG administration and maintenance protocols may significantly influence trial results.
Conclusion
The expansion of immunotherapy into earlier disease stages represents an important direction in urothelial cancer research. However, the available data from CREST, POTOMAC, and ALBAN suggest that systemic checkpoint inhibition should not yet be adopted routinely in BCG-naïve high-risk NMIBC. Optimized BCG therapy continues to produce strong outcomes, and the modest improvements in recurrence-based endpoints achieved with systemic therapy come at the cost of increased toxicity and financial burden.
Future progress in NMIBC will depend on more precise patient selection, improved biomarkers of progression risk, and endpoints that better reflect meaningful clinical outcomes, including bladder preservation and patient quality of life. Rather than broadly expanding systemic therapy, the focus should shift toward identifying patients at the highest risk of progression, such as those with T1 high-grade tumors with adverse features, who may derive the greatest benefit from systemic intensification.


