New Phase 2 Trial Published in The Lancet Oncology Shows Promising Results for Muscle-Invasive Bladder Cancer
The phase 2 SunRISe-4 Trial, recently published in The Lancet Oncology, offers encouraging findings for patients with muscle-invasive bladder cancer (MIBC) who are unable or unwilling to receive standard cisplatin-based chemotherapy before surgery.
SunRISe-4 is a randomized, open-label phase 2 trial conducted in 109 centers across 10 countries. Eligible MIBC patients (cT2–cT4 N0M0), unsuitable or unwilling for neoadjuvant cisplatin, were randomized 5:3 to receive four cycles of either TAR-200 plus cetrelimab or cetrelimab alone every 21 days before radical cystectomy. The primary endpoint was pathological complete response (pCR) assessed centrally.
Title: TAR-200 plus cetrelimab versus cetrelimab monotherapy as neoadjuvant therapy in patients with muscle-invasive bladder cancer who are ineligible for or decline neoadjuvant cisplatin-based chemotherapy (SunRISe-4): interim analysis of a randomised, open-label phase 2 trial
Authors: Andrea Necchi, Félix Guerrero-Ramos, Paul L Crispen, Bernardo Herrera-Imbroda, Rohan Garje, Thomas Powles, Charles C Peyton, Benjamin Pradere, Ja Hyeon Ku, Neal Shore, Martin Bögemann, Mark A Preston, Evanguelos Xylinas, Cristina Sanchez de Llano, Mohamad Hasan, Hind Stitou, Sumeet Bhanvadia, Hussein Sweiti, Sarah P Psutka

Results:
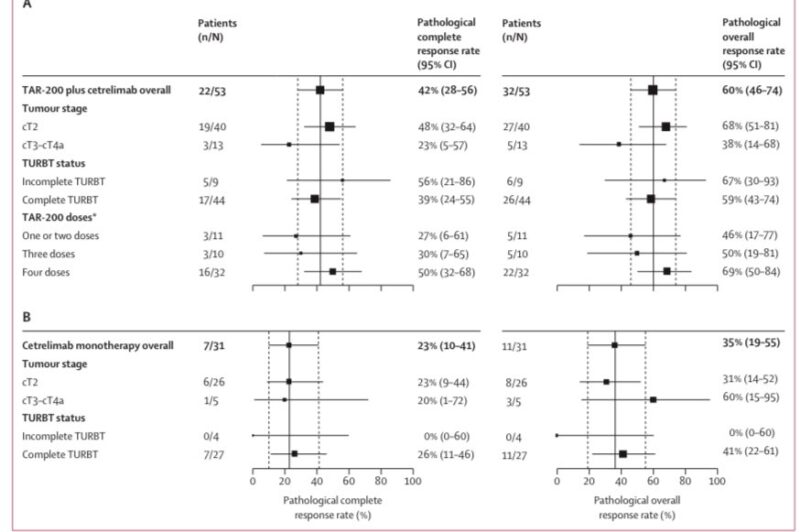
- 122 patients randomized—80 to combination therapy and 42 to monotherapy.
- The efficacy-evaluable set (patients undergoing cystectomy or with disease progression/death) included 53 in combination and 31 in monotherapy groups.
- Median follow-up of 23.5 weeks showed pCR rates of 42% with TAR-200 plus cetrelimab versus 23% with cetrelimab alone.
- Safety: Treatment-related adverse events were more common in the combination group (72% vs 44%), with grade 3 or higher events in 11% versus 5%. Serious adverse events occurred in 11% of combination and 2% of monotherapy patients. No treatment-related deaths occurred in the combination group.
Oncology Community Reacted to SunRISe-4 Trial results:
Andrea Necchi, Co-Author of SunRISe-4 Trial:
“Interim results of Sunrise4 are released The Lancet Oncology
Neoadjuvant TAR200 + cetrelimab vs cetrelimab monotherapy in patients with MIBC
TAR200+CET: pCR 42%, Major path Response 60%
in cT2 pts baseline: pCR
48% TAR200 exposure-response association: pCR 50% if TAR200x4 full cycles
Can we cure selected patients with MIBC with intravesical Rx + single agent IO?
Future: is there a way to avoid radical cystectomy by consolidating Response with TAR200 in selected patients?
Primary results with full biomarker results are awaited.”
Félix Guerrero-Ramos, Co-Author of SunRISe-4 Trial:
“Interim results of SR-4 study published in The Lancet Oncology
pCR rate 42%
Download it here.
Glad to co-author this paper with Andrea Necchi, Bernardo Herrera Imbroda, Tom Powles, Benjamin Pradere among others!”

Congrats Andrea Necchi, Sarah P Psutka, Félix Guerrero-Ramos and entire SunRise-4 team on the very promising results! Exciting to see promising options for cisplatin-ineligible MIBC patients.”
“Yes, that is the way for MIBC! Intravesical therapy + systemic therapy to avoid the radical Surgery!!”
“Applaud to the study lots of learn from here but this is picked cohort “ 97 (81%) patients had cT2 disease and 98 (82%) patients had visibly complete transurethral resection of bladder tumour at screening””
“Landmark study demonstrating the huge potential of the TAR device!! Congratulations to Andrea Necchi and the whole team behind this important effort!
Really excited to see which drugs will be formulated in the pretzel.”
“Cisplatin-ineligible MIBC: no more exclusion from neoadjuvant therapy!
SunRISe-4 (TAR-200+cetrelimab, 42% pCR) and EV-303 (EV+pembro) bring real promise for this population.”
“Exciting Results.”
“A new SunRISe for cisplatin-ineligible MIBC.
SunRISe-4 | Lancet Oncol 2025 Phase 2 RCT in cisplatin-ineligible/declining, cT2–T4a N0–1M0 pts (median age 69, ~75% male).
TAR-200 (intravesical gemcitabine) + cetrelimab (PD-1 inhibitor) vs cetrelimab
Outcomes
• pCR: 42% vs 23%
• ≤ypT1: 60% vs 35%
Safety
• Grade ≥3 TRAEs: 11% vs 5%
• Tx-related deaths: 0 vs 1
Device-based local chemo + systemic PD-1 = promising neoadjuvant path when cisplatin is off the table.”

More posts featuring Bladder Cancer on OncoDaily.


