George Kumar, Senior Director at AstraZeneca, shared a post on LinkedIn about a recently published article by Robert L. Ferris et al. in BMJ:
“One-Time Autologous TIL Therapy in patients with Recurrent/Metastatic HNSCC
What is Autologous TIL Therapy?
Autologous tumor-infiltrating lymphocyte (TIL) therapy is a personalized form of immunotherapy that harnesses a patient’s own immune system to fight cancer. TILs are T-cells that naturally migrate into tumors and recognize cancer-specific antigens. In this approach, tumor tissue is surgically resected, TILs are isolated and expanded to billions of cells in the laboratory, and patients undergo lymphodepleting chemotherapy before receiving a one-time infusion of their own expanded TILs. Interleukin-2 (IL-2) is administered afterwards to support the infused cells. Unlike ‘off-the-shelf’ therapies, this is highly individualized.
Results
- Responses: ORR was 11% (6/53, all partial responses).
- Durability: Median DOR was 7.6 months; median follow-up 17.9 months.
- Disease Control: DCR was 76% (40/53), with 64% showing stable disease.
- Safety: Toxicities were consistent with expected effects of lymphodepletion and IL-2; surgical resection carried typical risks of anesthesia and tumor excision.
- Manufacturing: All TIL products met release criteria, showing feasibility of consistent TIL generation from HNSCC tumors.
Conclusions
- Modest activity was observed in heavily pretreated HNSCC patients, with limited objective responses.
- Outcomes do not support further development of these TIL products as monotherapy in patients progressing after prior immunotherapy.
- Nonetheless, the study demonstrated feasibility and safety of TIL manufacturing and infusion.
- Findings support further development of next-generation TIL strategies, such as:
- Combination approaches with checkpoint inhibitors or other agents
- Novel TIL products with improved tumor reactivity
- Optimized manufacturing processes to enhance potency and durability
In essence:
Autologous TIL therapy is feasible and safe in HNSCC, but shows only modest standalone efficacy in patients who progressed after immunotherapy.
The future likely lies in smarter TIL designs or combination strategies rather than monotherapy.”
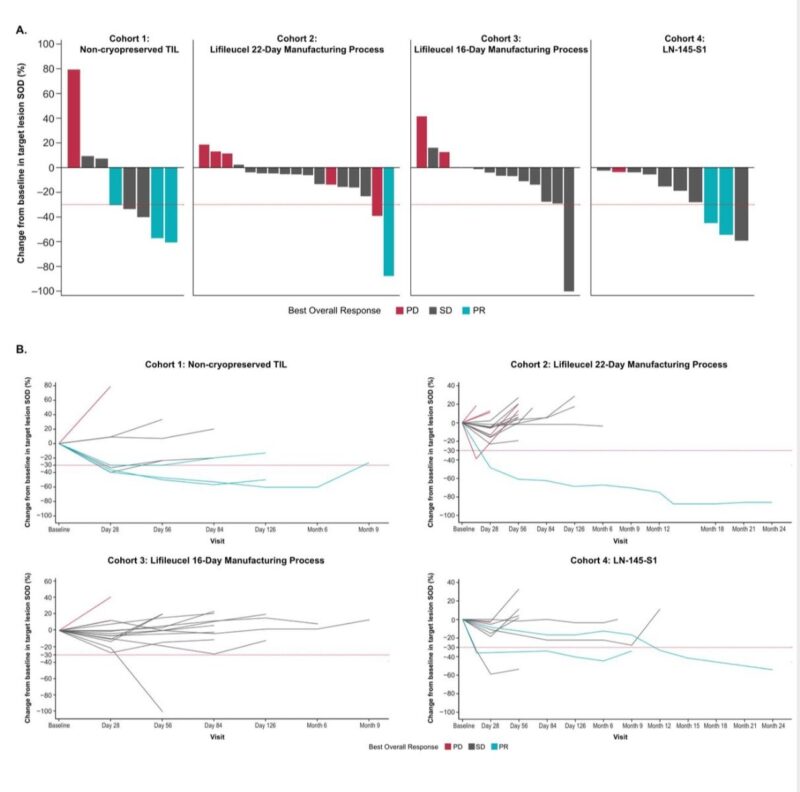
Figure Courtesy: Ferris RL, et al, Journal for ImmunoTherapy of Cancer. 2025. Best percent change from baseline in target lesion sum of diameters by cohort. (A) Percent change from baseline in target lesion sum of diameters for all patients who received TIL. (B) Percent change from baseline (ie, after tumor resection and before lymphodepletion) over time in target lesion sum of diameters for all patients who received TIL.
Title: Efficacy and safety of one-time autologous tumor-infiltrating lymphocyte cell therapy in patients with recurrent and/or metastatic head and neck squamous cell carcinoma
Journal: BMJ
Authors: Robert L. Ferris, Rom S. Leidner, Christine H. Chung, Antonio Jimeno, Sylvia M. Lee, Ammar Sukari, Jorge J. Nieva, Juneko E. Grilley-Olson, Rebecca Redman, Stuart J. Wong, Victoria M. Villaflor, Jamal Misleh, Friedrich Graf Finckenstein, Jeffrey Chou, Brian Gastman, Rana Fiaz, Melissa Catlett, Min Yi, Ezra E. W. Cohen

More posts featuring George Kumar on OncoDaily.


