Rahul Gosain, Co-Founder of Oncology Brothers podcast and Director of Regional Infusion Services and Medical Director at Wilmot Cancer Institute, recently shared on LinkedIn:
“Trastuzumab Deruxtecan now FDA approved for pan-tumor with HER2 IHC3 and solid tumors who have received prior systemic treatment and have no alternative treatment options.
Based on DESTINY-PanTumor02, DESTINY-Lung01, and DESTINY-CRC02:
- Dose 5.4 mg/kg
- Common AE: fatigue, decreased counts and nausea. ILD can be fatal.”
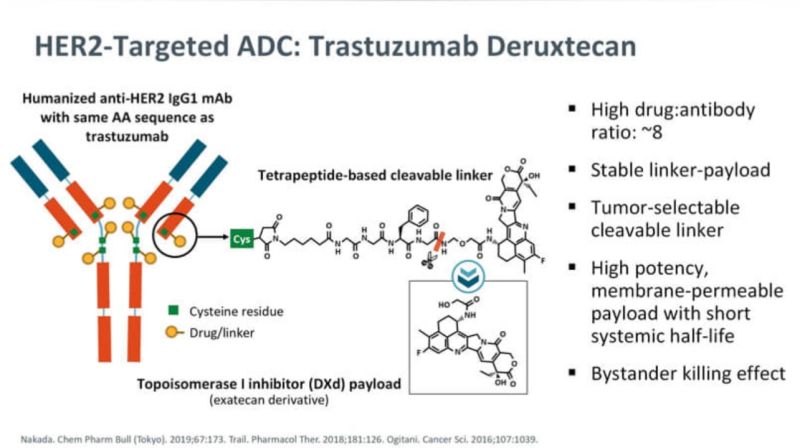
Source: Rahul Gosain/LinkedIn


