This week in oncology biotech, a $5 billion ADC acquisition out of Munich captured the sector’s attention, but it was far from the only signal worth tracking. From China regulatory approvals and bispecific franchise expansions to emerging science at the microbiome-oncology interface, the week offered a clear view of where capital and conviction are converging.
Below are 10 featured posts from scientists, investors, and executives shaping the conversation in oncology and life sciences this week.
Milad Alucozai – Co-Founder & General Partner, Pamir Ventures
European biotech thinks too small.” “Academic spinouts don’t work.” Six years later: $5B acquisition. This is what the Tubulis GmbH and Gilead Sciences deal should mean for the startup ecosystem.
Founded by scientists. No hype raise. No SPAC. No shortcuts. The team kept building. Deep science, a defensible platform, and clinical data that proved itself.
Tubulis spun out of a German research lab in 2019. They built next generation antibody drug conjugates. A technology that delivers cancer treatment directly into tumors while reducing toxicity.
They raised $600M privately. Gilead partnered with them in 2024. Today Gilead acquired them for $3.15B upfront with another $1.85B in milestones.
This is the part most people miss about biotech and deep tech. The timelines are longer. The risk is real. But when the science works, the outcomes are not 2x or 3x. They are generational.
University spinout to $5B acquisition in six years. While everyone was chasing the next SaaS tool, these founders were engineering molecules.
This is why I get excited about backing technical founders solving hard problems.
The best returns in venture are not built on hype. They are built on depth.
Levi Garraway – Executive Vice President, Head of Global Product Development and Chief Medical Officer, Genetech
Fifty years ago, Genentech’s founders started a company that launched the entire biotech industry. Celebrating our 50th Anniversary and Founders Day this week reminded me that our success is rooted in a unique culture,one that blends rigorous, world-class science with creativity, irreverence and joy.
Whether we’re pursuing intense work meant to shape the future of oncology or in full costume celebrating with colleagues, that pioneering spirit shines bright. For 50 years, our company has been fueled by the same relentless curiosity and refusal to settle for the status quo.
Reflecting on this milestone, I feel deeply proud of the remarkable things we’ve accomplished and galvanized by the conviction that we’re just getting started. At the same time, I’m humbled by the expectation that our culture of bold innovation will continue to transform promising science into life-changing medicines for decades to come.
Here’s to the next chapter.

Levi Garraway/LinkedIn

Levi Garraway/LinkedIn
Misagh Naderi – CEO & Co-Founder, Glint Lab
Investors keep funding oncology. Biology is still the bottleneck.
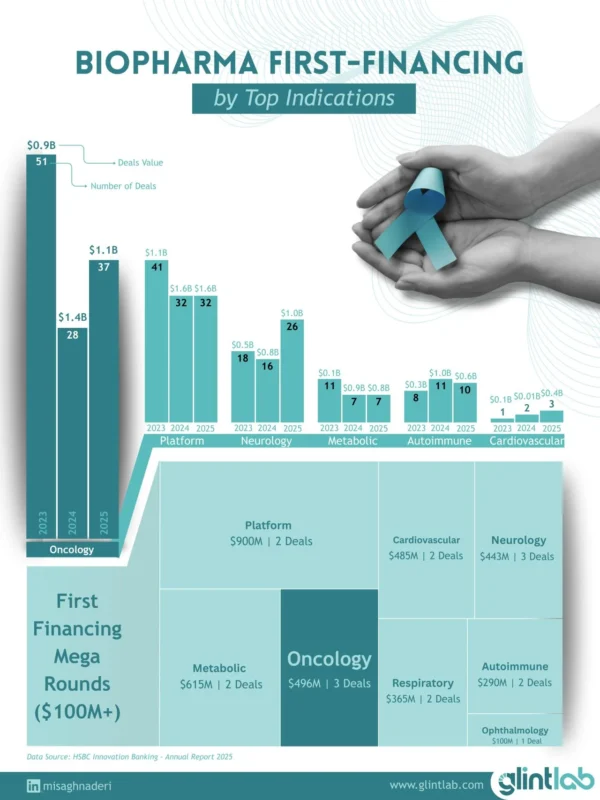
Early biotech financing data from 2023–2025 shows a familiar pattern. Oncology still dominates new company creation.In 2025 alone, 37 oncology startups raised about $1.1B in first financings across the US and Europe. Even with fluctuations in capital flows, oncology continues to produce the largest number of new therapeutic companies entering the pipeline.
But the more interesting point is where these companies actually are.
Most oncology first financings remain preclinical. In 2025, 27 deals were focused on preclinical assets. That tells us something important about the structure of oncology R&D.The bottleneck is rarely capital; it is the biological understanding early in the development.
Even the largest financings reflect this uncertainty. Among $100M+ mega rounds of early-stage funding (angel, pre-seed, seed, and Series A) in 2025, oncology accounted for three deals totaling about $496M, spanning both preclinical and early clinical programs.
As more oncology startups enter the pipeline, the quality of tumor characterization and tissue-level evidence becomes increasingly important. Better biology upstream leads to better decisions downstream.
Misagh Naderi/LinkedIn
Uli Stilz – Senior Advisor, Flagship Pioneering
Moving upstream in biotech innovation feels less like a change,and more like a continuation of a journey.
What excites me most is the opportunity to start earlier,at the intersection of emerging biology, new modalities, and increasingly AI,and to shape companies that are designed to scale from the outset.
The next wave of impact will depend not only on scientific breakthroughs, but on how we build the systems around them,combining structured experimentation and optionality in early discovery with the focus, execution, and partnering strategies needed to translate science into therapies.
At Flagship Pioneering, this is where company creation begins.
Thanks to Boston Business Journal for the feature.
More to come.
Alan Vanderborght – CEO, Kybora
CNS is where oncology was 20 years ago. The microbiome may be what connects them both.
I recently sat down with Nicolas Coudurier, CEO of Biocodex, for our KYBORA Healthcare Insights podcast.
Biocodex is a €676M family-owned pharma company that has been in the microbiome space for 7 decades.
Nicolas introduced a term worth knowing: microbiomecology.
The idea: microbiome solutions that don’t replace oncology treatments, but enhance them. Specifically, increasing patient responsiveness when they’re on immune checkpoint inhibitors.
Studies are already running at Institut Gustave Roussy in Paris and at hospitals in the US. Biocodex isn’t trying to become an oncology company. They’re exploring how the microbiota can synergize with existing treatments. If that works, it changes the treatment algorithm.
Then there’s CNS.
No single product will solve complex neurological conditions. We’ve done work with companies in the ALS space, and the reality is clear: it will take a combination of approaches.
The gut-brain axis is no longer theoretical. It’s generating real clinical data. Physicians are becoming more comfortable treating each patient with cocktails of approaches rather than single-product solutions.
And then women’s health. Alan called it a space with “a drought of innovation” but potentially fast-growing. Nicolas explained the connection: we don’t have only a gut microbiota. We have a vaginal microbiota, and the science converges. Biocodex already leads in intimate health in France and Europe with their Saforelle brand.
As Nicolas put it: “The holistic approach is more and more necessary. What better than microbiota can prove that, being an organ by itself with its action on all the other organs.”
What makes this moment different from the hype cycle:
- AI is now advanced enough to map the microbiome’s complexity. Biocodex has backed a Paris-based startup doing exactly that for 7 years.
- The science is moving from consumer probiotics toward mechanism-driven therapeutic applications.
- Companies like Biocodex and MaaT Pharma are building the evidence base that was missing during the initial wave.
- Biocodex runs a Microbiota Institute in 9 languages providing non-promotional scientific education. They’re investing in credibility, not just products.
I see real partnering opportunities emerging here. Companies with deep microbiome expertise partnering with larger organizations that lead in oncology or CNS could create significant value on both sides.
For biotech leaders in this space:
- The microbiome is shifting from standalone category to adjuvant platform. Oncology, CNS, women’s health. That changes who your partnering targets are.
- Companies with decades of microbiome data have a compounding advantage. Clinical history and manufacturing scale are hard to replicate.
- Oncology & CNS are both moving toward combination approaches. If your platform can synergize with existing therapies, that’s where the conversations are heading.
Markus Zettl – Head of Discovery – Therapeutic Lead Immuno-Oncology & Cell and Gene Therapies, Biological E. Limited
In reposting Biological E. Limited original announcement, Markus Zettl wrote:
I’m happy to share that I’m starting a new position as Head of Discovery – Therapeutic Lead Immuno-Oncology & Cell & Gene Therapies at Biological E. Limited!
In the original company post, Biological E. Limited wrote:
We’re pleased to welcome Markus Zettl to Biological E Limited as Head of Discovery – Therapeutic Lead, Immuno-Oncology & Cell and Gene Therapies.
With over 15 years of global experience in cancer immunotherapy, he brings deep expertise across biologics, mRNA-based therapeutics, and early-stage development.
As we continue to strengthen our capabilities in vaccines and biologics, his addition will support the advancement of our innovation pipeline in emerging therapies.
We look forward to the journey ahead.

Markus Zettl/Biological E. Limited
Jack Shuang Hou – Scientific Director, Jtests
Amgen’s Tarlatamab Wins China Approval in ES-SCLC, Expanding >$2B Bispecific Opportunity with BeOne Medicines
A major global oncology expansion milestone just landed.
China’s NMPA approved Amgen’s tarlatamab (Imdelltra) for previously treated extensive-stage small cell lung cancer (ES-SCLC), with regional development and commercialization led by BeOne Medicines.
Key leader referenced:
Jay Bradner, M.D. , Executive Vice President, Research & Development, AmgenChina unlocks major lung cancer market access
This is highly meaningful in one of the world’s largest lung cancer markets.
The approval targets:Post-chemotherapy ES-SCLC
High unmet need setting
Large incident patient population in China
This materially strengthens global commercial reach beyond the 2024 FDA approval.Bispecific T-cell engagers keep building momentum
Tarlatamab’s DLL3 × CD3 mechanism continues validating:
Precise T-cell redirection
Tumor-selective immune activation
Bispecific antibodies as next-wave oncology backbone assets
This remains one of the clearest DLL3 platform wins in solid tumors.Commercial upside remains underappreciated
Pricing and launch timing remain undisclosed, but the setup is compelling:
Analysts project over $2B in annual global sales potential
Partnership-led China execution de-risks market entry
Strong DeLLphi-301 efficacy supports uptake confidence
The regional partnership strategy here is highly efficient.Takeaway
The bigger signal is not just China approval.This reinforces that bispecific T-cell engagers are becoming globally scalable commercial franchises, not just U.S.-centric specialty launches.
China is now increasingly central to that equation.
Bottom line
Amgen and BeOne Medicines have expanded one of oncology’s most promising bispecific assets into a critical global growth market.

Jack Shuang Hou/LinkedIn
Devang Bhuva – Senior Vice President, Finance at Gilead Sciences
Proud moment for our Corporate Development team at Gilead , what a run in the last quarter!
In a short period of time, we’ve announced three important acquisitions , Arcellx, Ouro Medicines in partnership with Galapagos, and now Tubulis GmbH , each strengthening our pipeline and expanding our ability to deliver meaningful innovation for patients.
It’s been a true privilege to work alongside this incredibly talented group. Grateful for each and every member of the team for the commitment and excellence you bring every day , and excited about what these steps mean for Gilead’s future.
Thank you to everyone involved.
Melissa Leich, Founder & Principal Advisor, Strategic Advisory
Gilead Sciences just paid up to $5 billion for Tubulis GmbH.
A Munich-based, clinical-stage biotech. Private. German.
Let that sink in.
The upfront alone, $3.15 billion, for a company still in Phase 1b/2. Not for revenue. Not for a commercial product. For the science. For the platform. For the vision of what antibody-drug conjugates can do for cancer patients.
This is what it looks like when the work is right.
For everyone building in this space right now, the founders, the scientists, the teams running trials and refining platforms in relative obscurity, this deal is for you. It is proof that the market sees what you are building. That the conviction is justified.
For those watching the European biotech space: the exits are real, the valuations are serious, and US strategic appetite for German-origin science has not slowed.
The future of oncology is being written in places like Munich. And the world is paying attention.
That work starts long before anyone from Foster City comes calling.
But when it does, it can look like this.
Melissa Leich/LinkedIn
“Mgoes” – Podcast Host, Accelerate Bio
biotech has got insane velocity in 2026
here’s what just crossed the line from mice to humans:
– Life Biosciences enrolled first humans in epigenetic reprogramming trial – Sinclair’s life work, in people, now
– Azalea (Doudna’s lab) first human success with in vivo CAR-T, the engineering happens inside your body, no extraction required
– baby KJ, first personalized in-body CRISPR edit ever, 7 months from diagnosis to functional cure
– Kyverna’s CAR-T put lupus into drug-free remission, filing for first ever autoimmune CAR-T approval
– pig kidneys with 69 gene edits kept a man off dialysis 271 days, FDA cleared 30-patient trial
– Retro Bio (Sam Altman-backed) dosed first human with RTR242, targets Alzheimer’s and aging at the cellular root
– GLP-1s confirmed as first true longevity drugs in humans, reduces mortality, protects the brain, slows kidney aging, all in the same molecule
– Insilico’s AI-designed drug hit Phase II for fibrosis with positive efficacy, first AI-discovered molecule to work in humans
– Compass crushed Phase III for psilocybin in treatment-resistant depression, 50 years of regulatory hell, done
– rapamycin PEARL trial showed measurable healthspan improvements, the longevity drug finally has human data
slowly, then sudden
The pace of transformation in oncology biotech shows no signs of slowing. The deals, approvals, and scientific milestones from this week are a reminder that the sector’s most consequential outcomes are built long before they make headlines.
Read more biotech insights on OncoDaily Biotech.


