Sotorasib (brand name: Lumakras) is an innovative targeted therapy that has changed the landscape of cancer treatment for patients with the KRAS G12C mutation. Approved by the FDA in May 2021, it provides a new treatment option for previously treated non-small cell lung cancer (NSCLC) and, as of January 2025, metastatic colorectal cancer (mCRC) in combination with panitumumab. This article explores how Sotorasib works, its efficacy, side effects, and what patients can expect during treatment.
What Is Sotorasib and How Does It Work?
Sotorasib is the first FDA-approved KRAS G12C inhibitor. KRAS is a crucial protein that regulates cell growth, but mutations in the KRAS gene can cause uncontrolled cancer cell proliferation. The KRAS G12C mutation keeps the protein in an always-active state, promoting tumor growth.
Lumakras specifically targets and irreversibly binds to the KRAS G12C mutant protein in its inactive GDP-bound state. By blocking KRAS activation, it prevents downstream cancer-driving signaling pathways, such as MAPK and PI3K, ultimately inhibiting tumor growth and inducing cancer cell death.
What Cancers Does Sotorasib Treat?
Sotorasib is a medication that has been approved by the FDA to treat specific types of cancer caused by a mutation in the KRAS gene, known as KRAS G12C. It is primarily used for two types of cancer:
- Non-Small Cell Lung Cancer (NSCLC): Lumakras is approved for patients who have KRAS G12C-mutated NSCLC, which is a type of lung cancer that has spread or cannot be surgically removed (locally advanced or metastatic). It is specifically for patients who have already tried at least one prior systemic treatment. This means it is used when other treatments have not worked or have stopped working.
- Metastatic Colorectal Cancer (mCRC): In January 2025, the FDA approved Sotorasib for use in combination with another medication called panitumumab for patients with KRAS G12C-mutated mCRC. This is for patients who have already received chemotherapy and whose cancer has continued to progress.
Lumakras works by targeting the KRAS G12C mutation, which is found in some cancers, and helping to slow down or stop the growth of the cancer cells. If you have one of these conditions and are dealing with a KRAS G12C mutation, Sotorasib may be a treatment option to consider. Your doctor can help determine if it’s the right choice for your cancer.
What Is a Clinical Trial and Why Does It Matter?
A clinical trial is a research study designed to test new drugs and treatments in patients to determine their safety and effectiveness. Before Sotorasib was approved, it went through multiple phases of clinical trials to assess how well it worked, what side effects it caused, and whether it was better than existing treatments. Clinical trials are essential because they provide scientific evidence that a drug can help patients while ensuring it is safe for widespread use.
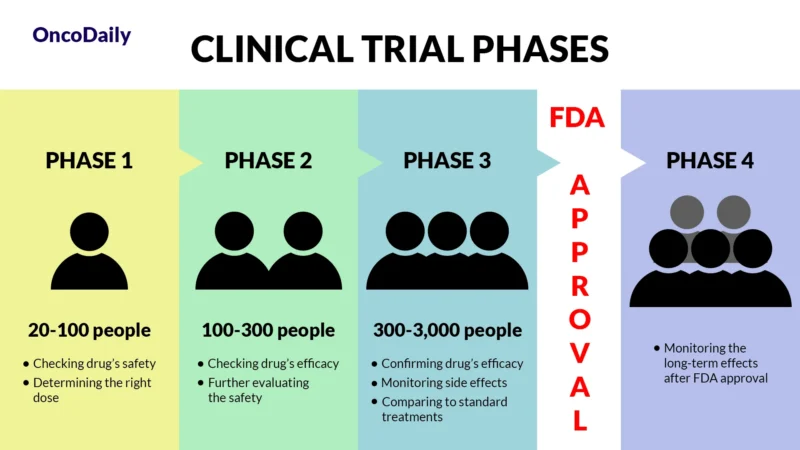
What Does FDA Approval Mean?
When a drug receives FDA approval, it means that after rigorous testing in clinical trials, it has been shown to be both safe and effective for treating a specific condition. This approval makes the drug widely available for doctors to prescribe and helps patients access new, cutting-edge treatments sooner.
Efficacy and Results from Clinical Trials
Sotorasib’s effectiveness has been demonstrated through several important clinical trials:
CodeBreaK 100 (NSCLC – Non-Small Cell Lung Cancer)
In this trial, 37.1% of patients experienced tumor shrinkage, and the duration of response lasted for an average of 11.1 months. The progression-free survival (PFS), or the time patients lived without the cancer worsening, was 6.8 months.
CodeBreaK 300 (mCRC with Panitumumab – Metastatic Colorectal Cancer)
In this trial, patients who received Sotorasib plus panitumumab had a significantly longer PFS of 5.6 months compared to just 2 months with the standard treatment. The objective response rate (ORR), or the percentage of patients whose tumors shrank, was 26%.
These results highlight Sotorasib as a promising option for patients with KRAS G12C mutations, offering potential for improved outcomes in certain cancers.
Ongoing Trials with Sotorasib
There are several ongoing studies exploring new ways that Sotorasib might help patients:
- BrainMet ADePPT Trial (NCT06807619): This study is testing whether Sotorasib can effectively treat brain metastases (cancer spread to the brain) in patients with KRAS G12C mutations. The goal is to see if Sotorasib can reach and work in the brain to shrink tumors.
- NCT06333678: This trial is looking at whether switching from the immunotherapy drug Durvalumab to Sotorasib in patients with locally advanced non-small cell lung cancer (NSCLC) and minimal remaining disease can improve treatment results.
These studies are important as they could expand Sotorasib’s use to help patients with brain metastases or provide better outcomes in NSCLC treatment.
What Can You Expect During Treatment?
Starting Sotorasib treatment may bring questions about what lies ahead. This targeted therapy works by blocking cancer growth and is taken as a daily pill. For some patients, it is combined with panitumumab, an IV infusion given under medical supervision. Regular monitoring, lifestyle adjustments, and open communication with healthcare providers help ensure the best possible outcome.
Sotorasib administration
Sotorasib comes in the form of a pill that is taken once a day. It can be taken with or without food, offering flexibility for patients. Taking it at the same time each day can help ensure consistent use and keep the treatment on track.
Combination with Panitumumab
For patients with metastatic colorectal cancer (mCRC), Sotorasib is often combined with panitumumab, which is given through an intravenous (IV) infusion. These infusions are usually done in a clinic or hospital setting, where healthcare providers will monitor the patient during the procedure to ensure their safety and comfort.
Missed Dose Guidance
If a dose is missed and more than six hours have passed, the patient should skip that dose and take the next one as scheduled. It’s important not to take an extra dose to make up for the missed one. If the patient vomits shortly after taking a dose, they should not retake it. Instead, they should continue with the next scheduled dose.
Monitoring and Adjustments
Throughout treatment, healthcare providers will monitor the patient’s progress, checking for side effects and adjusting the treatment as needed. Regular blood tests, scans, and checkups will help ensure that Sotorasib is working as expected. If any side effects arise, doctors will work with patients to manage them effectively.
Lifestyle Considerations
Patients are encouraged to maintain a healthy lifestyle while on treatment. Staying hydrated, eating a balanced diet, and getting enough rest can help manage side effects such as fatigue. If side effects become troublesome, patients should talk to their healthcare provider for advice on managing them.
By staying in close communication with their healthcare team, patients can navigate any challenges during treatment and ensure the best possible outcome. The healthcare team is there to provide ongoing support and make any necessary adjustments to the treatment plan.
Learn more about Immunotherapy for Lung Cancer on OncoDaily.
Side Effects and Management
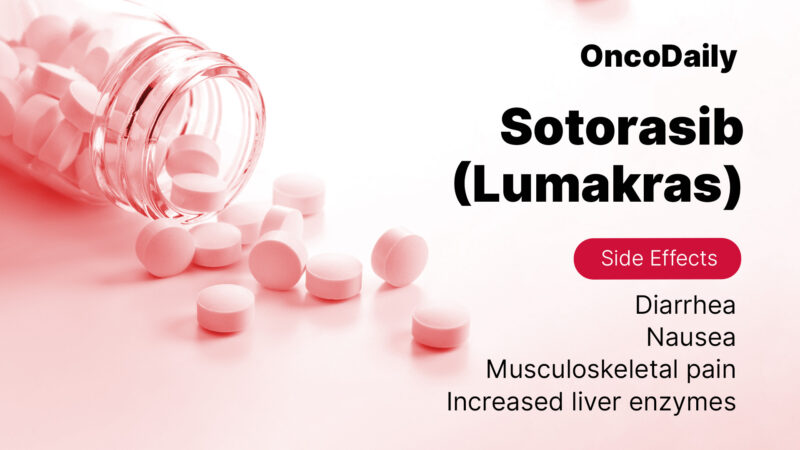
Sotorasib is generally well tolerated, but like any medication, it may cause some side effects. The most common side effects include diarrhea, nausea, fatigue, liver problems, and muscle pain. Diarrhea can be managed by staying hydrated and using anti-diarrheal medications such as loperamide. If patients experience nausea, antiemetic medications like ondansetron can help reduce symptoms. Fatigue is another common issue, but optimizing nutrition, getting enough rest, and engaging in light physical activity can make it more manageable.
Sotorasib can also affect liver function, so regular liver function tests (LFTs) are important to monitor any changes in liver enzymes. Muscle pain may occur, but it can typically be managed with over-the-counter pain relievers like acetaminophen or NSAIDs, unless contraindicated.
While most side effects are manageable, there are some serious side effects that require prompt attention. One of these is Interstitial Lung Disease (ILD) or pneumonitis, which can cause coughing or breathing difficulties. If symptoms of ILD appear, the patient should immediately stop the medication and receive corticosteroid treatment.
Another serious side effect is QT prolongation, which affects the heart’s rhythm. Patients who are at risk will need to have regular ECGs to monitor their heart’s condition. In rare cases, severe liver toxicity (when liver enzymes like AST/ALT rise beyond five times the normal limit) may occur, which may require discontinuing Sotorasib temporarily or permanently.
What to Avoid During Sotorasib Treatment
When taking Sotorasib, there are a few things to avoid to ensure the treatment works effectively. First, acid reducers like proton pump inhibitors (PPIs) or H2 blockers should be avoided because they can reduce how much of the medicine your body absorbs.
You should also be cautious with certain medications that affect how Sotorasib is processed in the body. Strong inducers (such as rifampin and phenytoin) can lower the level of Sotorasib in your system, making it less effective. On the other hand, strong inhibitors (like ketoconazole and clarithromycin) can increase Sotorasib levels, which might lead to unwanted side effects. Always inform your doctor about any other medications you’re taking.
Finally, grapefruit and grapefruit juice should be avoided, as they can interfere with the way your body processes Sotorasib, potentially affecting how well the medicine works.
Real-Life Effectiveness
Real-world studies have shown that Sotorasib can be highly effective in improving survival for patients with certain cancers. In one study, patients with advanced non-small cell lung cancer (NSCLC) who had already been treated with other therapies lived longer when treated with Sotorasib compared to those treated with docetaxel. The study found a median overall survival of 10.2 months for those on Sotorasib, compared to 6.0 months for those on docetaxel.
Moreover, benefits from Sotorasib were seen across various patient groups, even in those who had previously received immunotherapy. Additionally, a study presented at the ESMO GI 2024 conference showed that when Sotorasib was combined with panitumumab, it led to better response rates in patients with metastatic colorectal cancer (mCRC) who had not responded to previous treatments. These findings highlight the real-world potential of Sotorasib to improve outcomes for cancer patients.
If you’re considering Sotorasib for your treatment, discussing these factors with your doctor can help you get the most out of your therapy.
Looking Ahead – The Future of Sotorasib in Cancer Treatment
Research into Sotorasib is ongoing, and new studies are being conducted to explore its potential in treating more types of cancer. Scientists are testing Sotorasib for use in cancers like pancreatic cancer and other solid tumors to see if it can help even more patients.
In addition, researchers are studying how Sotorasib works when combined with other treatments, such as immune checkpoint inhibitors or chemotherapy. The goal is to improve patient outcomes and make treatment more effective.
Looking to the future, there’s hope that Sotorasib may be used earlier in cancer treatment, possibly as part of the first line of therapy for patients with KRAS G12C mutations. More research is needed to determine its full potential in this regard.
In conclusion, Sotorasib is a significant advancement in precision oncology, offering a new treatment option for patients with KRAS G12C-mutated cancers. By targeting this specific mutation, it helps slow cancer growth and is generally well tolerated by patients. If you or a loved one are considering Sotorasib as a treatment option, it’s important to talk with your oncologist to determine if it’s the right choice for your cancer care.
If you’re a healthcare provider, access the professional version here.
Written by Mariam Khachatryan, MD


