Sotorasib is a groundbreaking KRAS G12C inhibitor and the first targeted therapy for this mutation. Approved by the FDA in May 2021, it treats KRAS G12C-mutated non-small cell lung cancer (NSCLC) in previously treated patients. Marketed as Lumakras, it represents a major advancement in precision oncology, offering a new treatment option for patients with this aggressive mutation.
Which company produced Sotorasib?
Sotorasib (Lumakras) was developed by Amgen, a leading biotechnology company known for pioneering targeted therapies. Founded in 1980 and headquartered in California, Amgen specializes in innovative treatments for cancer, autoimmune diseases, and rare conditions. With Sotorasib, Amgen became the first to successfully target the KRAS G12C mutation, marking a breakthrough in precision oncology.
How does Sotorasib work?
Lumakras works by irreversibly binding to the KRAS G12C mutant in its inactive GDP-bound state, preventing it from switching back to the active form. KRAS is a key signaling protein that regulates cell growth and division. Under normal conditions, KRAS cycles between an active GTP-bound state and an inactive GDP-bound state, allowing controlled cell proliferation. However, in cancers with a KRAS G12C mutation, the protein becomes locked in the active state, continuously signaling uncontrolled tumor growth.
This blocks downstream signaling pathways, such as MAPK and PI3K, essential for cancer cell survival and proliferation. By shutting down these pathways, Sotorasib inhibits tumor growth and induces cancer cell death, offering a targeted approach to treating KRAS G12C-mutated cancers.
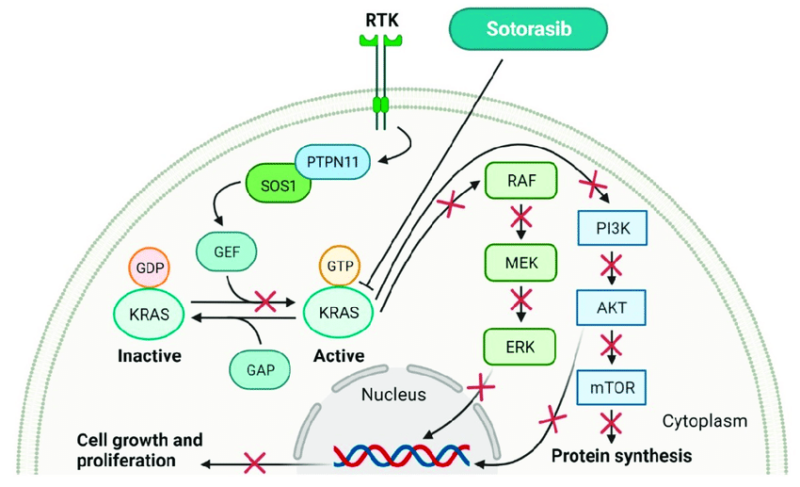
Sochacka-Ćwikła, Aleksandra & Mączyński, Marcin & Regiec, Andrzej. (2022). FDA-Approved Small Molecule Compounds as Drugs for Solid Cancers from Early 2011 to the End of 2021. Molecules. 27. 2259. 10.3390/molecules27072259.
What Cancers Is Sotorasib Approved to Treat?
Lumakras is FDA-approved to treat KRAS G12C-mutated cancers, including non-small cell lung cancer (NSCLC) and metastatic colorectal cancer (mCRC). It was first approved in May 2021 for patients with locally advanced or metastatic NSCLC who had received at least one prior systemic therapy, based on clinical trials demonstrating significant tumor shrinkage and durable responses. On January 16, 2025, the FDA expanded its approval to include KRAS G12C-mutated mCRC in combination with panitumumab (Vectibix) for patients who had previously undergone chemotherapy, offering a new targeted option for those with limited treatment choices.
What research is behind the approval?
The CodeBreaK 100 trial, published in the New England Journal of Medicine (NEJM) in September 2020, was a pivotal study leading to the FDA approval of Lumakras. This Phase 1/2 trial evaluated the drug’s safety and efficacy in KRAS G12C-mutated solid tumors, with particularly promising results in non-small cell lung cancer (NSCLC).
Among 126 previously treated NSCLC patients, Sotorasib achieved an objective response rate (ORR) of 37.1%, with a median duration of response of 11.1 months and progression-free survival of 6.8 months. The study demonstrated durable clinical benefit, establishing Sotorasib as the first targeted therapy for KRAS G12C-mutated NSCLC.
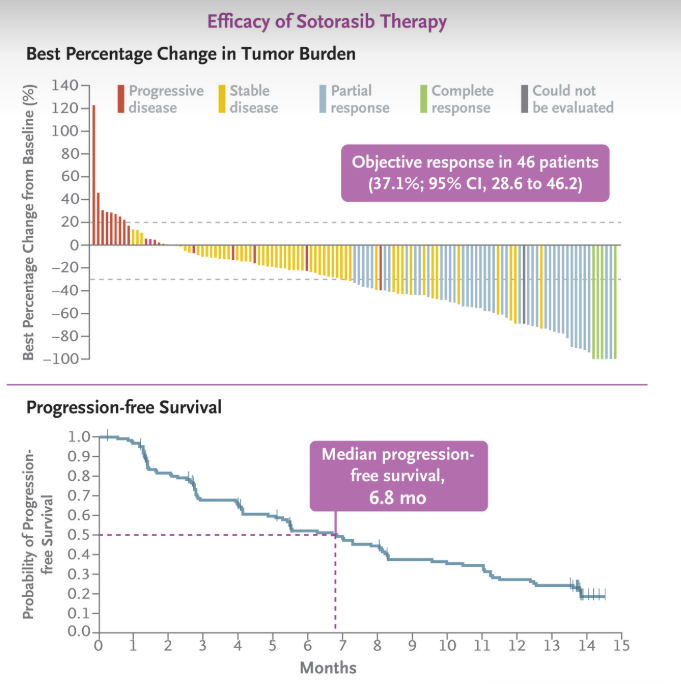
Sotorasib Combinations and Treatment Outcomes
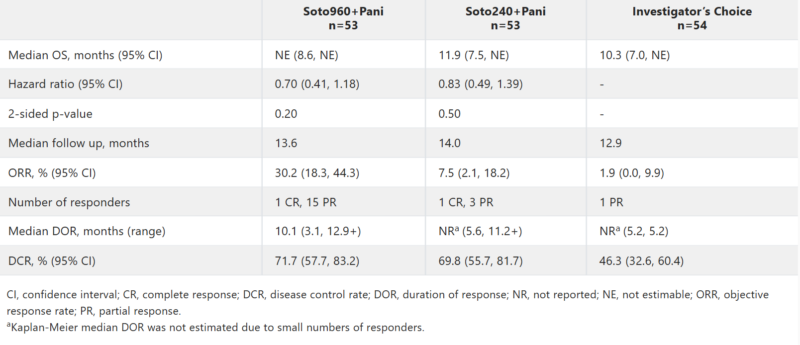
Sotorasib (Lumakras), in combination with panitumumab (Vectibix), was FDA-approved on January 16, 2025, for KRAS G12C-mutated metastatic colorectal cancer (mCRC) after prior chemotherapy, based on the CodeBreaK 300 Phase III trial. The results were published in the Journal of Clinical Oncology on June 5, 2024 (JCO).
In the trial, 160 patients with chemorefractory KRAS G12C-mutated mCRC were assigned to receive either Lumakras (960 mg) plus panitumumab, sotorasib (240 mg) plus panitumumab, or standard care. The combination of sotorasib 960 mg and panitumumab showed a median PFS of 5.6 months, significantly better than 2 months for standard care (HR 0.48, p=0.005). The objective response rate (ORR) was 26%. While overall survival (OS) data were not statistically significant, the median OS for the combination group was not reached, compared to 10.3 months for standard care. These results suggest Lumakras plus panitumumab as a potential new treatment for chemorefractory KRAS G12C-mutated mCRC.
Sotorasib side effects and its management
Sotorasib (Lumakras) is generally well tolerated, but like all targeted therapies, it has potential side effects. The most common adverse reactions include gastrointestinal issues, liver enzyme elevations, and respiratory complications.
Common Side Effects
Like most targeted therapies, Sotorasib is generally well tolerated, but patients may experience certain adverse effects.
- Diarrhea – One of the most commonly reported side effects. Patients should maintain proper hydration, adopt dietary modifications such as a low-fiber diet, and use anti-diarrheal medications like loperamide as needed.
- Nausea – Can significantly impact quality of life but is manageable with antiemetics such as ondansetron or metoclopramide. Patients should eat smaller, more frequent meals and avoid triggers like strong odors.
- Fatigue – This can affect daily activities, making rest and optimized nutrition essential. Encouraging moderate physical activity when possible can also help alleviate fatigue.
- Hepatotoxicity (Elevated AST/ALT Levels) – Regular monitoring of liver function tests (LFTs) is recommended. If significant elevations occur, dose adjustments or temporary discontinuation may be necessary.
- Musculoskeletal Pain – Patients may experience joint or muscle pain, which can be managed with acetaminophen or nonsteroidal anti-inflammatory drugs (NSAIDs) unless contraindicated.
Serious Side Effects
While uncommon, some adverse effects require immediate medical attention to prevent serious complications.
- Interstitial Lung Disease (ILD) or Pneumonitis – Patients should be closely monitored for symptoms such as new or worsening cough, dyspnea, or fever. If ILD is suspected, immediate drug discontinuation and corticosteroid therapy are necessary to mitigate severe respiratory distress.
- QT Prolongation – Sotorasib may cause QT interval prolongation in patients with predisposing factors. Routine ECG monitoring is advised, especially in those with preexisting cardiac conditions or concurrent QT-prolonging medications.
- Severe Hepatotoxicity (Grade 3-4) – If AST/ALT levels rise beyond five times the upper limit of normal (ULN), treatment should be held or permanently discontinued based on severity. Supportive care and close liver function monitoring are essential to prevent further complications.
Patients on Sotorasib should be closely monitored for these common and serious side effects. Regular check-ups and proactive management, including dose adjustments or medication changes, can help ensure the therapy remains effective and tolerable. Always consult with a healthcare provider for the best course of action if side effects arise.
What is the Recommended Dosage of Sotorasib?
Lumakras is available in 120 mg, 240 mg, and 320 mg tablets. The recommended dose for both KRAS G12C-mutated NSCLC and metastatic colorectal cancer (mCRC) is 960 mg orally once daily until disease progression or unacceptable toxicity. In mCRC, it is used in combination with panitumumab, and dosing for panitumumab should follow its prescribing guidelines.
How is Sotorasib administered?
Sotorasib is taken once daily at the same time, with or without food, and must be swallowed whole. When combined with panitumumab, the first dose of Lumakras should be taken before the initial infusion. If a dose is missed by more than six hours, it should be skipped, and the next scheduled dose should be taken as usual. Vomited doses should not be retaken.
For patients with swallowing difficulties, sotorasib tablets can be dispersed in 120 mL of noncarbonated water, stirred for three minutes, and consumed immediately or within two hours. The medication should be stored at 20-25°C (68-77°F), with short-term allowances between 15-30°C (59-86°F) to maintain its stability and effectiveness.
By targeting the KRAS G12C mutation, Lumakras has revolutionized treatment options for NSCLC and mCRC patients, offering a promising precision medicine approach for those with previously limited therapeutic alternatives.
Learn more about Immunotherapy for Colon Cancer: Types, Success Rate, Side Effects on OncoDaily.
What to Avoid During Sotorasib Treatment?
During Sotorasib treatment, avoid acid-reducing agents like proton pump inhibitors and H2 blockers, as they may reduce drug absorption. If needed, use antacids or adjust timing. Avoid strong CYP3A inducers (e.g., rifampin, phenytoin) as they lower Sotorasib levels and CYP3A inhibitors (e.g., ketoconazole, clarithromycin) as they increase toxicity. Sotorasib may also reduce the effectiveness of drugs like dabrafenib and apixaban. Grapefruit and grapefruit juice should be avoided due to potential interactions. Always consult a healthcare provider before changing medications.
Sotorasib effectiveness over time
A real-world study published in Elsevier (November 2024) found that Lumakras improved survival over docetaxel in pretreated KRAS G12C-mutated NSCLC. Median overall survival was 10.2 vs. 6.0 months in the second line (HR 0.62) and 10.2 vs. 7.2 months in the second line and beyond (HR 0.65). Benefits were consistent across subgroups, including those previously treated with anti-PD-(L)1 therapy.
Presented at ESMO GI 2024 on June 27, this MAIC analysis compared Lumakras + panitumumab to T/T + bevacizumab in chemorefractory KRAS G12C-mutated mCRC. Sotorasib showed higher response rates (OR 6.0) and a trend toward improved PFS (HR 0.85) with fewer Grade 3+ AEs (51% vs. 72%). OS data is pending.
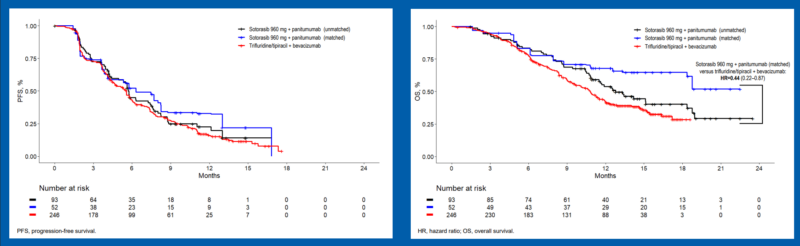
On January 16, 2025, the FDA approved Lumakras+ panitumumab for KRAS G12C-mutated mCRC based on CodeBreaK 300 (ESMO Annals of Oncology). The combo improved PFS (5.6 vs. 2 months, HR 0.48, p=0.005) with a 26% ORR. Common side effects included rash, diarrhea, and fatigue.
Ongoing trials with Sotorasib
The BrainMet ADePPT trial (NCT06807619) is studying how well Sotorasib penetrates KRAS G12C+ brain tumors. It also evaluates its safety and effectiveness in patients undergoing surgical resection of metastatic brain tumors, along with how the body processes the drug.
The NCT06333678 trial is evaluating whether switching from Durvalumab to Lumakras in patients with KRAS G12C+ locally advanced NSCLC (LA-NSCLC) with detectable minimal residual disease (MRD) improves outcomes. Patients who complete chemoradiation and have persistent MRD will be randomized to either continue Durvalumab or switch to Lumakras. The study aims to determine if Sotorasib can prolong progression-free survival (PFS) compared to ongoing Durvalumab treatment.
Written by Mariam Khachatryan, MD
FAQ
What is Lumakras used for?
Sotorasib is primarily used to treat KRAS G12C-mutated non-small cell lung cancer (NSCLC) and KRAS G12C-mutated metastatic colorectal cancer (mCRC) after prior treatment. It targets a mutation that drives cancer growth, offering a personalized treatment option.
How does Sotorasib differ from chemotherapy?
Unlike traditional chemotherapy, which kills both healthy and cancerous cells, Sotorasib is a targeted therapy. It specifically binds to the KRAS G12C mutation, stopping cancer growth while minimizing damage to normal cells.
What are the most common side effects of Sotorasib?
Common side effects include diarrhea, nausea, fatigue, liver enzyme elevations, and musculoskeletal pain. Serious but rare side effects include lung inflammation (pneumonitis) and QT prolongation.
What should I avoid while taking Lumakras?
Patients should avoid proton pump inhibitors (PPIs), CYP3A inducers and inhibitors, grapefruit, and grapefruit juice, as they can interfere with drug absorption and effectiveness.
Can Sotorasib be taken with other cancer drugs?
Yes, Sotorasib is now FDA-approved in combination with panitumumab (Vectibix) for KRAS G12C-mutated colorectal cancer. Clinical trials are also testing combinations with immunotherapy and other targeted therapies.


