Photo from Paolo Tarantino/LinkedIn
Apr 8, 2024, 03:54
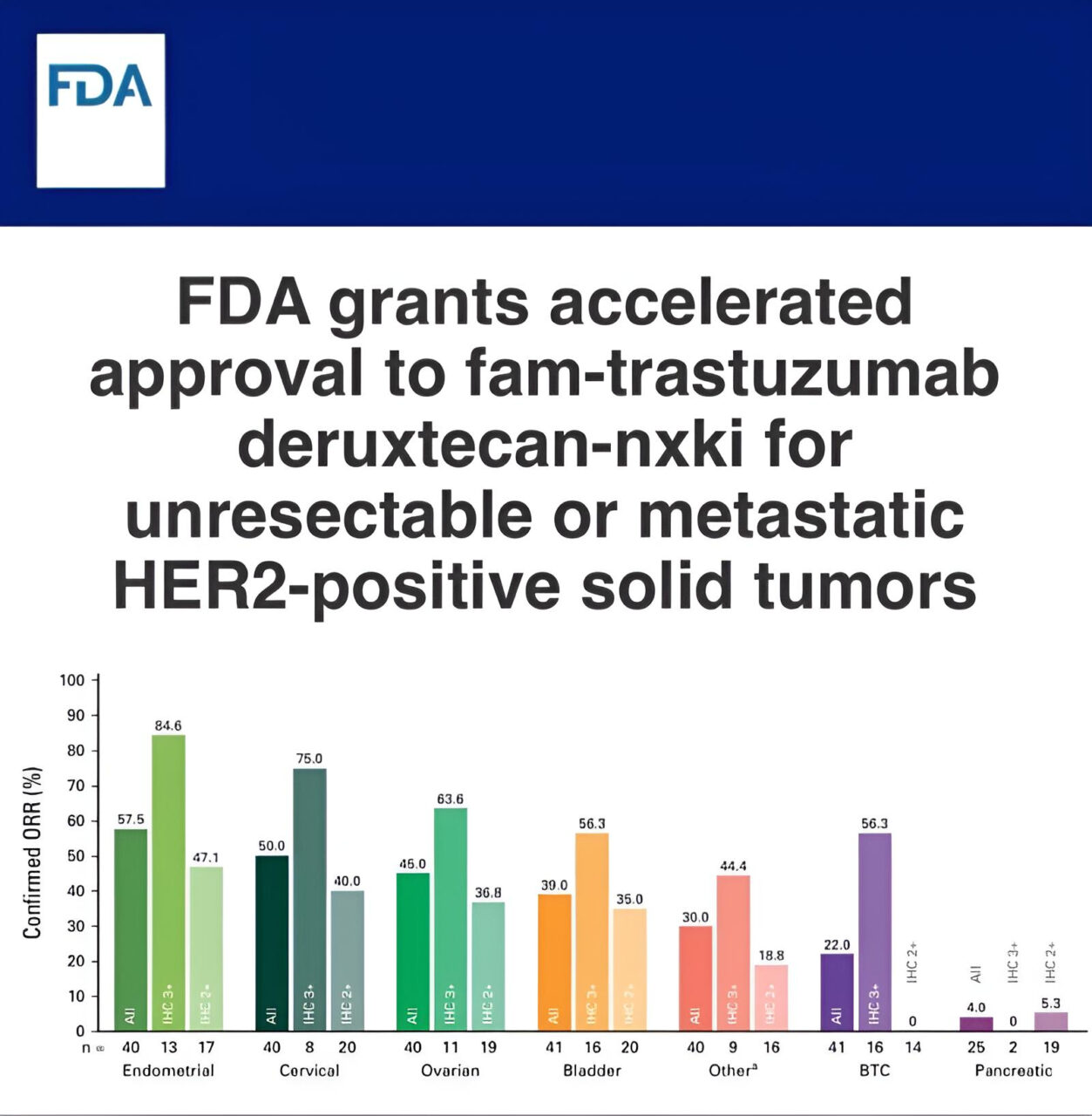
Paolo Tarantino: Trastuzumab deruxtecan is now FDA approved for ANY treatment-refractory HER2+ solid tumor
Paolo Tarantino, Advanced Research Fellow in the Breast Oncology Program at Dana-Farber Cancer Institute, shared a post on LinkedIn:
“Trastuzumab deruxtecan (T-DXd) is now FDA approved for the treatment of patients with ANY treatment-refractory HER2+ (IHC 3+) solid tumor, making it the first agnostic ADC. Unlikely to be the last. First priority: ensuring that HER2 testing is expanded across cancer types. Exciting times!”
Read further.
Source: Paolo Tarantino/LinkedIn
